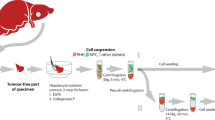
Abstract
For the purpose of clinical research of hepatocyte transplantation, procedures for isolation, cryopreservation, thawing, and functional assessment of hepatocytes are described. Although demands for human hepatocytes are increasing in not only cell therapy but also drug development, it is highly difficult to obtain good lots of hepatocytes from human liver tissue. This chapter describes essential issues such as alleviation of warm ischemia, prevention of shear stress, optimization of cryopreservation, and functional assessment, along with securement of quality. All procedures described here are compliant with good manufacturing procedure (GMP) in cell processing facility, approved by the act on measures to ensure safety of regenerative medicine and ethical regulations in Japan.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Enosawa S, Horikawa R, Yamamoto A, Sakamoto S, Shigeta T, Nosaka S et al (2014) Hepatocyte transplantation using the living donor reduced-graft in a baby with ornithine transcarbamylase deficiency: a novel source for hepatocytes. Liver Transpl 20:391–393
Brown HS, Griffin M, Houston JB (2007) Evaluation of cryopreserved human hepatocytes as an alternative in vitro system to microsomes for the prediction of metabolic clearance. Drug Metab Dispos 35:293–301
Ibars EP, Cortes M, Tolosa L, Gómez-Lechón MJ, López S, Castell JV, Mir J (2016) Hepatocyte transplantation program: Lessons learned and future strategies. World J Gastroenterol 22:874–886
Matsuno N, Enosawa S (2014) DCD for liver transplantation. Chapter 10 In: Asano T, Fukushima N, Kenmochi T, Matsuno N (eds) Marginal donors, current and future status, Springer, 1–282, 103–122
Kasahara M, Sakamoto S, Umeshita K, Uemoto S (2014) Effect of graft size matching on pediatric living-donor liver transplantation in Japan. Exp Clin Transplant 12:1–4
Hsu HC, Matsuno N, Machida N, Enosawa S (2013) Improvement of hepatocyte recovery from rat liver subjected to 1-hour warm ischemic injury by using citrate phosphate dextrose added to euro-collins perfusion solution. Transplant Proc 45:1700–1703. doi:10.1016/j.transproceed.2013.02.036
Bioreclamation IVT Product Sheet. Cryoplateable Hepatocytes. 02 SEP 2014 http://www.bioreclamationivt.com/sites/default/files/pdf/Instructions_Cryoplateable_%20Hepatocytes_10.pdf
Miyamoto Y, Suzuki S, Nomura K, Enosawa S (2006) Improvement of hepatocyte viability after cryopreservation by supplementation of long-chain oligosaccharide in the freezing medium in rats and humans. Cell Transplant 15:911–919
Enosawa S, Yuan W, Douzen M, Nakazawa A, Omasa T, Fukuda A et al (2012) Consideration of a safe protocol for hepatocyte transplantation using infantile pigs. Cell Medicine 3:13–18, doi: http://dx.doi.org/10.3727/215517912X639469
Enosawa S, Suzuki S, Kakefuda T, Amemiya H (1996) Examination of 7-ethoxycoumarin deethylation and ammonia removal activities in 31 hepatocyte cell lines. Cell Transplant 5:39–40
Acknowledgement
The author expresses sincere gratitude to all persons who donated liver tissue. The author would also like to thank Dr. Mureo Kasahara who proposed use of remnant liver tissues from living donor liver graft as a source of hepatocytes for transplantation.
The author declares no conflict of interest; no personal or financial interests and no source of financial support from any industry for this study and the manuscript.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media New York
About this protocol
Cite this protocol
Enosawa, S. (2017). Isolation of GMP Grade Human Hepatocytes from Remnant Liver Tissue of Living Donor Liver Transplantation. In: Stock, P., Christ, B. (eds) Hepatocyte Transplantation. Methods in Molecular Biology, vol 1506. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-6506-9_16
Download citation
DOI: https://doi.org/10.1007/978-1-4939-6506-9_16
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-6504-5
Online ISBN: 978-1-4939-6506-9
eBook Packages: Springer Protocols