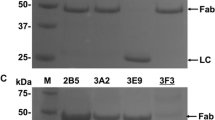
Abstract
Molecular and chemical chaperones /foldases can strongly contribute to improve the amounts and the structural quality of recombinant proteins. Several methodologies have been proposed to optimize their beneficial effects. This chapter presents a condensed summary of the biotechnological opportunities offered by this approach followed by a protocol describing the method we use for expressing disulfide bond-dependent recombinant antibodies in the cytoplasm of bacteria engineered to overexpress sulfhydryl oxidase and DsbC isomerase. The system is based on the possibility to trigger the foldase expression independently and before the induction of the target protein. As a consequence, the recombinant antibody synthesis starts only after enough foldases have accumulated to promote correct folding of the antibody.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Anfinsen CB (1972) The formation and stabilization of protein structure. Biochem J 128:737–749
Taipale M, Krykbaeva I, Koeva M et al (2012) Quantitative analysis of HSP90-client interactions reveals principles of substrate recognition. Cell 150:987–1001
Kerner MJ, Naylor DJ, Ishihama Y et al (2005) Proteome-wide analysis of chaperonin-dependent protein folding in Escherichia coli. Cell 122:209–220
de Marco A, Vigh L, Diamant S et al (2005) Native folding of aggregation-prone recombinant proteins in Escherichia coli by osmolytes, plasmid- or benzyl alcohol-overexpressed molecular chaperones. Cell Stress Chaperones 10:329–339
de Marco A, Deuerling E, Mogk A et al (2007) Chaperone-based procedure to increase yields of soluble recombinant proteins produced in E. coli. BMC Biotechnol 7:32
de Marco A (2007) Protocol for preparing proteins with improved solubility by co-expression with molecular chaperones in Escherichia coli. Nat Protoc 2:2632–2639
Diamant S, Rosenthal D, Azem A (2003) Dicarboxylic amino acids and glycine-betaine regulate chaperone-mediated protein disaggregation under stress. Mol Microbiol 49:401–410
Schultz T, Liu J, Capasso P et al (2007) The solubility of recombinant proteins expressed in Escherichia coli is increased by otsA and otsB co-transformation. Biochem Biophys Res Commun 355:234–239
Bandyopadhyay A, Saxena K, Kasturia N et al (2012) Chemical chaperones assist intracellular folding to buffer mutational variations. Nat Chem Biol 8:238–245
Diamant S, Eliahu N, Rosenthal D et al (2001) Chemical chaperones regulate molecular chaperones in vitro and in cells under combined salt and heat stresses. J Biol Chem 276:39586–39591
de Marco A (2014) Osmolytes as chemical chaperones to use in protein biotechnology. In: Doglia SM, Lotti M (eds) Protein aggregation in bacteria: functional and structural properties of inclusion bodies in bacterial cells. Wiley, Hoboken, NJ, pp 77–92
Esposito D, Chatterjee DK (2006) Enhancement of soluble protein expression through the use of fusion tags. Curr Opin Biotechnol 17:353–358
Swalley SE, Fulghum JR, Chambers SP (2006) Screening factors effecting a response in soluble protein expression: formalized approach using design of experiments. Anal Biochem 351:122–127
Bora N, Bawa Z, Bill RM et al (2012) The implementation of a design of experiments strategy to increase recombinant protein yields in yeast (review). Methods Mol Biol 866:115–127
Nozach H, Fruchart-Gaillard C, Fenaille F (2013) High throughput screening identifies disulfide isomerase DsbC as a very efficient partner for recombinant expression of small disulfide-rich proteins in E. coli. Microb Cell Fact 12:37
Djender S, Schneider A, Beugnet A et al (2014) Bacterial cytoplasm as an effective cell compartment for producing functional VHH-based affinity reagents and Camelidae IgG-like recombinant antibodies. Microb Cell Fact 13:140
Veggiani G, de Marco A (2011) Improved quantitative and qualitative production of single-domain intrabodies mediated by the co-expression of Erv1p sulfhydryl oxidase. Protein Expr Purif 79:111–114
Raynal B, Lenormand P, Baron B et al (2014) Quality assessment and optimization of purified protein samples: why and how? Microb Cell Fact 13:180
Nguyen VD, Hatahet F, Salo KE et al (2011) Pre-expression of a sulfhydryl-oxidase significantly increases the yields of eukaryotic disulfide bond containing proteins expressed in the cytoplasm of E. coli. Microb Cell Fact 10:1
Liu JL, Zabetakis D, Walper SA et al (2014) Bioconjugates of rhizavidin with single domain antibodies as bifunctional immunoreagents. J Immunol Methods 411:37–42
de Marco A (2015) Recombinant antibody production evolves into multiple options aimed at yielding reagents suitable for application-specific needs. Microb Cell Fact 14:125
Sala E, de Marco A (2010) Screening optimized protein purification protocols by coupling small-scale expression and mini-size exclusion chromatography. Protein Expr Purif 74:231–235
Nominé Y, Ristriani T, Laurent C et al (2001) A strategy for optimizing the monodispersity of fusion proteins: application to purification of recombinant HPV E6 oncoprotein. Protein Eng 14:297–305
Capasso P, Aliprandi M, Ossolengo G et al (2009) Monodispersity of recombinant Cre recombinase correlates with its effectiveness in vivo. BMC Biotechnol 9:80
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
de Marco, A. (2017). Acting on Folding Effectors to Improve Recombinant Protein Yields and Functional Quality. In: Burgess-Brown, N. (eds) Heterologous Gene Expression in E.coli. Methods in Molecular Biology, vol 1586. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6887-9_12
Download citation
DOI: https://doi.org/10.1007/978-1-4939-6887-9_12
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-6885-5
Online ISBN: 978-1-4939-6887-9
eBook Packages: Springer Protocols