Abstract
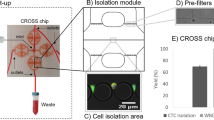
Molecular analysis of circulating tumor cells (CTCs) is hindered by low sensitivity and high level of background leukocytes of currently available CTC enrichment technologies. We have developed a novel device to enrich and retrieve CTCs from blood samples by using a microfluidic chip. The Celsee PREP100 device captures CTCs with high sensitivity and allows the captured CTCs to be retrieved for molecular analysis. It uses the microfluidic chip which has approximately 56,320 capture chambers. Based on differences in cell size and deformability, each chamber ensures that small blood escape while larger CTCs of varying sizes are trapped and isolated in the chambers. In this report, we used the Celsee PREP100 to capture cancer cells spiked into normal donor blood samples. We were able to show that the device can capture as low as 10 cells with high reproducibility. The captured CTCs were retrieved from the microfluidic chip. The cell recovery rate of this back-flow procedure is 100% and the level of remaining background leukocytes is very low (about 300–400 cells). RNA from the retrieved cells are extracted and converted to cDNA, and gene expression analysis of selected cancer markers can be carried out by using RT-PCR assays. The sensitive and easy-to-use Celsee PREP100 system represents a promising technology for capturing and molecular characterization of CTCs.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Pantel K, Alix-Panabieres C (2010) Circulating tumor cells in cancer patients: challenges and perspectives. Trends Mol Med 16(9):398–406
Maheswaran S, Haber DA (2010) Circulating tumor cells: a window into cancer biology and metastasis. Curr Opin Genet Dev 20(1):96–99
Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Reuben JM et al (2004) Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 351(8):781–791
Punnoose EA, Atwal SK, Spoerke JM, Savage H, Pandita A, Yeh RF et al (2010) Molecular biomarker analyses using circulating tumor cells. PLoS One 5(9):e12571
Allard WJ, Matera J, Miller MC, Repollet M, Connelly MC, Rao C et al (2004) Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res 10(20):6897–6904
Riethdorf S, Fritsche H, Muller V, Rau T, Schindlbeck C, Rack B et al (2007) Detection of circulating tumor cells in peripheral blood of patients with metastatic breast cancer: a validation study of the CellSearch system. Clin Cancer Res 13(3):920–928
Nagrath S, Sequist LV, Maheswaran S, Bell DW, Irimia D, Ulkus L et al (2007) Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 450(7173):1235–1239
Khoo BL, Warkiani ME, Tan DS, Bhagat AA, Irwin D, Lau DP et al (2014) Clinical validation of an ultrahigh-throughput spiral microfluidics for the detection and enrichment of viable circulating tumor cells. PLoS One 9(7):e99409
Stott SL, Hsu C-H, Tsukrov DI, Yu M, Miyamoto DT, Waltman BA et al (2010) Isolation of circulating tumor cells using a microvortex-generating herringbone-chip. Proc Natl Acad Sci U S A 107(43):18392–18397
Saliba AE, Saias L, Psychari E, Minc N, Simon D, Bidard FC et al (2010) Microfluidic sorting and multimodal typing of cancer cells in self-assembled magnetic arrays. Proc Natl Acad Sci U S A 107(33):14524–14529
Powell AA, Talasaz AH, Zhang H, Coram MA, Reddy A, Deng G et al (2012) Single cell profiling of circulating tumor cells: transcriptional heterogeneity and diversity from breast cancer cell lines. PLoS One 7(5):e33788
Sieuwerts AM, Kraan J, Bolt-de Vries J, van der Spoel P, Mostert B, Martens JW et al (2009) Molecular characterization of circulating tumor cells in large quantities of contaminating leukocytes by a multiplex real-time PCR. Breast Cancer Res Treat 118(3):455–468
Riahi R, Gogoi P, Sepehri S, Zhou Y, Handique K, Godsey J et al (2014) A novel microchannel-based device to capture and analyze circulating tumor cells (CTCs) of breast cancer. Int J Oncol 44(6):1870–1878
Gogoi P, Sepehri S, Zhou Y, Gorin M, Paolillo C, Capoluongo E et al (2016) Development of an automated and sensitive microfluidic device for capturing and characterizing circulating tumor cells (CTCs) from clinical blood samples. PLoS One 11(1):e0147400
Acknowledgment
We would like to thank Harris Soifer, Ranelle Salunga, and Cathy Schnabel from Biotheranostics, Inc for their excellent technical support and critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Gogoi, P., Sepehri, S., Chow, W., Handique, K., Wang, Y. (2017). Microfluidic-Based Enrichment and Retrieval of Circulating Tumor Cells for RT-PCR Analysis. In: M. Magbanua, M., W. Park, J. (eds) Circulating Tumor Cells. Methods in Molecular Biology, vol 1634. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7144-2_4
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7144-2_4
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7143-5
Online ISBN: 978-1-4939-7144-2
eBook Packages: Springer Protocols