Abstract
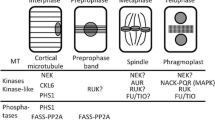
Reorganization of microtubules during cell cycle depends on the modulation of activity of microtubule-associated proteins. MAP65 is one of the main microtubule structural proteins in plants responsible for the formation of bundles of parallel and antiparallel microtubules. A member of MAP65 protein family, MAP65-1, binds to microtubules of preprophase band during early stages of cell division and later to the midzone of anaphase spindle and the phragmoplast, but exhibits no or reduced microtubule binding during metaphase. Artificially induced interaction of MAP65-1 with microtubules during metaphase promotes excessive formation of pole-to-pole microtubule bundles and causes delay of anaphase onset. The exact mechanism of this delay is not known, but it was suggested that microtubule bundles induced by MAP65 impose spatial constraints on the chromosome movement obstructing their alignment in the metaphase plate. Interaction of MAP65-1 with microtubules is controlled by phosphorylation. This chapter describes a strategy for the identification of phosphorylation residues responsible for the cell-cycle control of MAP65-1 activity.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Hussey PJ, Hawkins TJ, Igarashi H, Kaloriti D, Smertenko A (2002) The plant cytoskeleton: recent advances in the study of the plant microtubule-associated proteins MAP-65, MAP-190 and the Xenopus MAP215-like protein, MOR1. Plant Mol Biol 50:915–924
Chan J, Jensen CG, Jensen LCW, Bush M, Lloyd CW (1999) The 65-kDa carrot microtubule-associated protein forms regularly arranged filamentous cross-bridges between microtubules. Proc Natl Acad Sci U S A 96:14931–14936
Janson ME, Loughlin R, Loïodice I, Fu C, Brunner D, Nedelec FJ, Tran PH (2007) Crosslinkers and motors organize dynamic microtubules to form stable bipolar arrays in fission yeast. Cell 128:357–368
Gaillard J, Neumann E, Van Damme D, Stoppin-Mellet V, Ebel C, Barbier E, Geelen D, Vantard M (2008) Two microtubule-associated proteins of Arabidopsis MAP65s promote antiparallel microtubule bundling. Mol Biol Cell 19:4534–4544
Pellman D, Bagget M, Tu H, Fink GR (1995) Two microtubule-associated proteins required for anaphase spindle movement in Saccharomyces cerevisiae. J Cell Biol 130:1375–1385
Mollinari C, Kleman JP, Jiang W, Schoehn G, Hunter T, Margolis RL (2002) PRC1 is a microtubule binding and bundling protein essential to maintain the mitotic spindle midzone. J Cell Biol 157:1175–1186
Schuyler SC, Liu JY, Pellman DJ (2003) The molecular function of Ase1p: evidence for a MAP-dependent midzone-specific spindle matrix. J Cell Biol 160:517–528
Kurasawa Y, Earnshaw WC, Mochizuki Y, Dohmae N, Todokoro N (2004) Essential roles of KIF4 and its binding partner PRC1 in organized central spindle midzone formation. EMBO J 23:3237–3248
Müller S, Smertenko A, Wagner V, Heinrich M, Hussey PJ, Hauser M-T (2004) The plant microtubule associated protein, AtMAP65-3/PLE, is essential for cytokinetic phragmoplast function. Curr Biol 14:412–417
Verbrugghe KJC, White JG (2004) SPD-1 is required for the formation of the spindle midzone but is not essential for the completion of cytokinesis in C. elegans embryos. Curr Biol 14:1755–1760
Verni F, Somma MP, Gunsalus KC, Bonaccorsi S, Belloni B, Goldberg ML, Gatti M (2004) Feo, the Drosophila homolog of PRC1, is required for central-spindle formation and cytokinesis. Curr Biol 14:1569–1575
Smertenko A, Saleh N, Igarashi H, Mori H, Hauser-Hahn I, Jiang CJ, Sonobe S, Lloyd CW, Hussey PJ (2000) A new class of microtubule-associated proteins in plants. Nat Cell Biol 2:750–753
Smertenko AP, Chang HY, Wagner V, Kaloriti D, Fenyk S, Sonobe S, Lloyd C, Hauser MT, Hussey PJ (2004) The Arabidopsis microtubule-associated protein AtMAP65-1: molecular analysis of its microtubule bundling activity. Plant Cell 16:2035–2047
Chang H-Y, Smertenko AP, Igarashi H, Dixon DP, Hussey PJ (2005) Dynamic interaction of NtMAP65-1a with microtubules in vivo. J Cell Sci 118:3195–3201
Mao G, Chan J, Calder G, Doonan JH, Lloyd CW (2005) Modulated targeting of GFP-AtMAP65-1 to central microtubules during division. Plant J 43:469–478
Smertenko AP, Chang HY, Sonobe S, Fenyk SI, Weingartner M, Bogre L, Hussey PJ (2006) Control of the AtMAP65-1 interaction with microtubules through the cell cycle. J Cell Sci 119:3227–3237
Bögre L, Calderini O, Binarova P, Mattauch M, Till S, Kiegerl S, Jonak C, Pollaschek C, Barker P, Huskisson NS, Hirt H, Heberle-Bors E (1999) A MAP kinase is activated late in plant mitosis and becomes localized to the plane of cell division. Plant Cell 11:101–113
Weingartner M, Binarova P, Drykova D, Schweighofer A, David J-P, Heberle-Bors E, Doonan J, Bögre L (2001) Dynamic recruitment of Cdc2 to specific microtubule structures during mitosis. Plant Cell 13:1929–1943
Samaj J, Baluska F, Hirt H (2004) From signal to cell polarity: mitogen-activated protein kinases as sensors and effectors of cytoskeleton dynamicity. J Exp Bot 55:189–198
Hemsley R, McCutcheon S, Doonan J, Lloyd C (2001) P34(cdc2) kinase is associated with cortical microtubules from higher plant protoplasts. FEBS Lett 508:157–161
Bögre L, Zwerger K, Meskiene I, Binarova P, Csizmadia V, Planck C, Wagner E, Hirt H, HeberleBors E (1997) The cdc2Ms kinase is differently regulated in the cytoplasm and in the nucleus. Plant Physiol 113:841–852
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Smertenko, A. (2014). Determination of Phosphorylation Sites in Microtubule Associated Protein MAP65-1. In: Komis, G., Šamaj, J. (eds) Plant MAP Kinases. Methods in Molecular Biology, vol 1171. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-0922-3_13
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0922-3_13
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-0921-6
Online ISBN: 978-1-4939-0922-3
eBook Packages: Springer Protocols