Abstract
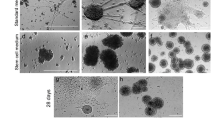
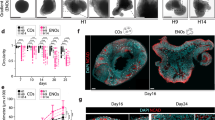
In the developing embryo, telencephalon arises from the rostral portion of the neural tube. The telencephalon further subdivides into distinct brain regions along the dorsal-ventral (DV) axis by exogenous patterning signals. Here, we describe a protocol for in vitro generation of various telencephalic regions from human embryonic stem cells (ESCs). Dissociated human ESCs are reaggregated in a low-cell-adhesion 96-well plate and cultured as floating aggregates. Telencephalic neural progenitors are efficiently generated when ESC aggregates are cultured in serum-free medium containing TGFβ inhibitor and Wnt inhibitor. In long-term culture, the telencephalic neural progenitors acquire cortical identities and self-organize a stratified cortical structure as seen in human fetal cortex. By treatment with Shh signal, the telencephalic progenitors acquire ventral (subpallial) identities and generate lateral ganglionic eminence (LGE) and medial ganglionic eminence (MGE). In contrast, by treatment with Wnt and BMP signals, their regional identities shift to more dorsal side that generates choroid plexus and medial palllium (hippocampal primordium).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Wilson SW, Houart C (2004) Early steps in the development of the forebrain. Dev Cell 6(2):167–181
Wonders CP, Anderson SA (2006) The origin and specification of cortical interneurons. Nat Rev Neurosci 7(9):687–696
Monuki ES, Porter FD, Walsh CA (2001) Patterning of the dorsal telencephalon and cerebral cortex by a roof plate-Lhx2 pathway. Neuron 32(4):591–604
Lehtinen MK, Bjornsson CS, Dymecki SM et al (2013) The choroid plexus and cerebrospinal fluid: emerging roles in development, disease, and therapy. J Neurosci 33(45):17553–17559. doi:10.1523/JNEUROSCI.3258-13.2013
Bystron I, Blakemore C, Rakic P (2008) Development of the human cerebral cortex: Boulder Committee revisited. Nat Rev Neurosci 9(2):110–122. doi:10.1038/nrn2252
Tole S, Christian C, Grove EA (1997) Early specification and autonomous development of cortical fields in the mouse hippocampus. Development 124(24):4959–4970
Sousa VH, Fishell G (2010) Sonic hedgehog functions through dynamic changes in temporal competence in the developing forebrain. Curr Opin Genet Dev 20(4):391–399. doi:10.1016/j.gde.2010.04.008
Sigurdsson T, Duvarci S (2015) Hippocampal-prefrontal interactions in cognition, behavior and psychiatric disease. Front Syst Neurosci 9:190. doi:10.3389/fnsys.2015.00190
Eiraku M, Sasai Y (2012) Self-formation of layered neural structures in three-dimensional culture of ES cells. Curr Opin Neurobiol 22(5):768–777. doi:10.1016/j.conb.2012.02.005
Sasai Y (2013) Next-generation regenerative medicine: organogenesis from stem cells in 3D culture. Cell Stem Cell 12(5):520–530. doi:10.1016/j.stem.2013.04.009
Sasai Y (2013) Cytosystems dynamics in self-organization of tissue architecture. Nature 493(7432):318–326. doi:10.1038/nature11859
Eiraku M, Watanabe K, Matsuo-Takasaki M et al (2008) Self-organized formation of polarized cortical tissues from ESCs and its active manipulation by extrinsic signals. Cell Stem Cell 3(5):519–532. doi:10.1016/j.stem.2008.09.002
Watanabe M, Kang YJ, Davies LM et al (2012) BMP4 sufficiency to induce choroid plexus epithelial fate from embryonic stem cell-derived neuroepithelial progenitors. J Neurosci 32(45):15934–15945. doi:10.1523/JNEUROSCI.3227-12.2012
Kadoshima T, Sakaguchi H, Nakano T et al (2013) Self-organization of axial polarity, inside-out layer pattern, and species-specific progenitor dynamics in human ES cell-derived neocortex. Proc Natl Acad Sci U S A 110:20284–20289. doi:10.1073/pnas.1315710110
Lancaster MA, Renner M, Martin CA et al (2013) Cerebral organoids model human brain development and microcephaly. Nature 501(7467):373–379. doi:10.1038/nature12517
Sakaguchi H, Kadoshima T, Soen M et al (2015) Generation of functional hippocampal neurons from self-organizing human embryonic stem cell-derived dorsomedial telencephalic tissue. Nat Commun 6:8896. doi:10.1038/ncomms9896
Watanabe K, Kamiya D, Nishiyama A et al (2005) Directed differentiation of telencephalic precursors from embryonic stem cells. Nat Neurosci 8:288–296
Watanabe K, Ueno M, Kamiya D et al (2007) A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat Biotechnol 25:681–686
Wataya T, Ando S, Muguruma K et al (2008) Minimization of exogenous signals in ES cell culture induces rostral hypothalamic differentiation. Proc Natl Acad Sci U S A 105(33):11796–11801. doi:10.1073/pnas.0803078105
Danjo T, Eiraku M, Muguruma K et al (2011) Subregional specification of embryonic stem cell-derived ventral telencephalic tissues by timed and combinatory treatment with extrinsic signals. J Neurosci 31(5):1919–1933. doi:10.1523/JNEUROSCI.5128-10.2011
Nasu M, Takata N, Danjo T et al (2012) Robust formation and maintenance of continuous stratified cortical neuroepithelium by laminincontaining matrix in mouse ES cell culture. PLoS One 7:e53024. doi:10.1371/journal.pone.0053024
Levine AJ, Brivanlou AH (2007) Proposal of a model of mammalian neural induction. Dev Biol 308(2):247–256
Fuccillo M, Rallu M, McMahon AP et al (2004) Temporal requirement for hedgehog signaling in ventral telencephalic patterning. Development 131(20):5031–5040
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Kadoshima, T., Sakaguchi, H., Eiraku, M. (2017). Generation of Various Telencephalic Regions from Human Embryonic Stem Cells in Three-Dimensional Culture. In: Tsuji, T. (eds) Organ Regeneration. Methods in Molecular Biology, vol 1597. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6949-4_1
Download citation
DOI: https://doi.org/10.1007/978-1-4939-6949-4_1
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-6947-0
Online ISBN: 978-1-4939-6949-4
eBook Packages: Springer Protocols