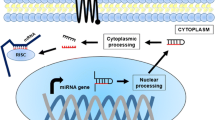
Abstract
A growing body of evidence suggests that noncoding RNA (ncRNA) transcripts play a fundamental role in regulating gene expression via targeting epigenetic modifications to particular loci in the genome. Classical examples of such regulation are X-chromosome inactivation and genomic imprinting; however it is now clear that ncRNAs exert their influence over a wider array of genes throughout the metazoan genome. Accumulating evidence suggests that the ncRNAs act as guides for epigenetic silencing complexes to specific sites within the genome. Those ncRNAs involved in regulating the expression of particular protein-coding genes offer panoply of targets that when suppressed can result in derepression or activation of the ncRNA-targeted locus. Recent work has determined the underlying mechanisms involved in ncRNA-targeted epigenetic regulation in a subset of genes. These findings have resulted in a paradigm shift whereby targeted gene activation can be achieved, by targeting endogenous regulatory ncRNAs, producing potential novel treatments for genetic and infectious diseases where increases in gene expression are required.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Lee J (2012) Epigenetic regulation by long noncoding RNAs. Science 338:1435–1439
Ponting C (2008) The functional repertoires of metazoan genomes. Nat Rev Genet 9:689–698
Morris K (2009) RNA-directed transcriptional gene silencing and activation in human cells. Oligonucleotides 19:299–306
Kung J, Colognori D, Lee J (2013) Long noncoding RNAs: past, present, and future. Genetics 193:651–669
Morris K (2009) Non-coding RNAs, epigenetic memory and the passage of information to progeny. RNA Biol 6(3):242–247
Rassoulzadegan M, Grandjean V, Gounon P, Vincent S, Gillot I, Cuzin F (2006) RNA-mediated non-mendelian inheritance of an epigenetic change in the mouse. Nature 441:469–474
Davidson B, Fasman GD (1969) The single-stranded polyadenylic acid-poly-L-lysine complex. A conformational study and characterization. Biochemistry 8:4116–4126
Jacob F, Monod J (1961) Genetic regulatory mechanisms in the synthesis of proteins. J Mol Biol 3:318–356
Banfai B, Jia H, Khatun J, Wood E, Risk B, Gundling WE Jr, Kundaje A, Gunawardena HP, Yu Y, Xie L, Krajewski K, Strahl BD, Chen X, Bickel P, Giddings MC, Brown JB, Lipovich L (2012) Long noncoding RNAs are rarely translated in two human cell lines. Genome Res 22:1646–1657
Carninci P, Kasukawa T, Katayama S, Gough J, Frith M, Maeda N, Oyama R, Ravasi T, Lenhard B, Wells C, Kodzius R, Shimokawa K, Bajic V, Brenner S, Batalov S, Forrest AR, Zavolan M, Davis M, Wilming L, Aidinis V, Allen J, Ambesi-Impiombato A, Apweiler R, Aturaliya R, Bailey T, Bansal M, Baxter L, Beisel K, Bersano T, Bono H, Chalk A, Chiu K, Choudhary V, Christoffels A, Clutterbuck D, Crowe M, Dalla E, Dalrymple B, de Bono B, Della Gatta G, di Bernardo D, Down T, Engstrom P, Fagiolini M, Faulkner G, Fletcher C, Fukushima T, Furuno M, Futaki S, Gariboldi M, Georgii-Hemming P, Gingeras T, Gojobori T, Green R, Gustincich S, Harbers M, Hayashi Y, Hensch T, Hirokawa N, Hill D, Huminiecki L, Iacono M, Ikeo K, Iwama A, Ishikawa T, Jakt M, Kanapin A, Katoh M, Kawasawa Y, Kelso J, Kitamura H, Kitano H, Kollias G, Krishnan S, Kruger A, Kummerfeld S, Kurochkin I, Lareau L, Lazarevic D, Lipovich L, Liu J, Liuni S, McWilliam S, Madan Babu M, Madera M, Marchionni L, Matsuda H, Matsuzawa S, Miki H, Mignone F, Miyake S, Morris K, Mottagui-Tabar S, Mulder N, Nakano N, Nakauchi H, Ng P, Nilsson R, Nishiguchi S, Nishikawa S (2005) The transcriptional landscape of the mammalian genome. Science 309:1559–1563
Amaral P, Mattick J (2008) Noncoding RNA in development. Mamm Genome 19:454–492
Lucchesi J, Kelly W, Panning B (2005) Chromatin remodeling in dosage compensation. Annu Rev Genet 39:615–651
Lee J (2010) The X as model for RNA’s niche in epigenomic regulation. Cold Spring Harb Perspect Biol 2:a003749
Wutz A (2003) RNAs templating chromatin structure for dosage compensation in animals. Bioessays 25:434–442
Lyon MF (1961) Gene action in the X-chromosome of the mouse (Mus musculus L.). Nature 190:372–373
Brown C, Lafreniere R, Powers V, Sebastio G, Ballabio A, Pettigrew A, Ledbetter D, Levy E, Craig I, Willard H (1991) Localization of the X inactivation centre on the human X chromosome in Xq13. Nature 349:82–84
Rastan S, Robertson EJ (1985) X-chromosome deletions in embryo-derived (EK) cell lines associated with lack of X-chromosome inactivation. J Embryol Exp Morphol 90:379–388
Cattanach B, Isaacson J (1967) Controlling elements in the mouse X chromosome. Genetics 57:331–346
Kay GF, Barton SC, Surani MA, Rastan S (1994) Imprinting and X chromosome counting mechanisms determine Xist expression in early mouse development. Cell 77:639–650
Lyon MF (1999) Imprinting and X-chromosome inactivation. Results Probl Cell Differ 25:73–90
Avner P, Heard E (2001) X-chromosome inactivation: counting, choice and initiation. Nat Rev Genet 2:59–67
Boumil RM, Lee JT (2001) Forty years of decoding the silence in X-chromosome inactivation. Hum Mol Genet 10:2225–2232
Namekawa SH, Payer B, Huynh KD, Jaenisch R, Lee JT (2010) Two-step imprinted X inactivation: repeat versus genic silencing in the mouse. Mol Cell Biol 30:3187–3205
Xu N, Donohoe ME, Silva SS, Lee JT (2007) Evidence that homologous X-chromosome pairing requires transcription and Ctcf protein. Nat Genet 39:1390–1396
Xu N, Tsai CL, Lee JT (2006) Transient homologous chromosome pairing marks the onset of X inactivation. Science 311:1149–1152
Bacher C, Guggiari M, Brors B, Augui S, Clerc P, Avner P, Eils R, Heard E (2006) Transient colocalization of X-inactivation centres accompanies the initiation of X inactivation. Nat Cell Biol 8:293–299
Edwards C, Ferguson-Smith A (2007) Mechanisms regulating imprinted genes in clusters. Curr Opin Cell Biol 19:281–289
Sleutels F, Barlow DP (2002) The origins of genomic imprinting in mammals. Adv Genet 46:119–163
Bartolomei MS, Ferguson-Smith AC (2011) Mammalian genomic imprinting. Cold Spring Harb Perspect Biol. 3(7). pii: a002592. doi: 10.1101/cshperspect.a002592
Lee JT (2003) Molecular links between X-inactivation and autosomal imprinting: X-inactivation as a driving force for the evolution of imprinting? Curr Biol 13(6):R242–54
Cai X, Cullen B (2007) The imprinted H19 noncoding RNA is a primary microRNA precursor. RNA 13:313–316
Kevin V, M. (2009) RNA-directed control of transcription in human cells: specifically turning genes ON or OFF. Gene Ther Reg 04
Weinberg M, Morris K (2013) Long non-coding RNA targeting and transcriptional de-repression. Nucleic Acid Ther 23:9–14
Leung AK, Sharp PA (2006) Function and localization of microRNAs in mammalian cells. Cold Spring Harb Symp Quant Biol 71:29–38
Turner AM, Morris KV (2010) Controlling transcription with noncoding RNAs in mammalian cells. Biotechniques 48:ix–xvi
Ackley A, Lenox A, Stapleton K, Knowling S, Lu T, Sabir KS, Vogt PK, Morris KV (2013) An algorithm for generating small RNAs capable of epigenetically modulating transcriptional gene silencing and activation in human cells. Mol Ther Nucleic Acids 2:e104
Hawkins P, Santoso S, Adams C, Anest V, Morris K (2009) Promoter targeted small RNAs induce long-term transcriptional gene silencing in human cells. Nucleic Acids Res 37:2984–2995
Kim DH, Villeneuve LM, Morris KV, Rossi JJ (2006) Argonaute-1 directs siRNA-mediated transcriptional gene silencing in human cells. Nat Struct Mol Biol 13:793–797
Suzuki K, Juelich T, Lim H, Ishida T, Watanebe T, Cooper DA, Rao S, Kelleher AD (2008) Closed chromatin architecture is induced by an RNA duplex targeting the HIV-1 promoter region. J Biol Chem 283:23353–23363
Weinberg MS, Villeneuve LM, Ehsani A, Amarzguioui M, Aagaard L, Chen ZX, Riggs AD, Rossi JJ, Morris KV (2006) The antisense strand of small interfering RNAs directs histone methylation and transcriptional gene silencing in human cells. RNA 12:256–262
Han J, Kim D, Morris K (2007) Promoter-associated RNA is required for RNA-directed transcriptional gene silencing in human cells. Proc Natl Acad Sci U S A 104:12422–12427
Knowling S, Morris KV (2011) Epigenetic regulation of gene expression in human cells by noncoding RNAs. Prog Mol Biol Transl Sci 102:1–10
Hawkins P, Morris K (2008) RNA and transcriptional modulation of gene expression. Cell Cycle 7:602–607
Johnsson P, Ackley A, Vidarsdottir L, Lui WO, Corcoran M, Grander D, Morris KV (2013) A pseudogene long-noncoding-RNA network regulates PTEN transcription and translation in human cells. Nat Struct Mol Biol 20(4):440–446
Braunschweig M, Jagannathan V, Gutzwiller A, Bee G (2012) Investigations on transgenerational epigenetic response down the male line in F2 pigs. PLoS One 7:e30583
Janowski B, Younger S, Hardy D, Ram R, Huffman K, Corey D (2007) Activating gene expression in mammalian cells with promoter-targeted duplex RNAs. Nat Chem Biol 3:166–173
Li LC, Okino ST, Zhao H, Pookot D, Place RF, Urakami S, Enokida H, Dahiya R (2006) Small dsRNAs induce transcriptional activation in human cells. Proc Natl Acad Sci U S A 103:17337–17342
Morris KV, Santoso S, Turner AM, Pastori C, Hawkins PG (2008) Bidirectional transcription directs both transcriptional gene activation and suppression in human cells. PLoS Genet 4:e1000258
Schwartz JC, Younger ST, Nguyen NB, Hardy DB, Monia BP, Corey DR, Janowski BA (2008) Antisense transcripts are targets for activating small RNAs. Nat Struct Mol Biol 15:842–848
Yu W, Gius D, Onyango P, Muldoon-Jacobs K, Karp J, Feinberg AP, Cui H (2008) Epigenetic silencing of tumour suppressor gene p15 by its antisense RNA. Nature 451:202–206
Hawkins PG, Morris KV (2010) Transcriptional regulation of Oct4 by a long non-coding RNA antisense to Oct4-pseudogene 5. Transcription 1:165–175
Modarresi F, Faghihi M, Lopez-Toledano M, Fatemi R, Magistri M, Brothers S, van der Brug M, Wahlestedt C (2012) Inhibition of natural antisense transcripts in vivo results in gene-specific transcriptional upregulation. Nat Biotechnol 30:453–459
Acknowledgements
The project was supported by NIHLB R01AI084406, NIAID R56 AI096861-01, and PO1 AI099783-01 to KVM.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Damski, C., Morris, K.V. (2014). Targeted Small Noncoding RNA-Directed Gene Activation in Human Cells. In: Werner, A. (eds) Animal Endo-SiRNAs. Methods in Molecular Biology, vol 1173. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-0931-5_1
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0931-5_1
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-0930-8
Online ISBN: 978-1-4939-0931-5
eBook Packages: Springer Protocols