Abstract
Vitamin D deficiency is one of the most common medical conditions. This chapter reviews the photobiology, metabolism, biologic actions of vitamin D as well as strategies to treat and prevent vitamin D deficiency. Vitamin D, the sunshine vitamin, is well recognized as being important for the development and maintenance of bone health throughout life. The major source of vitamin D for children and adults is from sun exposure. Solar ultraviolet B radiation converts 7-dehydrocholesterol to previtamin D3 which in turn thermally isomerizes to vitamin D3. Once formed it enters the circulation and along with vitamin D2 and vitamin D3 coming from dietary sources travels to the liver and is converted to the major circulating form 25-hydroxyvitamin D [25(OH)D]. 25(OH)D enters the circulation and travels to the kidneys where it is converted to its active form 1,25-dihydroxyvitamin D [1,25(OH)2D]. 1,25(OH)2D interacts with its vitamin D receptor (VDR) in the intestine resulting in an increase in intestinal calcium absorption. In the skeleton it increases the number of osteoclasts to mobilize calcium from the skeleton when necessary. Vitamin D deficiency, defined as a 25-hydroxyvitamin D <20 ng/mL, is one of the most common medical disorders worldwide. Strategies using sensible sun exposure with the app dminder.info along with vitamin D supplementation are discussed in detail. Vitamin D insufficiency has been defined as a 25(OH)D of 21–29 ng/mL and sufficiency as >30 ng/mL. Vitamin D toxicity is usually not observed until 25(OH)D levels are >200 ng/mL. Essentially every tissue and cell in the body has a VDR and many cells including macrophages have the ability to convert 25(OH)D to 1,25(OH)2D. Epidemiologic and association studies have suggested that vitamin D deficiency increases risk for many acute and chronic illnesses including autoimmune diseases such as multiple sclerosis and type 1 diabetes, cardiovascular disease, several cancers, type 2 diabetes, infectious diseases and neurocognitive dysfunction. Because vitamin D toxicity is an extremely rare occurrence based on the totality of evidence to date about the many health benefits of vitamin D it is reasonable to encourage sensible sun exposure in combination with vitamin D supplementation. The Endocrine Society's practice guidelines recommends children 1 year and older receive 600–1,000 IU daily and adults 1,500–2,000 IU daily with the caveat that obese people require 2–3 times more.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Vitamin D
- Sunlight
- 25-Hydroxyvitamin D
- 1,25-Dihydroxyvitamin D
- Rickets
- Osteoporosis
- Osteomalacia
- Cancer
- Autoimmune diseases
- Infectious diseases
-
Vitamin D, the sunshine vitamin, is well recognized as being important for the development and maintenance of bone health throughout life.
-
The major source of vitamin D for children and adults is from sun exposure. Solar ultraviolet B radiation converts 7-dehydrocholesterol to previtamin D3 which in turn thermally isomerizes to vitamin D3.
-
Vitamin D3 from the skin enters the circulation and along with vitamin D2 and vitamin D3 coming from dietary sources travels to the liver and is converted to the major circulating form 25-hydroxyvitamin D [25(OH)D].
-
25(OH)D enters the circulation and travels to the kidneys where it is converted to its active form 1,25-dihydroxyvitamin D [1,25(OH)2D]. 1,25(OH)2D interacts with its vitamin D receptor (VDR) in the intestine resulting in an increase in intestinal calcium absorption. In the skeleton it increases the number of osteoclasts to mobilize calcium from the skeleton when necessary.
-
Vitamin D deficiency, defined as a 25-hydroxyvitamin D <20 ng/mL and vitamin D insufficiency has been defined as a 25(OH)D of 21–29 ng/mL and sufficiency as >30 ng/mL.
-
Vitamin D toxicity is usually not observed until 25(OH)D levels are >200 ng/mL.
-
Essentially every tissue and cell in the body has a VDR and many cells including macrophages have the ability to convert 25(OH)D to 1,25(OH)2D.
-
Epidemiologic and association studies have suggested that vitamin D deficiency increases risk for many acute and chronic illnesses including autoimmune diseases such as multiple sclerosis and type 1 diabetes, cardiovascular disease, several cancers, type 2 diabetes, infectious diseases and neurocognitive dysfunction.
-
The Endocrine Society's practice guidelines recommends children 1 year and older receive 600–1,000 IU daily and adults 1,500–2,000 IU daily with the caveat that obese people require 2–3 times more.
1 Introduction
Vitamin D, the sunshine vitamin, is well recognized as being important for the development and maintenance of bone health throughout life. The major source of vitamin D for children and adults is from sun exposure. Solar ultraviolet B radiation converts 7-dehydrocholesterol to previtamin D3 which in turn thermally isomerizes to vitamin D3. Once formed it enters the circulation and along with vitamin D2 and vitamin D3 coming from dietary sources travels to the liver and is converted to the major circulating form 25-hydroxyvitamin D [25(OH)D]. 25(OH)D enters the circulation and travels to the kidneys where it is converted to its active form 1,25-dihydroxyvitamin D [1,25(OH)2D]. 1,25(OH)2D interacts with its vitamin D receptor (VDR) in the intestine resulting in an increase in intestinal calcium absorption. In the skeleton it increases the number of osteoclasts to mobilize calcium from the skeleton when necessary. Vitamin D deficiency, defined as a 25-hydroxyvitamin D <20 ng/mL, is one of the most common medical disorders worldwide. Strategies using sensible sun exposure with the app dminder.info along with vitamin D supplementation are discussed in detail. Vitamin D insufficiency has been defined as a 25(OH)D of 21–29 ng/mL and sufficiency as >30 ng/mL. Vitamin D toxicity is usually not observed until 25(OH)D levels are >200 ng/mL. Essentially every tissue and cell in the body has a VDR and many cells including macrophages have the ability to convert 25(OH)D to 1,25(OH)2D. Epidemiologic and association studies have suggested that vitamin D deficiency increases risk for many acute and chronic illnesses including autoimmune diseases such as multiple sclerosis and type 1 diabetes, cardiovascular disease, several cancers, type 2 diabetes, infectious diseases and neurocognitive dysfunction. Because vitamin D toxicity is an extremely rare occurrence based on the totality of evidence to date about the many health benefits of vitamin D it is reasonable to encourage sensible sun exposure in combination with vitamin D supplementation. The Endocrine Society’s practice guidelines recommends children 1 year and older receive 600–1,000 IU daily and adults 1,500–2,000 IU daily with the caveat that obese people require 2–3 times more.
2 Evolution of Vitamin D
Although it is not certain when vitamin D became critically important for calcium metabolism and bone health for our early ancestors, there is evidence that some of the earliest phytoplankton life forms were photosynthesizing vitamin D more than 750 million years ago [1–3]. Life evolved in a fertile soup that contained all of the organic and inorganic compounds necessary for life to evolve. One of the key elements that early life forms used was calcium for regulation of many metabolic processes. As invertebrates and vertebrates evolved, they took advantage of the high calcium content of their ocean environment (approx. 400 mmol) and used it as a major component for their exo- and endoskeletons, respectively. When vertebrate life forms ventured onto land, the calcium on which they became dependent was plentiful in the soils, but they had no mechanism to extract it. Plants, however, extracted the precious calcium out of the soils and distributed it throughout their structures. Thus, calcium was harvested by vertebrates from the soil indirectly by the ingestion of these plants. To utilize the dietary calcium there was a need for a mechanism to recognize the calcium status of the organism and to regulate the efficiency of intestinal calcium absorption depending on the organism’s calcium needs. It is likely that vitamin D played a crucial role in early vertebrate development by regulating intestinal calcium absorption and calcium metabolism [1–3].
3 Vitamin D Metabolism and Action on the Intestine
Once vitamin D is made in the skin, it enters the circulation. Vitamin D (vitamin D represents either vitamin D2 or vitamin D3) from the diet is incorporated in chylomicrons and absorbed into the lymphatic system, where it eventually is deposited into the venous circulation. Both dietary and skin sources of vitamin D are bound in the circulation to a vitamin D-binding protein (DBP) [4]. Some of the lipophylic vitamin D in the circulation is deposited in the body fat, while most of it is directed to the liver [5–7]. Once it enters hepatocytes, it is metabolized by the vitamin D-25-hydroxylase (CYP27A) and transformed to 25-hydroxyvitamin D [25(OH)D] [6, 7]. 25(OH)D leaves the hepatocyte and enters the circulation and is once again bound to the DBP. 25(OH)D is the major circulating form of vitamin D and, as a result, is used to determine the vitamin D status of both children and adults. The 25(OH)D–DBP complex is recognized by megalin that is located in the plasma membrane of the renal tubular cells. Megalin facilitates the endocytic transport of the 25(OH)D–DBP complex into the renal cell [8]. 25(OH)D is then released and enters the mitochondria, where the cytochrome P450-25-hydroxyvitamin D-1-hydroxylase (CYP27B1; 1-OHase) converts it to 1,25-dihydroxyvitamin D [1,25(OH)2D] [3, 6, 7] (Fig. 27.1). The renal 1-OHase is upregulated by hypocalcemia and hypophosphatemia. Parathyroid hormone (PTH) is a potent stimulator of the renal 1-OHase whereas fibroblast growth factor 23 (FGF23) produced by osteocytes and osteoblasts inhibits its activity (Fig. 27.1). During pregnancy and lactation, estrogen and prolactin are also thought to play a role in upregulating the 1-OHase [6, 7].
Schematic representation of the synthesis and metabolism of vitamin D for regulating calcium, phosphorus, and bone metabolism. During exposure to sunlight, 7-dehydrocholesterol in the skin is converted to previtamin D3. Previtamin D3 immediately converts by a heat-dependent process to vitamin D3. Excessive exposure to sunlight degrades previtamin D3 and vitamin D3 into inactive photoproducts. Vitamin D2 and vitamin D3 from dietary sources are incorporated into chylomicrons, transported by the lymphatic system into the venous circulation. Vitamin D (D represents D2 or D3) made in the skin or ingested in the diet can be stored in and then released from fat cells. Vitamin D in the circulation is bound to the vitamin D binding protein, which transports it to the liver, where vitamin D is converted by the vitamin D-25-hydroxylase to 25-hydroxyvitamin D (25(OH)D). This is the major circulating form of vitamin D that is used by clinicians to measure vitamin D status (although most reference laboratories report the normal range to be 20–100 ng/mL, the preferred healthful range is 30–60 ng/mL). It is biologically inactive and must be converted in the kidneys by the 25-hydroxyvitamin D-1α-hydroxylase (1-OHase) to its biologically active form 1,25-dihydroxyvitamin D (1,25(OH)2D). Serum phosphorus, calcium fibroblast growth factors (FGF-23), and other factors can either increase or decrease the renal production of 1,25(OH)2D. 1,25(OH)2D feedback regulates its own synthesis and decreases the synthesis and secretion of parathyroid hormone (PTH) in the parathyroid glands. 1,25(OH)2D increases the expression of the 25-hydroxyvitamin D-24-hydroxylase (24-OHase) to catabolize 1,25(OH)2D to the water-soluble, biologically inactive calcitroic acid, which is excreted in the bile. 1,25(OH)2D enhances intestinal calcium absorption in the smallFig. 27.1 (continued) intestine by stimulating the expression of the epithelial calcium channel (ECaC) and the calbindin 9K (calcium binding protein; CaBP). 1,25(OH)2D is recognized by its receptor in osteoblasts, causing an increase in the expression of the receptor activator of the NFκB ligand (RANKL). Its receptor RANK on the preosteoclast binds RANKL, which induces the preosteoclast to become a mature osteoclast. The mature osteoclast removes calcium and phosphorus from the bone to maintain blood calcium and phosphorus levels. Adequate calcium and phosphorus levels promote the mineralization of the skeleton. UVB, ultraviolet B. Reproduced with permission from Holick, copyright 2012
1,25(OH)2D is considered to be the biologically active form of vitamin D. It binds to its specific nuclear vitamin D receptor (VDR), which in turn binds with the retinoic acid X receptor (RXR) to form a herterodimeric complex. This complex interacts with specific sequences in the promoter region of vitamin D-responsive genes, known as vitamin D-responsive element (VDRE) [3, 7, 9, 10]. The binding of the VDR-1,25(OH)2D-RXR complex to the VDRE initiates the binding of several transcriptional factors that ultimately results in either an increased or decreased expression of vitamin D-responsive genes [9–11].
1,25(OH)2D is recognized by the VDR in the small intestine, resulting in an increase in the expression of the epithelial calcium channel on the mucosal surface of the intestinal absorptive cell [2, 3, 7]. In addition, there is an increase in the expression of the calcium-binding protein9K (calbindin), calcium-dependent ATPase, and several other brush border proteins [2, 3, 7, 10, 12]. The ultimate result is that 1,25(OH)2D enhances the efficiency of intestinal calcium absorption from a baseline of approx. 10–15 to 30–40 %. Most of the dietary calcium is absorbed in the duodenum and to a lesser extent in the jejunum and ileum.
Once 1,25(OH)2D carries out its function in the small intestine, it then induces the expression of the 25-hydroxyvitamin D-24-hydroxylase (CYP-24). This results in the initiation of a cascade of metabolic steps that culminates in the cleavage of the side chain between carbons 23 and 24 to yield the water-soluble, biologically inactive excretory product, calcitroic acid [3, 7, 10].
4 Vitamin D Action on Bone Calcium Mobilization
Although vitamin D is associated with bone health, the principal physiological function of vitamin D is to support the serum calcium within a physiologically acceptable range in order to maintain neuromuscular and cardiac function and a multitude of other metabolic activities [2]. Thus, when dietary calcium is inadequate to satisfy the body’s requirement for calcium, this results in vitamin D becoming a catabolic hormone that mobilizes calcium stores from the skeleton.
1,25(OH)2D increases the removal of calcium from the skeleton by increasing osteoclastic activity. It was originally believed that 1,25(OH)2D interacted with specific nuclear receptors in preosteoclasts to initiate the formation of mature osteoclasts. We now recognize that 1,25(OH)2D initiates the mobilization of preosteoclasts through its interaction with its VDR in osteoblasts. The osteoblast serves as the master cell for regulating bone metabolism. 1,25(OH)2D interacts with the VDR in mature osteoblasts and induces the expression of RANKL (receptor for RANKL) on its plasma membrane surface [3, 7, 13–15]. The precursor monocytic osteoclasts have a membrane receptor for RANKL, known as RANK (receptor activator NFκB). It is the intimate interaction of the preosteoclast’s RANK with the osteoblast’s RANKL that ultimately signals the preosteoclast to become a mature bone-resorbing multinucleated osteoclast (Fig. 27.1). Thus, in calcium-deficient states 1,25(OH)2D production is enhanced and in turn mobilizes an army of osteoclasts that resorb bone-releasing precious calcium stores into the circulation to maintain ionized calcium levels in the normal range.
5 Vitamin D and Bone Mineralization
1,25(OH)2D interacts with osteoblasts, not only to increase the expression of RANKL, but also to enhance the expression of osteocalcin, alkaline phosphatase, and osteopontin [6, 7, 10, 16, 17]. Despite all of these biological functions in the osteoblast, there is no evidence that 1,25(OH)2D is essential for the ossification process of the collagen matrix [18–20]. This is based on the observation that severely vitamin D-deficient rats that either received a high-calcium and high-phosphorus-with-lactose diet or received calcium intravenously had bones that had no evidence of rickets or other pathology (Fig. 27.2) [19]. This has also been confirmed in rachitic patients with a VDR defect known as 1,25(OH)2D-resistant rickets (vitamin D-dependent rickets type 2) and who received an infusion of calcium, resulting in the healing of their rickets [20].
Epiphyseal plates of tibias from rats that were fed (a) a vitamin D-deficient diet and supplemented with 125 ng (5 IU) of vitamin D3 orally five times a week, (b) a vitamin D-deficient diet containing 3 % calcium and 0.65 % phosphorus, and (c) a vitamin D-deficient diet with 20 % lactose, 4 % calcium, and 1 % phosphorus. Note the wide and disorganized hypertrophic zone in the vitamin D-deficient rat’s tibial epiphyseal (b) fed high calcium and normal phosphorus diet compared with normal tibial epiphyseal plates from the rats that were either vitamin D repleted (a) or maintained on normal serum calcium and phosphorus by being on a high-calcium lactose, high-phosphorus diet (c). (Reproduced with permission from ref. [17]
6 Dietary Sources of Vitamin D
There are very few foods that naturally contain vitamin D. These foods include oily fish including makerel, eel, and salmon, cod liver oil, sun- and UV-exposed mushrooms, and egg yolks (Table 27.1).
Steenbock [21] recognized the importance of promoting antirachitic activity in foods by irradiating them with ultraviolet radiation. He suggested irradiation of milk that was fortified with ergosterol (provitamin D2) as a mechanism to provide children with their vitamin D requirement. This recommendation was embraced by the United States, Canada, and Europe, and this simple food fortification program essentially eradicated rickets by 1940.
In the 1930s, the fortification of milk with vitamin D was a novelty and many companies became interested in fortifying their products with vitamin D. This included, among others, Bond bread, Rickter’s hot dogs, and Twang soda. Schlitz Brewery cleverly marketed their beer as containing the sunshine vitamin D (Fig. 27.3). In Europe, custards, milk, and other foods were fortified with vitamin D [22].
In the late 1930s, the US Food and Drug Administration forbade any nutritional claims for alcoholic beverages, and vitamin D fortification of beer was halted. In Europe in the 1950s there were several outbreaks of vitamin D intoxication, that is, hypercalcemia in children, which caused great alarm [23]. This resulted in most European countries forbidding the fortification of any food product with vitamin D.
Based on reports that these infants who were hypercalcemic also suffered from mental retardation, heart problems and had altered facial features is consistent with these children suffering from the rare genetic disorder Williams syndrome. Children with this genetic disorder have elfin faces, heart problems, mild mental retardation and have a hypersensitivity to vitamin D which can cause hypercalcemia. Recently Finland and Sweden have lifted restrictions on the fortification of milk with vitamin D [7].
In the United States, milk, orange juice, some breads, cereals, and yogurts are fortified with vitamin D. There is 100 IU (2.5 μg) of vitamin D in 8 oz of milk. In most European countries, margarine and some cereals are fortified with vitamin D (Table 27.1).
The reason milk was the vehicle for the vitamin D supplementation program was that children drank milk and they were at risk for developing rickets. However, with the awareness that vitamin D deficiency is an epidemic in both young, middle-aged, and older adults, there is a need for other dietary sources of vitamin D other than milk. Tangpricha et al. [24] observed that the fat content in milk does not influence vitamin D bioavailability. They also demonstrated that vitamin D added to orange juice was bioavailable for young and middle-aged adults. Thus, the recent introduction of vitamin D-fortified orange juice and other juice products heralded a new era in the vitamin D fortification process and should have a significant impact on vitamin D status of children and adults who consume these products.
7 Vitamin D from Sunlight Exposure
Because very few foods contain vitamin D, most children and adults receive their vitamin D requirement from exposure to sunlight. During sunlight exposure, the solar ultraviolet B photons (UVB; with energies 290–315 nm) penetrate into the epidermis and are absorbed by 7-dehydrocholesterol (provitamin D3) that resides in the plasma membrane of the epidermal cells [3, 15, 25]. This absorption results in a rearrangement of the double bonds that causes the B ring to open to form previtamin D3 (Fig. 27.1). Previtamin D3 exists in two conformeric forms, the s-cis, s-cis (czc) and its more thermodynamically stable counterpart the s-trans, s-cis (tzc) conformer (Fig. 27.4). It is only the czc conformer that can undergo rearrangement of its double bonds to form vitamin D3. In order for the skin to efficiently convert previtamin D3 to vitamin D3, the previtamin D3 is made in the plasma membrane and is locked into the czc conformation, which then can rapidly isomerize to vitamin D3 [26, 27]. Once formed, this molecule no longer is sterically compatible to reside in the cell’s plasma membrane and is released into the extracellular space, where it is picked up in the dermal capillary bed and bound to the DBP (Fig. 27.1).
Photolysis of provitamin D3 (pro-D3) into previtamin D3 (pre-D3) and its thermal isomerization of vitamin D3 in hexane and in lizard skin. In hexane pro-D3 is photolyzed to s-cis,s-cis-pre-D3. Once formed, this energetically unstable conformation undergoes a conformational change to the s-trans,s-cis-pre-D3. Only the s-cis,s-cis-pre-D3 can undergo thermal isomerization to vitamin D3. The s-cis,s-cis conformer of pre-D3 is stabilized in the phospholipid bilayer by hydrophilic interactions between the 3β-hydroxyl group and the polar head of the lipids, as well as by the van der Waals interactions between the steroid ring and side-chain structure and the hydrophobic tail of the lipids. These interactions significantly decrease the conversion of the s-cis,s-cis conformer to the s-trans,s-cis conformer, thereby facilitating the thermal isomerization of s-cis,s-cis-pre-D3 to vitamin D3. (Reproduced with permission from ref. [26])
Unlike vitamin D that is absorbed in the small intestine into the chylomicron fraction, where no more than two-thirds of it is bound to DBP, essentially 100 % of the vitamin D3 that comes from the skin and enters into the venous circulation is bound to the DBP [28]. This gives the cutaneous vitamin D3 a more prolonged half-life in the circulation and thus provides an advantage for obtaining vitamin D from exposure of the skin to the sun.
8 Factors That Influence the Cutaneous Production of Vitamin D3
Since the vitamin D3 synthetic process is dependent on the number of UVB photons that enters into the epidermis, anything that interferes with the number of photons reaching the Earth’s surface and ultimately penetrating into the viable epidermis results in an alteration in the production of vitamin D3 in the skin.
During exposure to sunlight, the UVB photons enter into the skin and initiate the photochemistry necessary for producing previtamin D3. The UVB photons also signal melanocytes to increase the production of melanin. Melanin acts as a natural sunscreen and is efficiently packaged into melanosomes that migrate upward to the upper layers of the epidermis, where they efficiently absorb UVB and ultraviolet A (321–400 nm) radiation. An increase in skin pigmentation is inversely related to the number of UVB photons that can penetrate into the epidermis and dermis. Thus, the efficiency in utilizing UVB photons to produce vitamin D3 in the skin is inversely related to the amount of skin pigmentation. This effect can be quite dramatic. A person with deep skin pigmentation of African origin (skin type 5), who is exposed to the same amount of sunlight as a person with minimum skin pigmentation of Celtic or Scandinavian origin (skin type 2), will produce no more than 5–10 % of that produced in the lighter-skinned individual [3, 15, 29] (Fig. 27.5).
Change in serum concentrations of vitamin D in two lightly pigmented white (skin type 2) (a) and three heavily pigmented black subjects (skin type 5) (b) after total-body exposure to 54 mJ/cm2 of UVB radiation. (c) Serial change in circulation vitamin D after reexposure of one black subject in b to a 320-mJ/cm2 dose of UVB radiation. (Reproduced with permission from ref. [29]
Sunscreens are heavily promoted for the prevention of skin cancer and wrinkles. Sunscreens, like melanin, efficiently absorb UVB radiation when applied topically to the skin. As a result, there is a marked diminishment in the penetration of UVB photons into the epidermis. The proper use of a sunscreen (2 mg sunscreen/cm2 skin surface, i.e., about 1 oz or 25 % of a 4-oz bottle applied to all sun exposed skin of a person wearing a bathing suit) with an SPF of 8 reduces the production of previtamin D3 by more than 95 % [30] (Fig. 27.6a). Clothing absorbs 100 % of the incident UVB radiation, and thus no vitamin D3 is made in the skin covered by clothing [31]. This is the reason why women who wear veils and cover all sun-exposed skin with clothing when outside are often vitamin D deficient [32, 33]. Glass also absorbs all UVB photons. Therefore, exposure of the skin from sunlight that has passed through glass will not promote vitamin D3 synthesis in the skin [34].
(a) Circulating concentrations of vitamin D after a single exposure to 1 minimal erythemal dose of simulated sunlight with either a sunscreen, with a sun protection factor of (SPF-8) 8, or a topical placebo cream. (b) Circulating concentrations of vitamin D in response to a wholebody exposure to 1 minimal erythemal dose in healthy young and elderly subjects. (Reproduced with permission from ref. [34]
Aging causes a decrease in the amount of 7-dehydrocholesterol in the epidermis [7, 34]. Elders exposed to the same amount of sunlight as a young adult will produce approx. 25 % of the amount of previtamin D3, compared to a young adult [34] (Fig. 27.6b).
The angle by which sunlight penetrates the Earth’s atmosphere also dramatically influences the production of previtamin D3 in the skin. This angle, known as the zenith angle, is related to season, time of day, and latitude. There is a direct relationship with increase in latitude and in the zenith angle of the sun. The higher the zenith angle, the longer is the path length that solar UVB photons have to travel through the ozone layer, which efficiently absorbs most of these vitamin D3-producing photons. Typically, in the summer no more than about 0.1 % of the solar UVB photons that hit the outer stratosphere reach the Earth’s surface. The lowest zenith angle, which permits more UVB photons to penetrate to the Earth’s surface, occurs at around noontime and in the middle of the summer at the Equator.
During the winter (i.e., November–February) above and below 35° latitude, the zenith angle is so oblique that essentially all of the UVB photons are absorbed by the stratospheric ozone layer. As a result, very little, if any, previtamin D3 can be produced in human skin. At very high latitudes, such as Bergen, Norway, and Edmonton, Canada, little, if any, previtamin D3 is produced between the months of October and March. Figure 27.7 shows how latitude, season, and time of day dramatically influence the production of previtamin D3 in the skin [35].
Influence of season, time of day, and latitude on the synthesis of previtamin D3 in Northern (a and c) and Southern Hemispheres (b and d). The hour indicated in c and d is the end of the 1-h exposure time. (Reproduced with permission from ref. [35]
There has been a lot of confusion about the mixed message of avoiding all direct sunlight because of skin cancer risk and the need for some sensible sun exposure to provide children and adults with their vitamin D requirement. Sensible sun exposure which means to never be exposed to an amount of sunlight that would cause a sunburn, which is the major cause for both skin cancer and skin damage, can provide children and adults with their vitamin D requirement. The recommendation is to always protect the face which is most sun damaged from direct sun exposure. However exposure of other body parts such as arms legs abdomen and back to about 50 % of the time that it would take to get a light pinkness 24 h later (known as one Minimal Erythemal Dose) can be a good source for vitamin D production. To overcome the vagaries of the variables associated with sun-induced vitamin D synthesis including time a day, cloud cover, season, latitude and skin type a free app has been developed that provides the user not only with useful information for how much vitamin D is being produced but also alerts the user when they are at risk for over exposure to sunlight. It is available at dminder.info.
9 Consequences of Vitamin D Deficiency on Musculoskeletal Health
Chronic vitamin deficiency in infants and young children causes the bonedeforming disease commonly known as rickets. Vitamin D deficiency disrupts chondrocyte maturation and inhibits the normal mineralization of the growth plates. This causes a widening of the epiphyseal plates that is commonly seen at the ends of the long bones in rachitic children, as well as bulging of the costo-chondral junctions that results in what is known as the rachitic rosary. The skeleton is also poorly mineralized, due to the low calcium × phosphate product. This poor mineralization makes the skeleton less rigid, and when the rachitic child begins to stand, gravity causes either inward or outward bowing of the long bones in the lower extremities, resulting in bowed legs or knocked knees, respectively (Fig. 27.8).
In adults after the epiphyseal plates have been fused, the skeletal abnormalities resulting from vitamin D deficiency are more subtle. Vitamin D deficiency results in a decrease in efficiency of intestinal calcium absorption. This causes a decrease in the serum ionized calcium, which is immediately recognized by the calcium sensor in the parathyroid glands [36]. This results in an increase in the expression and production of PTH. PTH, in turn, has three options to maintain serum calcium levels within a physiologically acceptable range. It can increase the efficiency of the renal tubules, especially the distal convoluted tubules, to increase the reabsorption of calcium from the ultrafiltrate. It also stimulates the kidney to produce more 1,25(OH)2D, which in turn increases intestinal calcium absorption (Fig. 27.1). If these actions are not adequate to maintain the serum calcium levels, then PTH will stimulate the expression of RANKL in osteoblasts to mobilize preosteoclasts to become mature bone-resorbing osteoclasts by a mechanism similar to 1,25(OH)2D [13, 37] (Fig. 27.1). Thus, an increase in osteoclastic activity results in the destruction of the matrix and release of calcium into the extracellular space. The net effect is to increase the porosity of the skeleton, thereby causing a decrease in bone mineral density and precipitating or exacerbating osteoporosis.
A more subtle, but important, effect of PTH on skeletal health is its effect on phosphorus metabolism in the kidney. PTH causes an increase in the urinary excretion of phosphorus. Although subtle in nature, the low-normal or low serum phosphorus is inadequate to maintain a supersaturated level of calcium × phosphorus product, resulting in a mineralization defect of the newly laid-down osteoid by osteoblasts. Histologically this appears as widened osteoid seams (Fig. 27.9) and is known as osteomalacia. Because osteoid has no mineral component, it provides little, if any, structural support to the skeleton and increases risk of fracture [38–41]. In addition, the lack of calcium hydroxyapatite deposition in newly laiddown osteoid results in no increase in bone mineral density. It is not possible to detect either by standard X-rays or bone densitometry the difference between osteoporosis, that is, holes in the skeleton, vs. osteomalacia, which is simply a collagen matrix without mineral [42, 43].
Bone histology demonstrating (a) normal mineralized trabecular bone, (b) increased osteoclastic bone resorption due to secondary hyperparathyroidism, and (c) osteomalacia with widened unmineralized osteoid light gray areas. (Reproduced with permission from ref. [15]
Unlike osteoporosis, which is a silent disease until a fracture occurs, osteomalacia is often associated with bone discomfort. Patients often complain of an aching in their skeleton that is unexplained. This can be detected on physical exam by palpating the sternum with minimum pressure of the thumb or forefinger on the sternum or on the anterior tibia. The patient often complains of discomfort with minimum to moderate applied pressure. Although the exact cause for this pain is not known, its possible that the collagen-rich osteoid that is laid down on the periosteal surface of the skeleton becomes hydrated similar to gelatin in Jell-O and causes an outward pressure on the periostial covering that is innervated with sensory pain receptors [44].
Patients with osteomalacia often complain of muscle aches and muscle weakness. There is mounting evidence that vitamin D deficiency results in muscle weakness and increases sway, which can result in increase in falling, thereby increasing risk of skeletal fractures [33, 45, 46].
Patients often complain to their physicians about nonspecific bone aches, muscle aches, and discomfort. Often after a thorough workup, including a sedimentation rate, rheumatoid factor, and even a bone scan, the physician will inform the patient that no specific cause has been found and often these patients are given the diagnosis of fibromyalgia. It has been estimated that upwards of 40–80 % of patients complaining of nonspecific bone pain and muscle aches and weakness are suffering not from fibromyalgia, but from chronic vitamin D deficiency [33, 44].
10 Prevalence of Vitamin D Deficiency in Children and Adults
It is both surprising and alarming that vitamin D deficiency continues to plague both children and adults [7, 38–55] (Fig. 27.10).
Infants who receive their total nutrition from breast feeding are at high risk of vitamin D deficiency because human milk contains very little, if any, vitamin D to satisfy their requirement [53]. This is especially true for infants of color, because their mothers are often vitamin D deficient as well and provide no vitamin D nutrition in breast milk [52, 54]. Even in Caucasian and African American women who had a mean intake of 457 IU/day, the concentrations of vitamin D and 25(OH)D in their milk was 12.6 IU/L and 37.6 IU/L, respectively [7, 53, 54]. It has been estimated that human milk contains no more than about 15 IU of vitamin D in 8 oz.
Children who are active and outdoors are at little risk of vitamin D deficiency as long as there is a short period of time when they wear no sun protection, such as clothing or sunscreen, on face, arms, and legs.
It has been recognized for more than three decades that globally children and adults are at high risk of developing vitamin D deficiency [7] (Fig. 27.11). Vitamin D deficiency is extremely common in older adults in Europe because essentially no foods are fortified with vitamin D. In the United States and Canada, vitamin D deficiency is also more common than expected [49, 50]. Gloth et al. [47] reported 54 % of community dwellers and 38 % of nursing home residents in the Baltimore area were severely vitamin D deficient [25(OH)D < 10 ng/mL]. Numerous studies have reported that between 25 % and more than 60 % of adults aged 50+ years were vitamin D deficient. In Boston, we observed in independently living elders (83 ± 8 years; 50 white, 14 Hispanic, and 5 African American subjects) in August of 1997 30, 43, and 84 % of white, Hispanic, and black elders were vitamin D deficient [15]. Inpatients are especially at high risk of vitamin D deficiency [55]. It was reported that 57 % of middle-aged and older adults were vitamin D deficient. Sixty percent of the patients consumed less than the recommended adequate intake of vitamin D, and 37 % who had intakes above the recommended daily allowance were found to be vitamin D deficient [7].
Reported incidence of vitamin D deficiency defined as a 25-hydroxyvitamin D <20 ng/mL around the globe including Australia (AU), Canada (CA), China (CH), India (IN), Korea (KR), Malaysia (MA), Middle East (ME), Mongolia (MO), New Zealand (NZ), North Africa (NA), Northern Europe (NE), United States (USA). Holick copyright 2012; reproduced with permission
It would be expected that young and middle-aged active adults would not be at risk of vitamin D deficiency. However, they have several risk factors for vitamin D deficiency, including long hours of work indoors with little exposure to sunlight, and they are also more likely to wear sun protection on all sun-exposed areas because of their worry about increased risk of skin cancer and wrinkles. As a result, when exposed to sunlight they make little vitamin D3 in their skin. In Boston, we observed 32 % of medical students and young doctors, aged 18–29 years, were vitamin D deficient [51]. Fifteen percent had secondary hyperparathyroidism, and 4 % of the students and residents remained vitamin D deficient at the end of the summer.
11 Causes of Vitamin D Deficiency
The major cause of vitamin D deficiency is that it is not appreciated that very few foods naturally contain vitamin D and that most (80–100 %) of our vitamin D requirement comes from casual exposure to sunlight [7, 15, 56] (Fig. 27.12). Even though oily fish contain vitamin D, it is highly variable depending on what season they were caught and whether they were farm raised and what their vitamin D intake was from their diet. Furthermore, it would require that a person eat oily fish at least 2–3 times a week. To satisfy the vitamin D requirement by drinking milk, would require ingesting six and eight glasses a day for children and adults up to the age of 70, and adults aged 70+ years, respectively [53].
Intestinal malabsorption syndromes, especially of the small intestine where vitamin D is absorbed, can lead to severe vitamin D deficiency [15, 57, 58] (Fig. 27.13). Patients with end-stage hepatic failure not only are unable to produce an adequate amount of 25(OH)D, but often suffer from fat malabsorption and are unable to absorb dietary vitamin D. Patients who are on total parenteral nutrition often suffer from a severe metabolic bone disease that is characteristic of vitamin D deficiency osteomalacia. However, the inclusion of 400 IU of vitamin D in the total parenteral nutrition solution does not protect the patient from vitamin D deficiency bone disease [59, 60].
(a) Serum vitamin D concentrations in seven patients with intestinal fat malabsorption syndromes after a single oral dose of 50,000 IU (1.25 mg) of vitamin D2. For comparison, the means and standard errors of vitamin D concentrations measured in seven normal control subjects after a similar dose are indicated by the filled circles and dotted lines. Note that two patients, one with Crohn’s ileocolitis (patient F) and one with ulcerative colitis (patient G), had essentially normal absorption curves. Five patients, however, absorbed very little, if any, vitamin D2. (b) Vitamin D absorption in young (filled circles) and elderly (open circles) adults. Each subject received an oral dose of 50,000 IU of vitamin D2 and at various times blood determinations were made for circulating concentrations of vitamin D. (Reproduced with permission from ref. [57])
The principal cause of vitamin D deficiency is lack of adequate exposure to sunlight. The skin has a large capacity to produce vitamin D3. Exposure of an adult in a bathing suit to simulated sunlight that mimicked the amount of time that would be 1 minimal erythemal dose (1 MED), that is, cause a minimum pinkness to the skin, resulted in an increase in blood levels of vitamin D3 comparable to ingesting between 10,000 and 25,000 IU of vitamin D [15] (Fig. 27.14). Although aging substantially reduces the amount of 7-dehydrocholesterol in the skin, it still has an adequate capacity to make vitamin D [15, 61–63] (Fig. 27.15).
(a) Ampoules containing 7-dehydrocholesterol were placed in a tanning bed at various times and conversion of 7-dehydrocholesterol to previtamin D3 was measured by high performance liquid chromatography. (b) Healthy adults were exposed to 0.75 MED in a tanning bed three times a week for 7 weeks. Circulating concentrations of 25(OH)D were determined at baseline and once a week thereafter. (c) A 76-year-old healthy male was exposed to tanning bed radiation equivalent to 0.75 MED three times a week for 7 weeks. His circulating concentrations of 25(OH)D were obtained at weekly intervals. Reproduced with permission Holick, M.F., Chen, T.C., Sauter, E.R. Vitamin D and Skin Physiology: A D-Lightful Story. J Bone Miner Res. 2007. 22(S2):V28-V33
Patients with obesity often complain of bone aches, muscle aches, and weakness, which exacerbates their inability to be active and their obesity. It is recognized that obesity is associated with vitamin D deficiency [5, 64]. This is due to the fact that body fat acts as a sink for vitamin D. Thus, whether vitamin D is produced in the skin or ingested in the diet, a majority of it is deposited in an almost irreversible manner into the body fat and is not bioavailable to the body (Fig. 27.16).
(a) Mean (±SEM) serum vitamin D3, concentrations before (filled square) and 24 h after (open square) whole-body irradiation (27 mJ/cm2) with UVB radiation. The response of the obese subjects was attenuated when compared with that of the control group. There was a significant time-by-group interaction, p = 0.003. *Significantly different from before values (p < 0.05). (b) Mean (±SEM) serum vitamin D2 concentrations in the control (filled circle) and obese (open circle) groups before and after 25 h after oral intake of vitamin D2 (50,000 IU, 1.25 mg). Vitamin D2 rose rapidly until ~10 h after intake and then declined slightly thereafter. *Significant time and group effects by ANOVA (p < 0.05) but no significant time-by-group interaction. The difference in peak concentrations between the obese and nonobese control subjects was not significant. (Reproduced with permission from ref. [5])
12 Diagnosis of Vitamin D Deficiency
Often physicians assume that the most sensitive indicator to detect vitamin D deficiency is to observe a below-normal serum calcium value. Unfortunately, as explained previously, the body is vigilant to maintain the serum calcium within the normal range in order to maintain most bodily functions. As a result, a person with vitamin D deficiency develops secondary hyperparathyroidism and maintains serum calcium within the normal range until most available calcium is depleted from the skeleton. The secondary hyperparathyroidism results in mild to moderate hypophosphatemia. However, this is also difficult to detect, especially if the patient’s blood is taken in a nonfasting state. Serum phosphorus levels are influenced by dietary phosphorus intake, sugar intake, and by acidosis and alkalosis [6].
With the exception of observing widened epiphyseal plates and Looser’s pseudo-fractures in the long bones, it is not possible to detect vitamin D deficiency by X-rays.
The only method to determine vitamin D deficiency is to measure the blood level of the major circulating form of vitamin D, 25(OH)D. Although 1,25(OH)2D is the biologically active form of vitamin D and would appear to be the ideal marker for vitamin D deficiency, it is not. There are several reasons for this. The circulating concentration of 1,25(OH)2D is 1,000th the concentration of 25(OH)D (pg vs. ng/mL). The half-life for 1,25(OH)2D is only 4–6 h, compared to 2 weeks for 25(OH)D [15]. Finally, as a person becomes vitamin D deficient and develops secondary hyperparathyroidism, the kidney’s 1-OHase produces more 1,25(OH)2D [3, 6, 7, 15]. Thus, when a patient is vitamin D insufficient there is often a normal or even elevated blood level of 1,25(OH)2D [7, 15, 56]. The measurement of 1,25(OH)2D as a gauge of vitamin D status is not only useless, but often misleads physicians into thinking their patient is vitamin D sufficient since the 1,25(OH)2D levels can be normal.
13 Vitamin D Requirement: Adequate Intake vs. Healthy Intake
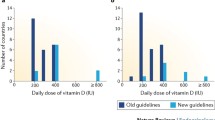
In 2010, the Institute of Medicine announced the new Recommended Daily Allowances (RDA) for vitamin D (1 μg = 40 IU) for children and adults aged 0–1, 1–70, and 71+ years to be 400, 600, and 800 IU/day, respectively (Fig. 27.10) [53]. In 2011 the Endocrine Society issued its guidelines for the treatment and prevention of vitamin D deficiency. They recommended that children in the first year of life should be receiving 400–1,000 IU daily and children 1 year and older should be receiving 600–1,000 IU daily. For all adults it was recommended that they receive 1,500–2,000 IU daily. They also recommended for those who are obese, i.e. BMI >30, require at least 2–3 times more vitamin D to both treat and prevent vitamin D deficiency [65]. Several investigators have reported on the effect of vitamin D intake on circulating concentrations of 25(OH)D. Vieth et al. [66] gave healthy adults (41 ± 9 years) 4,000 IU of vitamin D a day for 2–5 months and did not observe any untoward toxicity. Their 25(OH)D levels during the winter increased from 10.2 ± 4 to 24.1 ± 4 ng/mL. Barger-Lux et al. [67] evaluated a dose response of vitamin D and 25(OH)D intake in healthy males for 4 and 8 weeks, respectively. The groups of adults treated with 1,000, 10,000, or 50,000 IU of vitamin D3/day for 8 weeks demonstrated increases in their serum vitamin D3 levels of 5.0, 52.6, and 300.2 ng/mL, respectively. In the same groups, the 25(OH)D increased by 11.6, 58.4, and 257.2 ng/mL, respectively. Male adults who received 10, 20, or 50 μg of 25(OH)D3/day for 4 weeks demonstrated increases of 25(OH)D by 11.6, 58.4, and 257.2 ng/mL, respectively. None of the men demonstrated any significant change in either their calcium or 1,25(OH)2D levels. In a follow-up study, Heaney et al. [68] gave 67 men who were in general good health either 0, 25, 125, or 250 μg of vitamin D3 for approx. 20 weeks during the winter. They observed serum 25(OH)D levels increased in direct proportion to dose with a slope of approximately 0.28 ng/mL for each additional 1 μg of vitamin D3 ingested. The calculated oral input required to sustain serum 25(OH)D concentrations present in the men during autumn was 12.5 μg/day. The total amount from all sources (supplement, food, tissue stores) needed to sustain the starting 25(OH)D level was estimated at 96 μg (approx. 3,800 IU/day). They concluded that healthy men used between 3,000 and 5,000 IU vitamin D3/day to meet greater than 80 % of their winter vitamin D requirement that was provided by cutaneous production of vitamin D3 during the previous spring, summer, and fall (Fig. 27.17). Tangpricha et al. observed that healthy young and middle-aged female and male adults who ingested 1,000 IU of vitamin D/day for 3 months increased their blood levels of 25(OH)D from 15 ± 3 to 38 ± 8 ng/mL after 2 months. Continued intake of 1,000 IU of vitamin D/day did not increase blood levels of 25(OH)D above 40 ng/mL.
Time course of serum 25-hydroxyvitamin D3 [25(OH)D] concentration for the four dose groups. The points represent the mean values, and error bars are 1 SEM. The curves are fitted to the mean 25(OH)D3 values for each dosage group. The curves, from the lowest upward, are for 0.25, 125, and 250 μg vitamin D3 (labeled dose)/day. The horizontal dashed line reflects zero change from baseline. (Reproduced with permission from ref. [68])
14 Interpreting Serum 25(OH)D Levels
The normal blood level of 25(OH)D varies from different laboratories, but generally is in the range of 20–100 ng/mL [15, 65]. A normal range for an assay is typically obtained by collecting blood from hundreds of healthy volunteers and then determining the blood level of the analyte and using the mean ± 2 SD as the normal range. Because most healthy volunteers are likely to be vitamin D deficient or insufficient a different strategy has been used to define vitamin D deficiency. This became obvious when Malabanan et. al. [48] reported that healthy adults who received 50,000 IU of vitamin D2 once a week for 8 weeks along with calcium supplementation had a more than 30 % decline in their PTH values when there blood levels of 25(OH)D were between 11 and 19 ng/mL. No significant change was observed in adults whose blood levels of 25(OH)D were at least 20 ng/mL. The IOM used this data and other observations and concluded that for maximum bone health the 25(OH)D level should be >20 ng/mL. Thus vitamin D deficiency was defined as a 25(OH)D <20 ng/mL [53]. The Endocrine Society also concluded that vitamin D deficiency should be defined as a 25(OH)D <20 ng/mL. They further recommended that vitamin D insufficiency should be defined as a 25(OH)D of 21–29 ng/mL and that vitamin D sufficiency was defined as a 25(OH)D of 30–100 ng/mL with the preferred range of 40–60 ng/mL [65]. These recommendations were based on observations relating serum 25(OH)D with PTH levels. Many studies reported that PTH levels declined and plateaued when a serum 25(OH)D was between 30–40 ng/mL [69]. A study of 675 otherwise healthy German adults who died in an accident had a blood level of 25(OH)D determined and related to bone biopsy that was evaluated for evidence of unmineralized matrix, i.e. osteomalacia, a hallmark of vitamin D deficiency bone disease. The authors concluded that to guarantee no evidence of vitamin D deficiency bone disease that a blood level of 25(OH)D should be at least 30 ng/mL [70].
The upper range of normal by most assays is 80–100 ng/mL. However, this upper normal range is more an estimate than based on any reports of toxicity. Indeed, lifeguards routinely have blood levels of 25(OH)D of 100 ng/mL with no untoward consequences. Heaney et al. [68] observed blood levels of 25(OH)D3 of 100 ng/mL without any untoward side effects or hypercalcemia. Based on reports of vitamin D intoxication, that is, associated with hypercalcemia, and suppressed PTH levels, 25(OH)D need to be at least 200 ng/mL (Fig. 27.18) [65, 71–74].
Serum calcium level (upper panel) and 25(OH)D level (lower panel) in a patient who had vitamin D intoxication after ingestion of an over-the-counter vitamin D supplement that contained as much as one million units of vitamin D3 in a teaspoon. The patient stopped all vitamin D intake and wore sunscreen before going outside after his hospitalization (month 0). The dotted line (lower panel) represents the upper limit for the 25(OH)D assay that was 46.7 ng/mL. (Reproduced with permission from ref. [71]
Thus, based on the available literature today, it has been suggested that in the absence of any exposure to sunlight, the vitamin D requirement for children and adults is at least 600–1,000 IU and 1,500–2,000 IUof vitamin D/day (Fig. 27.12) [65]. Furthermore, a 25(OH)D level of between 40 and 60 ng/mL should be considered as a healthy range for 25(OH)D [65].
15 Treatment for Vitamin D Deficiency
The best method to treat vitamin D deficiency is to give pharmacological doses of vitamin D. This can be accomplished by giving an oral dose of 50,000 IU of vitamin D once a week for 8 weeks [48]. To prevent recurrence of vitamin D deficiency a maintenance dose of 50,000 IU of vitamin D every 2 weeks which is equivalent to ingesting approximately 3,000 IU of vitamin D daily is effective in normal weight adults in maintaining their blood levels of 25(OH)D in the range of 40–60 ng/mL. This strategy has been effective for at least 6 years without any toxicity (Fig. 27.19) [75, 76]. Alternatively, intramuscular injection of up to 500,000 IU of vitamin D has been demonstrated to prevent vitamin D deficiency in elderly nursing home residents when given twice a year [77]. However, the intramuscular preparation has been ineffective in many patients in raising blood levels of 25(OH)D when given intramuscularly. This may be a bioavailability problem. In addition, a relatively large volume of oil in which the vitamin D is dissolved when given intramuscularly can be quite uncomfortable, which again is a good reason to give a pharmacological doses of vitamin D orally to correct vitamin D deficiency. Aging does not alter vitamin D absorption [15].
(a) Mean serum 25-hydroxyvitamin D [25(OH)D] levels in all patients: Includes patients treated with 50,000 IU vitamin D2 every 2 weeks (maintenance therapy, N = 81), including those patients with vitamin D insufficiency who were initially treated with 8 weeks of 50,000 IU vitamin D2 weekly prior to maintenance therapy (N = 39). Error bars represent standard error of the mean, mean result over 5 years shown. Time 0 is initiation of treatment, results shown as mean values averaged for 6-month intervals. When mean 25(OH)D in each 6-month group was compared to mean initial 25(OH)D, p < 0.001 up until month 43; p < 0.001 when all remaining values after month 43 were compared to mean initial 25(OH)D. (b) Mean serum 25(OH)D levels in patients receiving maintenance therapy only: Levels for 37 patients who were vitamin D insufficient (25[OH]D levels <30 ng/mL) and five patients who were vitamin D sufficient (25[OH]D levels ≥30 ng/ml) who were treated with maintenance therapy of 50,000 IU vitamin D2 every 2 weeks. Error bars represent standard error of the mean, mean result over 5 years shown. Time 0 is initiation of treatment, results shown as mean values averaged for 6-month intervals. When mean 25(OH)D in each 6-month group were compared to mean initial 25(OH)D, p < 0.001 up until month 37; p < 0.001 when all remaining values after month 43 were compared to mean initial 25(OH)D. (c) Serum calcium levels: Results for all 81 patients who were treated with 50,000 IU of vitamin D2. Error bars represent standard error of the mean. Time 0 is initiation of treatment, results shown as mean values averaged for 6-month intervals. Normal serum calcium: 8.5–10.2 mg/dL. (Reproduced with permission from ref. [75])
An alternative and an inexpensive method to treat vitamin D deficiency is to encourage patients to be exposed to some sensible sunlight. The amount depends on the person’s skin sensitivity, time of day, season of the year, and latitude. For example, for an adult in Boston with a skin type 2, who would get a sunburn after being outside for 30 min at noontime in July, the recommendation is exposure to approx. 30–50 % of that time or 9–15 min 2–3 times a week. Always protect the face because of increased risk of wrinkles or skin damage. Exposure of arms, legs, back, abdomen and legs when possible and using the app dminder.info can provide guidance for sensible sun exposure. No sunscreen or sun protection should be used for this brief period of time. However, if the person wishes to stay outdoors for a longer period of time, then use of a sunscreen with an SPF of at least 30 and sun protection with clothing is recommended. For those with marked increased skin pigmentation, the time outside could be as much as 30–60 min, again depending on the person’s skin sensitivity, time of day, season of the year, and latitude [3, 7, 10, 15, 78].
Patients with severe intestinal malabsorption syndrome and who are on total parenteral nutrition can obtain their vitamin D requirement from sun exposure. However, if they cannot go outside or the season will not permit them to make any vitamin D in their skin, then the use of a UVB radiation source, either a home device or a tanning bed at a tanning salon, would be appropriate. In one patient who had only 2 ft of small intestine left, Koutia et al. [79] reported that exposure to 0.75 MED of tanning bed radiation three times a week markedly increased blood levels of 25(OH)D by 700 % and decreased PTH values into the normal range (Fig. 27.20). In addition, the patient, who suffered from severe bone pain and muscle aches and weakness, had complete relief of her symptoms.
Serum 25(OH)D, PTH, and calcium levels in a patient with Crohn’s disease who had whole-body UVB exposure for 10 min, three times in a week for 6 months. (Reproduced with permission from ref. [77])
Chuck et al. [62] also have demonstrated that the use of subliminal UVB lighting in an activity room in a nursing home was the most effective means to sustain 25(OH)D levels within the normal range and was far superior to taking a multivitamin that contained 400 IU of vitamin D a day (Fig. 27.21).
Exposure of nursing home residents to ultraviolet-B lamps that were installed near the ceiling in the day room. This was found to be the most effective method of maintaining serum 25(OH)D levels in these residents. (Reproduced with permission from ref. [62]
16 Nonskeletal Consequences of Vitamin D Deficiency
As early as 1941 it was reported that people who lived in higher latitudes were at higher risk of dying of cancer [80]. A multitude of epidemiological studies have now confirmed this early observation [81–89]. There is firm evidence that people living at higher latitudes are at higher risk of developing and dying of breast, colon, ovarian, and prostate cancers [81–87]. Indeed, mortality rates in both men and women are related to their exposure to sunlight [85] (Fig. 27.22).
(a) Premature mortality due to cancer, white females, vs. total ozone mapping spectrometer (TOMS), July 1992, DNA-weighed UV-B. (b) Premature mortality due to cancer with insufficient UV-B, white males, U.S., 1970–1994, vs. July 1992 DNA-weighted UV-B radiation. (Reproduced with permission from ref. [85])
There is also a latitudinal association with increased risk of developing hypertension and multiple sclerosis [88, 89].
It is now recognized that most tissues and cells possess a VDR. The exact function of 1,25(OH)2D in tissues, such as the brain, breast, prostate, skin, β-islet cells in the pancreas, monocytes, and activated T- and B-lymphocytes, is not fully understood. However, it is known that 1,25(OH)2D is extremely effective in downregulating cellular growth in cells that possess a VDR. Indeed, the potent antiproliferative activity of 1,25(OH)2D has been taken advantage of by the development of activated vitamin D analogs for the treatment of the hyperproliferative disorder psoriasis [90].
It is recognized that the β-islet cells have a VDR and that 1,25(OH)2D modulates insulin production and secretion [3, 7, 10]. 1,25(OH)2D also modulates the immune system by regulating the activity of both activated T- and B-lymphocytes and activated macrophages [7, 10, 15, 91, 92]. This may be the explanation for why Hyponnen et al. [93] observed that children treated with at least 2,000 IU of vitamin D a day reduced their risk of developing type 1 diabetes by 80 %. This was similar to what was observed when NOD mice, which invariably develop type 1 diabetes, received 1,25(OH)2D3 [91, 92]: they showed an 80 % reduction in developing the disease.
The kidney is an endocrine organ for producing 1,25(OH)2D for regulating calcium metabolism. Recently, it was recognized that 1,25(OH)2D also downregulates the production of renin in the kidney [94]. This may be the explanation for why vitamin D deficiency is associated with hypertension and increased risk of coronary artery disease and congestive heart failure [44, 95–99]. Krause et al. [97] reported that exposure of hypertensive adults to a tanning bed that emitted UVB radiation raised the blood levels of 25(OH)D by more than 100 % and controlled their hypertension. A similar group of hypertensive adults exposed to a similar tanning bed for 3 months that emitted UVA but no UVB radiation not only did not increase their blood levels of 25(OH)D, but also had no effect on their hypertension (Fig. 27.23).
Effect of UV-B and UV-A irradiation on ambulatory daytime and night-time blood pressure in hypertensive adults. ns nonsignificant. Thick line = mean. (Reproduced with permission from ref. [97]
17 Vitamin D and the Cancer Connection
Although in the 1990s there were several reports that some of the most common cancers occurred in people living at higher latitudes and that colon cancer and prostate cancer rates were significantly reduced in individuals with higher circulating levels of 25(OH)D, it was difficult to understand how increased exposure to sunlight could impact on decreasing risk of common cancers.
The reason for this is that it was well known that any significant increase in vitamin D intake or exposure to sunlight did not raise blood levels of 1,25(OH)2D. Thus, it was difficult to understand how increasing one’s 25(OH)D levels would be able to regulate cellular growth and prevent some cancers, since circulating levels of 1,25(OH)2D, the antiproliferative hormone, were not increased. The mystery was solved when it was observed that prostate cells and prostate cancer cells expressed a functional 1-OHase similar to what was observed in the skin [3, 15, 99–101]. Since this initial observation, it is now recognized that normal colon tissue and colon cancer, breast and breast cancer cells, as well as variety of other cell types have the enzymatic machinery to convert 25(OH)D directly to 1,25(OH)2D [3, 15, 16, 102–104]. Thus, it appears that when 25(OH)D levels are adequate, probably above 30 ng/mL, it acts a substrate for the extra renal 1-OHase in these tissues. The local production of 1,25(OH)2D may be necessary to maintain and regulate genes responsible for cellular growth and to prevent the cells from becoming autonomous, that is, carcinogenic. It has been suggested that once it carries out its function, it induces the 25-hydroxyvitamin D-24-hydroxylase, which in turn catabolizes 1,25(OH)2D to the inactive water-soluble calcitroic acid (Fig. 27.1).
18 Conclusion
Vitamin D deficiency is extremely common and needs to be recognized. Vitamin D deficiency in children and teenagers can result in poor bone health and the inability to attain the genetically predetermined peak bone mass. In young, middle-aged, and older adults, vitamin D deficiency causes osteomalacia and can precipitate and exacerbate osteoporosis. In addition, many of the symptoms associated with vitamin D deficiency mimic fibromyalgia, and as a result, many patients go undiagnosed.
Vitamin D deficiency, however, may have extremely important health consequences that heretofore have not been fully appreciated. Maintenance of an adequate 25(OH)D level of at least 20 ng/mL and preferably 40–60 ng/mL throughout life may help reduce the risk of developing many chronic diseases, including type 1 diabetes, hypertension, multiple sclerosis, infectious diseases, and cancers of the breast, prostate, colon, and ovary. Vitamin D deficiency has also important health implications for pregnant women and their newborns [7] (Fig. 27.24).
Thus, there needs to be a reawakening about the appreciation of maintaining a healthy vitamin D status throughout life. The best method to determine vitamin D adequacy is to measure 25(OH)D. Similar to evaluating patients for their blood pressure and blood lipid profile on their yearly exam, they should also be evaluated with a 25(OH)D to measure their vitamin D status. This will ensure vitamin D health and mitigate the consequences of vitamin D deficiency.
References
Holick MF. Phylogenetic and evolutionary aspects of vitamin D from phytoplankton to humans. In: Pang PKT, Schreibman MP, editors. Vertebrate endocrinology: fundamentals and biomedical implications, vol. 3. Orlando, FL: Academic; 1989. p. 7–43.
Holick MF. Calcium and vitamin D in human health. Ann Nestle 2002;60(3).
Holick MF. Vitamin D: a millennium perspective. J Cell Biochem. 2003;88:296–307.
Cooke NE, David EV. Serum vitamin D-binding protein is a third member of the albumin and alpha fetoprotein gene family. J Clin Invest. 1985;76:2420–4.
Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF. Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr. 2000;72:690–3.
Holick MF. Evaluation and treatment of disorders in calcium, phosphorus, and magnesium metabolism. In: Noble J, editor. Textbook of primary care medicine. 3rd ed. St. Louis, IL: Mosby; 2001. p. 886–98.
Hossein-nezhad A, Holick MF. Optimize dietary intake of vitamin D: an epigenetic perspective. Curr Opin Clin Nutr Metab Care. 2012;15:567–79.
Nykjaer A, Dragun D, Walther D, et al. An endocytic pathway essential for renal uptake and activation of the steroid 25-(OH) vitamin D3. Cell. 1999;96:507–15.
MacDonald PN. Molecular biology of the vitamin D receptor. In: Holick MF, editor. Vitamin D: physiology. Totowa, NJ: Molecular Biology and Clinical Applications. Humana; 1999. p. 109–28.
Hossein-nezhad A, Spira A, Holick MF. Influence of vitamin D status and vitamin D3 supplementation on genome wide expression of white blood cells: a randomized double-blind clinical trial. PLoS One. 2013;8(3):e58725. doi:10.1371/journal.pone.0058725.
Freedman LP. Multimeric coactivator complexes for steroid/nuclear receptors. Trends Endocrinol Metab. 1999;10:403–7.
Raval-Pandya M, Porta AR, Christakos S. Mechanism of action of 1,25-dihydroxyvitamin D3 on intestinal calcium absorption and renal calcium transport. In: Holick MF, editor. Vitamin D: physiology. Totowa, NJ: Molecular Biology and Clinical Applications. Humana; 1999. p. 163–73.
Khosla S. The OPG/RANKL/RANK system. Endocrinology. 2001;142:5050–5.
Jimi E, Nakamura I, Amano H, et al. Osteoclast function is activated by osteoblastic cells through a mechanism involving cell-to-cell contact. Endocrinology. 1996;137:2187–90.
Holick MF. Vitamin D deficiency. New Engl J Med. 2007;357:266–81.
Zerwekh JE, Sakhaee K, Pak CYC. Short-term 1,25-dihydroxyvitamin D3 administration raises serum osteocalcin in patients with postmenopausal osteoporosis. J Clin Endocrinol Metab. 1985;60:615–7.
Holick MF. Evolution, biologic functions, and recommended dietary allowances for vitamin D. In: Holick MF, editor. Vitamin D: physiology, molecular biology, and clinical applications. Totowa, NJ: Humana; 1999. p. 1–16.
Underwood JL, DeLuca HF. Vitamin D is not directly necessary for bone growth and mineralization. Am J Physiol. 1984;246:E493–8.
Holtrop ME, Cox KA, Carnes DL, Holick MF. Effects of serum calcium and phosphorus on skeletal mineralization in vitamin D-deficient rats. Am J Physiol. 1986;251(2 pt 1):E234–40.
Balsan S, Garabedian M, Larchet M, et al. Long-term nocturnal calcium infusions can cure rickets and promote normal mineralization in hereditary resistance to 1,25-dihydroxyvitamin D. J Clin Invest. 1986;77:1661–7.
Steenbock H. The induction of growth-prompting and calcifying properties in a ration exposed to light. Science. 1924;60:224–5.
Holick MF. Vitamin D: importance for bone health, cellular health and cancer prevention. In: Holick MF, editor. Biologic effects of light. Boston, MA: Kluwer; 2001. p. 155–73.
Oppé TE. Infantile hypercalcemia, nutritional rickets, and infantile survey in Great Britain. Br Med J. 1964;1:1659–61.
Tangpricha V, Koutkia P, Rieke SM, Chen TC, Perez AA, Holick MF. Fortification of orange juice with vitamin D: a novel approach to enhance vitamin D nutritional health. Am J Clin Nutr. 2003;77:1478–83.
MacLaughlin JA, Anderson RR, Holick MF. Spectral character of sunlight modulates the photosynthesis of previtamin D3 and its photo isomers in human skin. Science. 1982;1001–1003.
Holick MF, Tian XQ, Allen M. Evolutionary importance for the membrane enhancement of the production of vitamin D3 in the skin of poikilothermic animals. Proc Natl Acad Sci U S A. 1995;92(8):3124–6.
Tian XQ, Chen TC, Matsuoka LY, Wortsman J, Holick MF. Kinetic and thermodynamic studies of the conversion of previtamin D3 to vitamin D3 in human skin. J Biol Chem. 1993;268(20):14888–92.
Haddad JG, Matsuoka LY, Hollis BW, Hu YZ, Wortsman J. Human plasma transport of vitamin D after its endogenous synthesis. J Clin Invest. 1993;91:2552–5.
Clemens TL, Henderson SL, Adams JS, Holick MF. Increased skin pigment reduces the capacity of skin to synthesize vitamin D3. Lancet. 1982;74–76.
Matsuoka LY, Ide L, Wortsman J, MacLaughlin J, Holick MF. Sunscreens suppress cutaneous vitamin D3 synthesis. J Clin Endocrinol Metab. 1987;64:1165–8.
Matsuoka LY, Wortsman J, Dannenberg MJ, Hollis BW, Lu Z, Holick MF. Clothing prevents ultraviolet-B radiation-dependent photosynthesis of vitamin D3. J Clin Endocrinol Metab. 1992;75(4):1099–103.
Taha SA, Dost SM, Sedrani SH. 25-Hydroxyvitamin D and total calcium: extraordinarily low plasma concentrations in Saudi mothers and their neonates. Pediatr Res. 1984;18:739–41.
Glerup H, Mikkelsen K, Poulsen L, et al. Hypovitaminosis D myopathy without osteomalacic bone involvement. Calcif Tissue Int. 2000;66(6):419–24.
Holick MF. McCollum award lecture, 1994: vitamin D: new horizons for the 21st century. Am J Clin Nutr. 1994;60:619–30.
Chen TC. Photobiology of vitamin D. In: Holick MF, editor. Vitamin D: physiology, molecular biology, and clinical applications. Totowa, NJ: Humana; 1999. p. 17–37.
Brown EM, Pollak M, Seidman CE, et al. Calcium-ion-sensing cell-surface receptors. N Engl J Med. 1995;333:234–40.
Juppner H, Brown EM, Kronenberg HM. Parathyroid hormone. In: Favus MJ, editor. Primer on the metabolic bone diseases and disorders of mineral metabolism. 4th ed. Philadelphia: Lippincott-Raven; 1999. p. 80–7.
Hordon LD, Peacock M. Osteomalacia and osteoporosis in femoral neck fracture. Bone Miner. 1990;11:247–59.
Chapuy MC, Arlot M, Duboeuf F, et al. Vitamin D3 and calcium to prevent hip fractures in elderly women. N Engl J Med. 1992;327:1627–42.
Dawson-Hughes B, Harris SS, Krall EA, Dallal GE. Effect of calcium and vitamin D supplementation on bone density in men and women 65 years of age or older. N Engl J Med. 1997;337:670–6.
Schnitzler CM, Solomon L. Osteomalacia in elderly white South African women with fractures of the femoral neck. S Afr Med J. 1983;64:527–30.
Al-Ali H, Fuleihan GEH. Nutritional osteomalacia: substantial clinial improvement and gain in bone density post-therapy. J Clin Densitom. 2000;3:97–101.
Malabanan AO, Turner AK, Holick MF. Severe generalized bone pain and osteoporosis in a premenopausal black female: effect of vitamin D replacement. J Clin Densitom. 1998;1:201–4.
Holick MF. Sunlight and vitamin D, both good for cardiovascular health (editorial). J Gen Intern Med. 2002;17:733–5.
Rimaniol J, Authier F, Chariot P. Muscle weakness in intensive care patients: initial manifestation of vitamin D deficiency. Intensive Care Med. 1994;20:591–2.
Bischoff HA, Stahelin HB, Dick W, et al. Effect of vitamin D and calcium supplementation on falls: a randomized controlled study. J Bone Miner Res. 2003;18(2):343–51.
Gloth FM, Gundberg CM, Hollis BW, Haddad HG, Tobin JD. Vitamin D deficiency in homebound elderly persons. JAMA. 1995;274:1683–6.
Malabanan A, Veronikis IE, Holick MF. Redefining vitamin D insufficiency. Lancet. 1998;351:805–6.
Lips P. Vitamin D, deficiency and secondary hyperparathyroidism in the elderly: consequences for bone loss and fractures and therapeutic implications. Endocr Rev. 2001;22(4):477–501.
Chapuy MC, Preziosi P, Maaner M, et al. Prevalence of vitamin D insufficiency in an adult normal population. Osteoporosis Int. 1997;7:439–43.
Tangpricha V, Pearce EN, Chen TC, Holick MF. Vitamin D insufficiency among free-living healthy young adults. Am J Med. 2002;112:659–62.
Hollis BW. Circulating 25-hydroxyvitamin D levels indicative of vitamin D sufficiency: implications for establishing a new effective dietary intake recommendation for vitamin D. J Nutr. 2005;135:317–22.
IOM (Institute of Medicine). Dietary reference intakes for calcium and vitamin D. Committee to review dietary reference intakes for calcium and vitamin D. Washington, DC: The National Academies Press; 2011.
Holick MF. Resurrection of vitamin D deficiency and rickets. J Clin Invest. 2006;116(8):2062–72.
Thomas MK, Lloyd-Jones DM, Thadhani RI, et al. Hypovitaminosis D in medical inpatients. N Engl J Med. 1998;338:777–83.
Hossein-nezhad A, Holick MF. Vitamin D for health: a global perspective. Mayo Clin Proc. 2013;88(7):720–55.
Lo CW, Paris PW, Clemens TL, Nolan J, Holick MF. Vitamin D absorption in healthy subjects and in patients with intestinal malabsorption syndromes. Am J Clin Nutr. 1985;42:644–9.
Shane E, Silverberg SJ, Donovan D, et al. Osteoporosis in lung transplantation candidates with end-stage pulmonary disease. Am J Med. 1996;101:262–9.
Shike M, Harrison J, Sturtridge C, et al. Metabolic bone disease in patients receiving long-term parenteral nutrition. Ann Intern Med. 1994;92:343–50.
Shike M, Shils ME, Heller A, et al. Bone disease in prolonged parenteral nutrition: osteopenia without mineralization defect. Am J Clin Nutr. 1986;44:89–98.
Chel VGM, Ooms ME, Popp-Snijders C, et al. Ultraviolet irradiation corrects vitamin D deficiency and suppresses secondary hyperparathyroidism in the elderly. J Bone Miner Res. 1998;13:1238–42.
Chuck A, Todd J, Diffey B. Subliminal ultraviolet-B irradiation for the prevention of vitamin D deficiency in the elderly: a feasibility study. Photochem Photoimmun Photomed. 2001;17(4):168–71.
Lund B, Sorensen OH. Measurement of 25-hydroxyvitamin D in serum and its relation to sunshine, age and Vitamin D intake in the Danish population. Scand J Clin Lab Invest. 1979;39:23–30.
Bell NH, Epstein S, Greene A, Shary J, Oexmann MJ, Shaw S. Evidence for alteration of the vitamin D-endocrine system in obese subjects. J Clin Invest. 1985;76:370–3.
Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, Murad MH, Weaver CM. Evaluation, treatment & prevention of vitamin D deficiency: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911–30.
Vieth R, Chan PC, MacFarlane GD. Efficacy and safety of vitamin D3 intake exceeding the lowest observed adverse effect level1-3. Am J Clin Nutr. 2001;73:288–94.
Barger-Lux MJ, Heaney RP, Dowell S, Chen TC, Holick MF. Vitamin D and its major metabolites: serum levels after graded oral dosing in healthy men. Osteopor Int. 1998;8:222–30.
Heaney RP, Davies KM, Chen TC, Holick MF, Barger-Lux MJ. Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am J Clin Nutr. 2003;77:204–10.
Chapuy MC, Schott AM, Garnero P, et al. Healthy elderly French women living at home have secondary hyperparathyroidism and high bone turnover in winter. J Clin Endocrinol Metab. 1996;81:1129–33.
Priemel M, von Domarus C, Klatte TO, Kessler S, Schlie J, Meier S, Proksch N, Pastor F, Netter C, Streichert T, Puschel K, Amling M. Bone mineralization defects and vitamin D deficiency: histomorphometric analysis of iliac crest bone biopsies and circulating 25-hydroxyvitamin D in 675 patients. J Bone Miner Res. 2010;25(2):305–12.
Koutkia P, Chen TC, Holick MF. Vitamin D intoxication associated with an over-the-counter supplement. N Engl J Med. 2001;345(1):66–7.
Bauer JM, Freyberg RH. Vitamin D intoxication and metastatic calcification. JAMA. 1946;1208–1215.
Adams JS, Lee G. Gains in bone mineral density with resolution of vitamin D intoxication. Ann Intern Med. 1997;127:203–6.
Jacobus CH, Holick MF, Shao Q, et al. Hypervitaminosis D associated with drinking milk. N Engl J Med. 1992;326:1173–7.
Pietras SM, Obayan BK, Cai MH, Holick MF. Research letter: vitamin D2 treatment for vitamin D deficiency and insufficiency for up to 6 years. Arch Intern Med. 2009;169(19):1806–8.
Demetriou ETW, Travison TG, Holick MF. Treatment with 50,000 IU vitamin D2 every other week and effect on serum 25-hydroxyvitamin D2, 25-hydroxyvitamin D3 and total 25-hydroxyvitamin D in a clinical setting. Endocr Pract. 2012;18(3):399–402.
Heikinheimo RJ, Ubjivaaram JA, Jantti PO, Maki-Jokela PL, Rajala SA, Sievanen H. Intermittant parenteral vitamin D supplementation in the elderly in nutritional aspects of osteoporosis. In: Burckhard P, Heaney RP, editors. Challenges of modern medicine. Geneva, Switzerland: Ares-Serono; 1994. p. 335–40.
Wacker M, Holick MF. Sunlight and vitamin D: a global perspective for health. DermatoEndocrinology. 2013;5(1):51–108.
Koutkia P, Lu Z, Chen TC, Holick MF. Treatment of vitamin D deficiency due to Crohn’s disease with tanning bed ultraviolet B radiation. Gastroenterology. 2001;121(6):1485–8.
Apperly FL. The relation of solar radiation to cancer mortality in North America. Cancer Res. 1941;1:191–5.
Garland CF, Garland FC, Shaw EK, Comstock GW, Helsing KJ, Gorham ED. Serum 25-hydroxyvitamin D and colon cancer: eight-year prospective study. Lancet. 1989;18:1176–8.
Garland C, Shekelle RB, Barrett-Connor E, Criqui MH, Rossof AH, Oglesby P. Dietary vitamin D and calcium and risk of colorectal cancer: a 19-year prospective study in men. Lancet. 1985;9:307–9.
Garland CF, Garland FC, Gorham ED, Raffa J. Sunlight, vitamin D, and mortality from breast and colorectal cancer in Italy. In: Holick MF, Kligman A, editors. Biologic effects of light. Walter de Gruyter: New York; 1992. p. 39–43.
Garland FC, Garland CF, Gorham ED, Young JF. Geographic variation in breast cancer mortality in the United States: a hypothesis involving exposure to solar radiation. Prevent Med. 1990;19:614–22.
Hanchette CL, Schwartz GG. Geographic patterns of prostate cancer mortality. Cancer. 1992;70:2861–9.
Ahonen MH, Tenkanen L, Teppo L, Hakama M, Tuohimaa P. Prostate cancer risk and prediagnostic serum 25-hydroxyvitamin D levels (Finland). Cancer Causes Control. 2000;11:847–52.
Grant WB. An ecologic study of dietary and solar ultraviolet-B links to breast carcinoma mortality rates. Am Cancer Soc. 2002;94:272–81.
Rostand SG. Ultraviolet light may contribute to geographic and racial blood pressure differences. Hypertension. 1979;30:150–6.
Hernan MA, Olek MJ, Ascherio A. Geographic variation of MS incidence in two prospective studies of US women. Neurology. 1999;51:1711–8.
Holick MF. Clinical efficacy of 1,25-dihydroxyvitamin D3 and its analogues in the treatment of psoriasis. Retinoids. 1998;14(1):12–7.
Mathieu C, Waer M, Laureys J, Rutgeerts O, Bouillon R. Prevention of autoimmune diabetes in NOD mice by 1,25 dihydroxyvitamin D3. Diabetologia. 1994;37:552–8.
Liu PT, Stenger S, Li H, Wenzel L, Tan BH, Krutzik S, Ochoa MT, Schauber J, Wu K, Meinken C, Kamen DL, Wagner M, Bals R, Steinmeyer A, Zugel U, Gallo RL, Eisenberg D, Hewison M, Hollis BW, Adams JS, Bloom BR, Modlin RL. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Sciencexpress. 2006;3:1770–3.
Hypponen E, Laara E, Jarvelin M-R, Virtanen SM. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet. 2001;358:1500–3.
Li YC, Kong J, Wei M, Chen ZF, Liu SQ, Cao LP. 1,25-Dihydroxyvitamin D3 is a negative endocrine regulator of the renin-angiotensin system. J Clin Invest. 2002;110:229–38.
Scragg R, Jackson R, Holdaway IM, Lim T, Beaglehole R. Myocaardial infarction is inversely associated with plasma 25-hydroxyvitamin D3 levels: a community-based study. Int J Epidemiol. 1990;19:559–63.
Zitterman A, Schulze Schleithoff S, Tenderich C, Berthold H, Koefer R, Stehle P. Low vitamin D status: a contributing factor in the pathogenesis of congestive heart failure? J Am Coll Cardiol. 2003;41(1):105–12.
Krause R, Buhring M, Hopfenmuller W, Holick MF, Sharma AM. Ultraviolet B and blood pressure. Lancet. 1998;352(9129):709–10.
Scragg R. Seasonality of cardiovascular disease mortality and the possible protective effect of utlra-violet radiation. Int J Epidemiol. 1981;10:337–41.
Schwartz GG, Whitlatch LW, Chen TC, Lokeshwar BL, Holick MF. Human prostate cells synthesize 1,25-dihydroxyvitamin D3 from 25-hydroxyvitamin D3. Cancer Epidemiol Biomarkers Prev. 1998;7(5):391–5.
Bikle DD, Nemanic MK, Gee E, Elias P. 1,25-Dihydroxyvitamin D3 production by human keratinocytes: kinetics and regulation. J Clin Invest. 1986;78:557–66.
Lehmann B, Knuschke P, Meurer M. UVB-induced conversion of 7-dehydrocholesterol to 1α,25-dihydroxyvitamin D3 (calcitriol) in the human keratinocyte line HaCaT. Photochem Photobiol. 2000;72(6):803–9.
Cross HS, Bareis P, Hofer H, Bischof MG, Bajna E, Kriwanek S. 25-Hydroxyvitamin D3-1α-hydroxylase and vitamin D receptor gene expression in human colonic mucosa is elevated during early cancerogenesis. Steroids. 2001;66:287–92.
Tangpricha V, Flanagan JN, Whitlatch LW, et al. 25-Hydroxyvitamin D-1a-hydroxylase in normal and malignant colon tissue. Lancet. 2001;357(9269):1673–4.
Holick MF. Sunlight D-lemma: risk of skin cancer or bone disease and muscle weakness. Lancet. 2001;357:4–6.
Acknowledgments
This work was supported in part by National Institutes of Health Grants CTSI UL-1-TR 000157.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this chapter
Cite this chapter
Holick, M.F. (2015). Vitamin D. In: Holick, M., Nieves, J. (eds) Nutrition and Bone Health. Nutrition and Health. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-2001-3_27
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2001-3_27
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-2000-6
Online ISBN: 978-1-4939-2001-3
eBook Packages: MedicineMedicine (R0)