Abstract
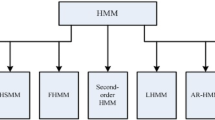
A number of real-world systems have common underlying patterns among them and deducing these patterns is important for us in order to understand the environment around us. These patterns in some instances are apparent upon observation while in many others especially those found in nature are well hidden. Moreover, the inherent stochasticity in these systems introduces sufficient noise that we need models capable to handling it in order to decipher the underlying pattern. Hidden Markov model (HMM) is a probabilistic model that is frequently used for studying the hidden patterns in an observed sequence or sets of observed sequences. Since its conception in the late 1960s it has been extensively applied in biology to capture patterns in various disciplines ranging from small DNA and protein molecules, their structure and architecture that forms the basis of life to multicellular levels such as movement analysis in humans. This chapter aims at a gentle introduction to the theory of HMM, the statistical problems usually associated with HMMs and their uses in biology.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ghahramani Z, (2001) An introduction to hidden Markov models and Bayesian networks. Int J Pattern Recognit Artif Intell 15(1): 9–42. doi: 10.1142/S0218001401000836
Baum LE, Petrie T (1966) Statistical inference for probabilistic functions of finite state Markov chains. Ann Math Statist 37(6):1554–1563. doi:10.1214/aoms/1177699147
Rabiner LR (1989) A tutorial on hidden Markov models and selected applications in speech recognition. Proc IEEE 77(2):257–286. doi:10.1109/5.18626
Schuster-Bockler B, Bateman A (2007) An introduction to hidden Markov models. Current protocols in bioinformatics/editoral board, Andreas D. Baxevanis [et al] Appendix 3:Appendix 3A. doi:10.1002/0471250953.bia03as18
Yoon BJ (2009) Hidden Markov models and their applications in biological sequence analysis. Curr Genomics 10(6):402–415
Durbin R, Eddy S, Krogh A, Mitchison G (2006) Biological sequence analysis. doi:citeulike-article-id:3346650
Krogh A, Larsson B, von Heijne G, Sonnhammer EL (2001) Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J Mol Biol 305(3):567–580. doi:10.1006/jmbi.2000.4315
Shukla D, Hernandez CX, Weber JK, Pande VS (2015) Markov state models provide insights into dynamic modulation of protein function. Acc Chem Res 48(2):414–422. doi:10.1021/ar5002999
Won KJ, Hamelryck T, Pruegel-Bennett A, Krogh A (2007) An evolutionary method for learning HMM structure: prediction of protein secondary structure. BMC Bioinformatics 8:357. doi:10.1186/1471-2105-8-357
Stanke M, Waack S (2003) Gene prediction with a hidden Markov model and a new intron submodel. Bioinformatics 19:Ii215–Ii225. doi:10.1093/bioinformatics/btg1080
Bian JW, Liu CL, Wang HY, Xing J, Kachroo P, Zhou XB (2013) SNVHMM: predicting single nucleotide variants from next generation sequencing. BMC Bioinformatics 14:225. doi:10.1186/1471-2105-14-225
Seiser EL, Innocenti F (2014) Hidden Markov model-Based CNV detection algorithms for illumina genotyping microarrays. Cancer Inform 13(Suppl 7):77–83. doi:10.4137/CIN.S16345
Bao Y, Vinciotti V, Wit E, t Hoen PA (2014) Joint modeling of ChIP-seq data via a Markov random field model. Biostatistics 15(2):296–310. doi:10.1093/biostatistics/kxt047
Okamoto K, Sako Y (2012) Variational Bayes analysis of a photon-based hidden Markov model for single-molecule FRET trajectories. Biophys J 103(6):1315–1324. doi:10.1016/j.bpj.2012.07.047
Arakawa T, Tanave A, Ikeuchi S, Takahashi A, Kakihara S, Kimura S, Sugimoto H, Asada N, Shiroishi T, Tomihara K, Tsuchiya T, Koide T (2014) A male-specific QTL for social interaction behavior in mice mapped with automated pattern detection by a hidden Markov model incorporated into newly developed freeware. J Neurosci Methods 234:127–134. doi:10.1016/j.jneumeth.2014.04.012
Karg M, Venture G, Hoey J, Kulic D (2014) Human movement analysis as a measure for fatigue: a hidden Markov-based approach. IEEE Trans Neural Syst Rehabil Eng 22(3):470–481. doi:10.1109/TNSRE.2013.2291327
Jr. GDF (2005) The Viterbi algorithm: a personal history. CoRR abs/cs/0504020
Viterbi A (1967) Error bounds for convolutional codes and an asymptotically optimum decoding algorithm. IEEE Trans Inform Theory 13(2):260–269. doi:10.1109/tit.1967.1054010
Welch LR (2003) Hidden Markov models and the Baum-Welch algorithm. IEEE Inform Theory Soc Newsl 53(4):10–13
Eddy SR (1996) Hidden markov models. Curr Opin Struct Biol 6(3):361–365
Eddy SR (1998) Profile hidden Markov models. Bioinformatics 14(9):755–763
Finn RD, Clements J, Arndt W, Miller BL, Wheeler TJ, Schreiber F, Bateman A, Eddy SR (2015) HMMER web server: 2015 update. Nucleic Acids Res 43(W1):W30–W38. doi:10.1093/nar/gkv397
Finn RD, Clements J, Eddy SR (2011) HMMER web server: interactive sequence similarity searching. Nucleic Acids Res 39(Web Server issue):W29–W37. doi:10.1093/nar/gkr367
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Vijayabaskar, M.S. (2017). Introduction to Hidden Markov Models and Its Applications in Biology. In: Westhead, D., Vijayabaskar, M. (eds) Hidden Markov Models. Methods in Molecular Biology, vol 1552. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6753-7_1
Download citation
DOI: https://doi.org/10.1007/978-1-4939-6753-7_1
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-6751-3
Online ISBN: 978-1-4939-6753-7
eBook Packages: Springer Protocols