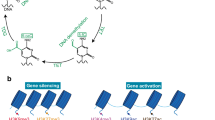
Abstract
Regulation of gene expression is essential for the differentiation of pluripotent precursor cells into specialized effector cells and, thus, for the evolution of multiorgan systems. The regulation of gene expression is controlled by a variety of “extra-genic” mechanisms, termed as epigenetic mechanisms. Obviously, alterations in such control mechanisms of gene expression may result in alterations of cellular effector functions, resulting in for example defects in cellular functions, but also, if immune cells are involved, leading to the development of immunologic disorders such as malignancies and autoimmune diseases. The analysis of epigenetic modifications is therefore pertinent not only for the understanding of the regular function of the immune system, but also for the understanding of the pathophysiology of such diseases. As interleukin-9 (IL-9) is the signature cytokine for Th9 cells, and since IL-9 plays important roles in the orchestration of a protective immune responses, the analysis of epigenetic mechanisms underlying the development of IL-9 producing effector T cells is of great interest. Here, we describe a protocol to analyze epigenetic regulation of the IL-9 gene by chromatin immunoprecipitation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Holliday R (2006) Epigenetics: a historical overview. Epigenetics 1(2):76–80
Rodriguez RM, Lopez-Larrea C, Suarez-Alvarez B (2015) Epigenetic dynamics during CD4(+) T cells lineage commitment. Int J Biochem Cell Biol 67:75–85. doi:10.1016/j.biocel.2015.04.020
Ramming A, Druzd D, Leipe J, Schulze-Koops H, Skapenko A (2012) Maturation-related histone modifications in the PU.1 promoter regulate Th9-cell development. Blood 119(20):4665–4674. doi:10.1182/blood-2011-11-392589
Santos P, Arumemi F, Park KS, Borghesi L, Milcarek C (2011) Transcriptional and epigenetic regulation of B cell development. Immunol Res 50(2–3):105–112. doi:10.1007/s12026-011-8225-y
MacDonald IA, Hathaway NA (2015) Epigenetic roots of immunologic disease and new methods for examining chromatin regulatory pathways. Immunol Cell Biol 93(3):261–270. doi:10.1038/icb.2014.105
Zhou Q, Haupt S, Kreuzer JT, Hammitzsch A, Proft F, Neumann C, Leipe J, Witt M, Schulze-Koops H, Skapenko A (2015) Decreased expression of miR-146a and miR-155 contributes to an abnormal Treg phenotype in patients with rheumatoid arthritis. Ann Rheum Dis 74(6):1265–1274. doi:10.1136/annrheumdis-2013-204377
Zhu J, Paul WE (2008) CD4 T cells: fates, functions, and faults. Blood 112(5):1557–1569. doi:10.1182/blood-2008-05-078154
Van Snick J, Goethals A, Renauld JC, Van Roost E, Uyttenhove C, Rubira MR, Moritz RL, Simpson RJ (1989) Cloning and characterization of a cDNA for a new mouse T cell growth factor (P40). J Exp Med 169(1):363–368
Uyttenhove C, Simpson RJ, Van Snick J (1988) Functional and structural characterization of P40, a mouse glycoprotein with T-cell growth factor activity. Proc Natl Acad Sci U S A 85(18):6934–6938
Dardalhon V, Awasthi A, Kwon H, Galileos G, Gao W, Sobel RA, Mitsdoerffer M, Strom TB, Elyaman W, Ho IC, Khoury S, Oukka M, Kuchroo VK (2008) IL-4 inhibits TGF-beta-induced Foxp3+ T cells and, together with TGF-beta, generates IL-9+ IL-10+ Foxp3(−) effector T cells. Nat Immunol 9(12):1347–1355
Beriou G, Bradshaw EM, Lozano E, Costantino CM, Hastings WD, Orban T, Elyaman W, Khoury SJ, Kuchroo VK, Baecher-Allan C, Hafler DA (2010) TGF-beta induces IL-9 production from human Th17 cells. J Immunol 185(1):46–54. doi:10.4049/jimmunol.1000356
Elyaman W, Bradshaw EM, Uyttenhove C, Dardalhon V, Awasthi A, Imitola J, Bettelli E, Oukka M, van Snick J, Renauld JC, Kuchroo VK, Khoury SJ (2009) IL-9 induces differentiation of TH17 cells and enhances function of FoxP3+ natural regulatory T cells. Proc Natl Acad Sci U S A 106(31):12885–12890
Soussi-Gounni A, Kontolemos M, Hamid Q (2001) Role of IL-9 in the pathophysiology of allergic diseases. J Allergy Clin Immunol 107(4):575–582. doi:10.1067/mai.2001.114238
Nowak EC, Weaver CT, Turner H, Begum-Haque S, Becher B, Schreiner B, Coyle AJ, Kasper LH, Noelle RJ (2009) IL-9 as a mediator of Th17-driven inflammatory disease. J Exp Med 206(8):1653–1660. doi:10.1084/jem.20090246
Lu LF, Lind EF, Gondek DC, Bennett KA, Gleeson MW, Pino-Lagos K, Scott ZA, Coyle AJ, Reed JL, Van Snick J, Strom TB, Zheng XX, Noelle RJ (2006) Mast cells are essential intermediaries in regulatory T-cell tolerance. Nature 442(7106):997–1002. doi:10.1038/nature05010
Acknowledgment
This work was supported by the Deutsche Forschungsgemeinschaft (Grants SK59/4-1, SK59/7-1, SCHU786/2-5, Schu786/8-1 and SCHU1683/9-1), by the DFG Sonderforschungsbereich Grant SFB571 (Autoimmunity), project D9, by the DFG Training Grant GK1202 (Oligonucleotides), project E2, and by the Verbundanträge “ArthroMark” (projects 1 and 7, OIEC100913 and 01EC1401B) and “Impam” (project 10, OIEC1008H) both by the Federal Ministry of Education and Research of Germany.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Skapenko, A., Schulze-Koops, H. (2017). Defining Epigenetic Regulation of the Interleukin-9 Gene by Chromatin Immunoprecipitation. In: Goswami, R. (eds) Th9 Cells. Methods in Molecular Biology, vol 1585. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6877-0_14
Download citation
DOI: https://doi.org/10.1007/978-1-4939-6877-0_14
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-6876-3
Online ISBN: 978-1-4939-6877-0
eBook Packages: Springer Protocols