Abstract
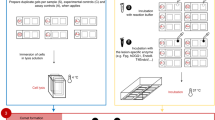
The rapid and enormous development of nanotechnology has been accompanied by a deep concern for the effects that nanoparticles may have on human health and the environment. In this context, it is essential to assess the ability of nanoparticles to cause DNA damage. Single cell gel electrophoresis assay (comet assay) is widely used for evaluating nanoparticle-induced DNA damage in cells and is the most used assay for genotoxicity testing of nanomaterial. Here, we describe the standard alkaline version of the comet assay, both in vitro and in vivo, as well as the lesion-specific enzyme-modified comet assay (for detection of oxidized DNA lesions) to test nanoparticles. We also highlight critical points that need to be taken into consideration when assessing nanomaterial genotoxicity.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Knaapen AM, Borm PJA, Albrecht C et al (2004) Inhaled particles and lung cancer. Part A: mechanisms. Int J Cancer 109:799–809
Donaldson K, Poland CA, Schins RPF (2010) Possible genotoxic mechanisms of nanoparticles: criteria for improved test strategies. Nanotoxicology 4:414–420
Magdolenova Z, Collins AR, Kumar A et al (2013) Mechanisms of genotoxicity. Review of recent in vitro and in vivo studies with engineered nanoparticles. Nanotoxicology 8(3):233–278. doi:10.3109/17435390.2013.773464
Bowman L, Castranova V, Ding M (2012) Single cell gel electrophoresis assay (comet assay) for evaluating nanoparticles-induced DNA damage in cells. Methods Mol Biol 906: 415–422
Karlsson HL (2010) The comet assay in nanotoxicology research. Anal Bioanal Chem 398: 651–666
Alenius H, Catalán J, Lindberg H et al (2014) Nanomaterials and human health. In: Vogel U, Savolainen K, Wu Q, van Tongeren M, Brouwer D, Berges M (eds) Handbook of nanosafety. Measurement, exposure and toxicology. Academic, San Diego
Dusinska M, Collins AR (1996) Detection of oxidised purines and UV-induced photoproducts in DNA, by inclusion of lesion-specific enzymes in the comet assay (single cell gel electrophoresis). Alternatives Lab Anim 24: 405–411
Magdolenova Z, Drlickova M, Henjum K, et al. (2014) Coating-dependent induction of cytotoxicity and genotoxicity of iron oxide nanoparticles. Nanotoxicology (in press)
Jha AN (2008) Ecotoxicological applications and significance of the comet assay. Mutagenesis 23:207–21
Doak SH, Manshian B, Jenkins GJS et al (2012) In vitro genotoxicity testing strategy for nanomaterials and the adaptation of current OECD guidelines. Mutat Res 745:104–111
Lovell DP, Omori T (2008) Statistical issues in the use of the comet assay. Mutagenesis 23: 171–182
Harris G, Palosaari T, Magdolenova Z, et al. (2014) Iron oxide nanoparticle toxicity testing using high throughput analysis and high content imaging. Nanotoxicology (in press)
Dusinska M, Fjellsbø LM, Magdolenova Z et al (2011) Safety of nanomaterial in nanomedicine. In: Hunter RJ, Preedy VR (eds) Nanomedicine in health and disease. CRC, Boca Raton
Dusinska M, Rundén-Pran E, Carreira SC et al (2012) In vitro and in vivo toxicity test methods. Chapter 4. Critical evaluation of toxicity tests. In: Fadeel B, Pietroiusti A, Shvedova A (eds) Adverse effects of engineered nanomaterials: exposure, toxicology and impact on human health. Elsevier, New York
Dusinska M, Magdolenova Z, Fjellsbø LM (2013) Toxicological aspects for nanomaterial in humans. In: Ogris M, Oupicky D (eds) Nanotechnology, methods in molecular biology. Humana, Totowa
Handy RD, van den Brink N, Chappell M et al (2012) Practical considerations for conducting ecotoxicity test methods with manufactured nanomaterials: what have we learnt so far? Ecotoxicology 21:933–72
Magdolenova Z, Bilaničová D, Pojana G et al (2012) Impact of agglomeration and different dispersions of titanium dioxide nanoparticles on the human related in vitro cytotoxicity and genotoxicity. J Environ Monit 14:455–64
Tulinska J, Kazimirova A, Kuricova M, et al. (2014) Immunotoxicity and genotoxicity testing of PLGA-PEO nanoparticles in human blood cell model. Nanotoxicology (in press)
Guadagnini R, Halamoda Kenzaoui B, et al. (2014) Toxicity screenings of nanomaterials: challenges due to interference with assay processes and components of classic in vitro tests. Nanotoxicology (in press)
Dusinska M, Collins A (2008) The Comet assay in human biomonitoring: gene–environment interactions. Mutagenesis 23:191–205
OECD (2008) OECD work on the safety of manufactured nanomaterials. Environment, health and safety division. Environment Directorate. OECD. www.oecd.org/dataoecd/54/27/41567645.ppt
Stone V, Nowack B, Baun A et al (2010) Nanomaterials for environmental studies: classification, reference material issues, and strategies for physico-chemical characterization. Sci Total Environ 408:1745–54
NanoGenotox project report. http://www.nanogenotox.eu/files/PDF/nanogenotox_web.pdf
Kroll A, Pillukat MH, Hahn D et al (2012) Interference of engineered nanoparticles with in vitro toxicity assays. Arch Toxicol 86: 1123–1136
Magdolenova Z, Lorenzo Y, Collins A et al (2012) Can standard genotoxicity tests be applied to nanoparticles? J Toxicol Environ Health A 75:800–806
Rajapakse K, Drobne D, Kastelec D et al (2013) Experimental evidence of false-positive Comet test results due to TiO2 particle-assay interactions. Nanotoxicology 7:1043–1051
Landsiedel R, Kapp MD, Schulz M et al (2009) Genotoxicity investigations on nanomaterials: methods, preparation and characterization of test material, potential artifacts and limitations—many questions, some answers. Mutat Res 681:241–458
Stone V, Johnston H, Schins RPF (2009) Development of in vitro systems for nanotoxicology: methodological considerations. Crit Rev Toxicol 39:613–626
Kain J, Karlsson HL, Möller L (2012) DNA damage induced by micro- and nanoparticle—interaction with FPG influences the detection of DNA oxidation in the comet assay. Mutagenesis 27:491–500
Tice R, Vasquez M (1999) Protocol for the application of the pH > 13 alkaline single cell gel (SCG) assay to the detection of DNA damage in mammalian cells. http://cometassay.com/index_files/Page290.htm
Collins AR (2004) The comet assay for DNA damage and repair: principles, applications, and limitations. Mol Biotechnol 26:249–261
OECD (2011) A steering group 7 case-study for hazard identification of inhaled nanomaterials: an integrated approach with short-term inhalation studies. ENV/CHEM/NANO(2011) 6/REV1. In: 10th meeting of the Working party on manufactured nanomaterials, OECD; 27–29 June 2012; Paris, France
Bright J, Aylott M, Bate S et al (2011) Recommendations on the statistical analysis of the Comet assay. Pharm Stat 10:485–493
OECD (2009). Preliminary review of OECD test guidelines for their applicability to manufactured nanomaterials. Environment Directorate, Organisation for Economic Co-operation and Development, Paris. OECD Environment, Health and Safety Publications Series on the Safety of Manufactured Nanomaterials, No. 15
Collins AR, Dušinská M, Mišľanová C, among other ESCODD members (2002) Inter-laboratory validation of procedures for measuring 8-oxo-7,8-dihydroguanine/8-oxo-7,8-digydro-2′-deoxyguanosine in DNA. Free Radic Res 36:239–245
Juillerat L, Dusinska M, Fjellesbo LM, et al. (2014) Biological impact assessment of nanomaterial used in nanomedicine. Introduction to the NanoTEST project. Nanotoxicology (in press)
ECHA (2012) Guidance on information requirements and chemical safety assessment. Appendix R7-1 Recommendations for nanomaterials applicable to Chapter R7a Endpoint specific guidance. European Chemicals Agency. http://echa.europa.eu/documents/10162/13632/appendix_r7a_nanomaterials_en.pdf
Acknowledgments
We thank Dr. Jonas Nygren for providing useful advice when developing our in vivo approach.
We thank Andrew R Collins, Zuzana Magdolenova, Lise M Fjellsbø, Elise Runden-Pran for adopting protocol and preparation of standard operating procedure. We also thank Mr. Leszek Huk for making plate for incubation slides and Mr. Michal Zagrodzki for helping with the graphic design.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Catalán, J., Suhonen, S., Huk, A., Dusinska, M. (2014). Analysis of Nanoparticle-Induced DNA Damage by the Comet Assay. In: Sierra, L., Gaivão, I. (eds) Genotoxicity and DNA Repair. Methods in Pharmacology and Toxicology. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1068-7_14
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1068-7_14
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1067-0
Online ISBN: 978-1-4939-1068-7
eBook Packages: Springer Protocols