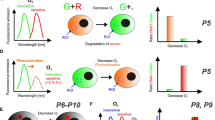
Abstract
Recent progress in bio-imaging has allowed detailed mechanistic studies of neural cell function in complex 3D tissue models including multicellular aggregates , neurospheres , excised brain slices , ganglia , and organoids . Molecular oxygen (O2 ) is an important metabolite and an environmental parameter which determines the viability and physiological status of neural cells within tissue. Here we describe standard method for monitoring O2 in 3D tissue models using phosphorescence lifetime imaging microscopy (PLIM ) and cell-penetrating O2-sensing probes . The O2 probes can be multiplexed with many conventional fluorescence based live cell biomarkers and also end-point immunofluorescence staining . The multi-parametric O2 imaging method is particularly useful for areas such as stem cell development and differentiation , hypoxia research, neurodegenerative disorders , regeneration of brain tissue, evaluation of new drugs , and development of novel tissue models.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Northington FJ, Chavez-Valdez R, Martin LJ (2011) Neuronal cell death in neonatal hypoxia-ischemia. Ann Neurol 69:743–758
Green DR, Galluzzi L, Kroemer G (2011) Mitochondria and the autophagy–inflammation–cell death axis in organismal aging. Science 333:1109–1112
Sims NR, Muyderman H (2010) Mitochondria, oxidative metabolism and cell death in stroke. Biochim Biophys Acta 1802:80–91
Kalyanaraman B, Darley-Usmar V, Davies KJA et al (2012) Measuring reactive oxygen and nitrogen species with fluorescent probes: challenges and limitations. Free Radic Biol Med 52:1–6
Tait SW, Green DR (2010) Mitochondria and cell death: outer membrane permeabilization and beyond. Nat Rev Mol Cell Biol 11:621–632
Jespersen SN, Østergaard L (2011) The roles of cerebral blood flow, capillary transit time heterogeneity, and oxygen tension in brain oxygenation and metabolism. JCBFM 32:264–277
Devor A, Sakadžić S, Srinivasan VJ et al (2012) Frontiers in optical imaging of cerebral blood flow and metabolism. JCBFM 32:1259–1276
Papkovsky DB, Dmitriev RI (2013) Biological detection by optical oxygen sensing. Chem Soc Rev 42:8700–8732
Lecoq J, Roussakis A, Parpaleix E et al (2011) Simultaneous two-photon imaging of oxygen and blood flow in deep cerebral vessels. Nat Med 17:893–898
Quaranta M, Borisov SM, Klimant I (2012) Indicators for optical oxygen sensors. Bioanal Rev 4:115–157
Dmitriev RI, Kondrashina AV, Koren K et al (2014) Small molecule phosphorescent probes for O2 imaging in 3D tissue models. Biomater Sci 2:853–866
Dmitriev RI, Zhdanov AV, Nolan YM et al (2013) Imaging of neurosphere oxygenation with phosphorescent probes. Biomaterials 34:9307–9317
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255:1707–1710
Monni E, Congiu T, Massa D et al (2011) Human neurospheres: from stained sections to three-dimensional assembly. Translational Neurosci 2:43–48
Pampaloni F, Reynaud EG, Stelzer EH (2007) The third dimension bridges the gap between cell culture and live tissue. Nat Rev Mol Cell Biol 8:839–845
Gil‐Perotín S, Duran‐Moreno M, Cebrián‐Silla A et al (2013) Adult neural stem cells from the subventricular zone: a review of the neurosphere assay. Anat Rec 296:1435–1452
Gähwiler BH, Capogna M, Debanne D et al (1997) Organotypic slice cultures: a technique has come of age. Trends Neurosci 20:471–477
Ohnishi T, Matsumura H, Izumoto S et al (1998) A novel model of glioma cell invasion using organotypic brain slice culture. Cancer Res 58:2935–2940
Zimmer J, Kristensen BW, Jakobsen B et al (2000) Excitatory amino acid neurotoxicity and modulation of glutamate receptor expression in organotypic brain slice cultures. Amino Acids 19:7–21
Dionne KR, Smith Leser J, Lorenzen KA et al (2011) A brain slice culture model of viral encephalitis reveals an innate CNS cytokine response profile and the therapeutic potential of caspase inhibition. Exp Neurol 228:222–231
Su T, Paradiso B, Long Y-S et al (2011) Evaluation of cell damage in organotypic hippocampal slice culture from adult mouse: a potential model system to study neuroprotection. Brain Res 1385:68–76
McCord AM, Jamal M, Shankavarum UT et al (2009) Physiologic oxygen concentration enhances the stem-like properties of CD133+ human glioblastoma cells in vitro. Mol Cancer Res 7:489–497
Lancaster MA, Renner M, Martin C-A et al (2013) Cerebral organoids model human brain development and microcephaly. Nature 501:373–379
Finikova OS, Lebedev AY, Aprelev A et al (2008) Oxygen microscopy by two‐photon‐excited phosphorescence. Chem Phys Chem 9:1673–1679
Kazmi SMS, Salvaggio AJ, Estrada AD et al (2013) Three-dimensional mapping of oxygen tension in cortical arterioles before and after occlusion. Biomed Opt Express 4:1061–1073
Becker W (2012) Fluorescence lifetime imaging – techniques and applications. J Microsc 247:119–136
Bershteyn M, Kriegstein AR (2013) Cerebral organoids in a dish: progress and prospects. Cell 155:19–20
Stoppini L, Buchs P-A, Muller D (1991) A simple method for organotypic cultures of nervous tissue. J Neurosci Meth 37:173–182
Barry DS, Pakan JMP, O’Keeffe GW et al (2013) The spatial and temporal arrangement of the radial glial scaffold suggests a role in axon tract formation in the developing spinal cord. J Anat 222:203–213
Pampaloni F, Ansari N, Stelzer EH (2013) High-resolution deep imaging of live cellular spheroids with light-sheet-based fluorescence microscopy. Cell Tiss Res 352:161–167
Wilt BA, Burns LD, Wei Ho ET et al (2009) Advances in light microscopy for neuroscience. Annu Rev Neurosci 32:435–506
Graf BW, Boppart SA (2010) Imaging and analysis of three-dimensional cell culture models. In: Live cell imaging, vol 591, Methods in molecular biology. Springer, New York, pp 211–227
Dmitriev RI, Papkovsky DB (2012) Optical probes and techniques for O2 measurement in live cells and tissue. Cell Mol Life Sci 69:2025–2039
Mehta G, Hsiao AY, Takayama S (2012) Opportunities and challenges for use of tumor spheroids as models to test drug delivery and efficacy. J Control Release 164:192–204
Frigault MM, Lacoste J, Swift JL et al (2009) Live-cell microscopy–tips and tools. J Cell Sci 122:753–767
Kondrashina AV, Dmitriev R, Borisov SM et al (2012) A phosphorescent nanoparticle-based probe for sensing and imaging of (intra)cellular oxygen in multiple detection modalities. Adv Funct Mater 22:4931–4939
Fercher A, Borisov SM, Zhdanov A et al (2011) Intracellular O2 sensing probe based on cell-penetrating phosphorescent nanoparticles. ACS Nano 5:5499–5508
Tsytsarev V, Arakawa H, Borisov SM et al (2013) In vivo imaging of brain metabolism activity using a phosphorescent oxygen-sensitive probe. J Neurosci Meth 216:146–151
Acknowledgments
This work was supported by the Science Foundation Ireland, grants 12/RC/2276 and 12/TIDA/B2413, and the EC FP7 Program, grant NanoBio4Trans (No. 304842-2). We thank Dr. J. M.P. Pakan and Dr. Y.M. Nolan (Department of Anatomy and Neuroscience, University College Cork) for the help in preparation of brain slices and neurosphere cultures.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Dmitriev, R.I., Papkovsky, D.B. (2015). Multi-parametric O2 Imaging in Three-Dimensional Neural Cell Models with the Phosphorescent Probes. In: Lossi, L., Merighi, A. (eds) Neuronal Cell Death. Methods in Molecular Biology, vol 1254. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-2152-2_5
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2152-2_5
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-2151-5
Online ISBN: 978-1-4939-2152-2
eBook Packages: Springer Protocols