Abstract
Despite improvement in some chronic kidney disease (CKD) metabolic derangements of bone and mineral metabolism after kidney transplantation, bone disease remains a significant problem. Kidney transplant recipients are at increased risk of bone mineral density (BMD) loss, fractures, and osteonecrosis, all of which are associated with substantial morbidity [1–11]. The pathogenesis of post-transplant bone disease is multifactorial [12–32], including but not limited to interactions of the complex abnormalities in pre-transplant mineral homeostasis [12] and renal osteodystrophy [24, 25], the degree of persistently impaired kidney function, peri-transplant alternations in calciotropic and phosphotropic regulatory hormonal pathways [26], and the potential deleterious skeletal effects of immunosuppressive therapy [27–32].
Management of post-transplant bone disease is challenging given the limitations of the available diagnostic methods and the lack of proven benefit of available therapies to reduce fractures [33]. Despite the evidence that both bisphosphonates and vitamin D receptor agonists (VDRA) attenuate the loss in BMD following successful kidney transplantation, these agents have not been clearly shown to reduce fractures [33–49]. Moreover, there is no consensus on how to monitor the response to therapy and the optimal duration of therapy is unknown. Therefore, therapeutic decisions should be guided by consideration of the interplay of multiple factors including the severity of BMD loss, the presence of risk factors for osteoporotic fragility fractures, disordered mineral metabolism, underlying bone disorder, and the level of kidney function. This chapter provides an approach to screening and diagnosis of post-transplant bone disease and an overview of treatment options.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Bone Mineral Density
- Chronic Kidney Disease
- Bisphosphonate Therapy
- Kidney Transplant Recipient
- Renal Osteodystrophy
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Introduction of Post-transplant Bone Loss
The pathogenesis of post-transplant bone disease is multifactorial (Fig. 24.1). Bone mineral density (BMD) loss is common after kidney transplantation, particularly in the first post-transplant year [1–3] and is more pronounced in the lumbar spine than the femoral neck [50]. The rate of decline in BMD during the first 6–12 months post-transplantation ranges between 3 and 9 % [8–11, 45, 46]. The diminished degree of bone loss observed in some of the more recent studies may be a consequence of the use of lower glucocorticoid doses [10, 45, 46, 51]. However, bone loss persists after the first-year post-transplant as BMD reductions continue at rates of 1–2 % per year [52, 53]. Kidney transplant recipients are at 4.5-fold greater risk of fractures in comparison to the general population [4, 5, 7]. They have a 30 % higher risk of hip fracture during the first 3 years post-transplantation in comparison to dialysis patients on the waiting list for kidney transplant [54]. Post-kidney transplant fractures tend to occur more commonly in cortical bone with lower extremity fractures accounting for approximately 35 %, followed by fractures in the ribs, upper extremities, and vertebrae [4]. Unfortunately, measurements of BMD or trends of decreasing BMD have not been predictive of fractures [55, 56].
Osteonecrosis is another major debilitating bone complication seen in transplant patients that is associated with chronic glucocorticoid use [6]. Osteonecrosis generally occurs in weight-bearing bones like the femoral head and knees. Most cases of osteonecrosis occur in the first year following transplant [6]. The reported incidence of osteonecrosis in the femoral head among kidney transplant recipients ranges between 4 and 25 % [57–60], with lower occurrences associated with decreased cumulative glucocorticoid use [61].
Screening Diagnosis and Monitoring of Post-transplant Bone Disease
Biochemical Evaluation
Determinations of calcium, phosphate, vitamin D (25-hydroxycholecalciferol), and parathyroid hormone (PTH) concentrations are used to define, monitor, and target treatment of post-transplant CKD-mineral bone disorders (CKD-MBD) [62, 63]. Table 24.1 provides recommendations on the frequency of monitoring biochemical parameters of post-transplant CKD-MBD (based on KDIGO and KDOQI guidelines) with more frequent monitoring advised during the first 12 weeks [12, 56, 64]. Additionally, serum vitamin D should be checked in the early post-transplant period [64]. Unfortunately, the biochemical targets representing optimal management of post-transplant bone CKD-MBD have not yet been defined. In the absence of studies specific to transplant patients, targets tailored to the degree of renal impairment seem reasonable. Though it has been recognized that PTH concentrations are a poor discriminator of bone turnover in CKD, they are even less reliable in the transplant patient [65].
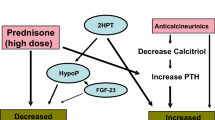
The risk factors and pathogenesis of post-transplant bone loss and post-transplant fracture: the pathogenesis of post-transplant bone disease is multifactorial including but not limited to pre-transplant-related factors, transplant-related factors, and factors that increase the risk of osteoporosis and fractures. All these factors result in uncoupling bone formation and bone resorption with higher rate of the latter and subsequently increase bone loss and bone fractures. CKD chronic kidney disease, PTH parathyroid hormone, BMI body mass index [9–29]
Bone Biomarkers: Biomarkers of bone formation (bone-specific alkaline phosphatase, osteocalcin, procollagen C-terminal propeptide (PICP), and procollagen N-terminal PINP) and bone resorption (urinary collagen breakdown products, TRAP5b) have been used to predict osteoporosis and fractures in the normal population. However, these biomarkers correlate poorly with bone loss as measured by densitometry and bone histology in the transplant patient and have not been shown to predict fracture risk. Until further information is available, there is probably no utility to measure these markers in the post-transplant patient.
Radiographic Evaluation
Densitometry
Bone densitometry, most commonly performed by dual-energy X-ray absorptiometry (DXA), is a readily available, noninvasive, rapid, cost-effective, and relatively precise method of screening BMD with low radiation exposure. However, the usefulness of DXA scans in patients with CKD and post-transplantation is limited because they do not reflect bone quality, turnover, or architecture [66]. The scans do not distinguish the underlying cause of bone loss. Low BMD measurements in post-kidney transplant recipients may be a consequence of osteoporosis and/or osteodystrophic bone disease [67]. The World Health Organization (WHO) [68] criteria defining fracture risk for osteopenia and osteoporosis are based on studies performed in normal postmenopausal Caucasian populations, thus limiting their applicability to kidney transplant recipients. Furthermore, there is no proven predictive relationship between BMD and fracture risk in kidney transplant recipients; in fact at least one study demonstrated that BMD did not distinguish those who went on to experience a fracture from those who did not [55]. BMD measurements in kidney transplant patients should not be looked at in isolation but should be analyzed and considered in the setting of a clinical assessment with biochemical data and possibly bone histology. However despite all of these limitations, BMDs may have a role in the transplant population. Sequential measurements of BMD may be helpful to guide antiresorptive therapy. KDIGO guidelines recommend measuring BMD at the time of transplantation or within the first 3 months after transplantation [23, 56, 69] when estimated GFR is >30 mL/min/m2, particularly in patients with risk factors for rapid bone loss. However, KDIGO does not recommend DXA scans in patients with eGFR < 30 mL/min/1.73 m2 as they are of no proven benefit in predicting fracture risk or the type of post-transplant bone disease [56]. DXA scans can be repeated at 12-month intervals if BMD decreases by greater than 5 % or half of a standard deviation; otherwise, DXA scan can be obtained every 2 years [70] to monitor response to therapy or to help decide whether therapy should be continued.
Conventional Radiographic Studies
Conventional radiography is helpful in identifying fractures, however is of limited value in distinguishing the different bone lesions of renal osteodystrophy. Lateral spine radiographs may be obtained to screen for asymptomatic spinal fragility fractures.
High-Resolution Quantitative Computed Tomography
High-resolution quantitative computed tomography (HR-QCT) is an accurate, noninvasive method of measuring volumetric BMD. It provides high-resolution three-dimensional images with information about volumetric BMD and microarchitecture with separation between cortical and trabecular bone [71]. Changes in volumetric vertebral BMD were found to correlate with changes detected in trabecular bone volume by bone biopsy [71]. Additionally, the trabecular microarchitecture in the radius and tibia using HR-QCT appears to better predict fracture risk in postmenopausal women [72] and is better than DXA in determining risk of fracture in patients with CKD [73, 74]. Unfortunately, this technology is not yet widely available, is costly, and has the disadvantage of high radiation exposure. The utility of this technique in the evaluation of post-transplant bone disease needs to be further evaluated before used outside of the research environment.
Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) is a noninvasive screening and diagnostic modality for the early detection of osteonecrosis [75]. The newest generation of high-resolution MRI scanners can also be used to evaluate bone microarchitecture, resulting in images that may represent a “virtual bone biopsy” [76]. A pilot study of micro-MRI in 17 young adults on maintenance hemodialysis compared with healthy controls demonstrated cortical thinning and disruption of the trabecular architecture in the former [77]. Two other preliminary studies have demonstrated that transplant recipients had significantly lower MRI measurements of bone volume and microarchitecture compared with controls; however, spine QCT, trabecular BMD, and DXA hip and spine BMD did not differ between transplant recipients and controls [78, 79] Micro-MRI appears to be a promising tool; however, more data are required to determine its utility in assessing bone disease in the post-transplant patient.
General Measures
The goals of therapy for post-transplant bone disease include increasing bone mass to avoid fractures, normalizing bone turnover, minimizing the risk of osteonecrosis, and optimizing calciotropic and phosphotropic mineral metabolism. Prevention and treatment of post-transplant bone loss and fracture reduction should begin in the pre-transplant period by following preventive measures that are of proven benefit in both the CKD and general population [80], such as limiting the use of steroids, engaging in 30 min of daily vigorous physical exercise, avoiding nicotine and alcohol consumption, and fall risk reduction. Management of the disorders associated with CKD-MBD including hyperparathyroidism, calcium and phosphorus derangements, and treatment of vitamin D insufficiency and deficiency is appropriate.
In the post-transplant period, calcium and vitamin D supplementation can be considered in all kidney transplant recipients who do not have hypercalcemia; however, the effects of vitamin D on BMD in post-transplant recipients are still controversial [51, 81]. Although short-term use of gonadal hormone therapy in solid organ transplant recipients with hypogonadism may slow bone loss, this therapy may increase the risk of coronary artery disease, cancer, and thrombotic events and should not be used routinely [82]. Therapy of post-transplant hypophosphatemia should be considered only in symptomatic patients or patients with serum levels below 1–1.5 mg/dL, given the risk of hypocalcemia, nephrocalcinosis [83], and persistent hyperparathyroidism [84]. When used phosphate supplementation should be closely monitored.
Glucocorticoids Minimizing Protocols
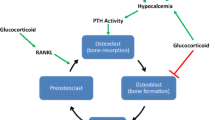
The use of high steroid doses in the early post-transplant period has been associated with a substantial and accelerated bone loss in the first 6–12 months post-kidney transplant [1–3, 69, 85]. In the short term, glucocorticoids profoundly inhibit bone formation and increase bone resorption. With long-term exposure, steroids are associated with low bone turnover and increased apoptosis of both osteoblasts and osteoclasts [86] (Fig. 24.2). The increasing use of steroid-free immunosuppression [87] with steroid withdrawal initiated at a variety of post-transplant time points including 4 days, 3 or 12 months has been associated with preserved and improved BMD at the femoral neck and lumbar spine [88–90]. In addition, minimizing steroid therapy has resulted in less osteonecrosis [61]. To date, there are no data linking the improvement in BMD observed in transplant patients withdrawn from steroids with a reduction in fracture risk. Some observational studies suggest that steroid-free regimens [87] and early steroid withdrawal (discharge from the hospital off steroids after renal transplantation) [88] are associated with decreased incident fractures after the first-year post-transplantation. This finding is not uniform as at least one observational study demonstrated that steroid withdrawal 2–6 months after solid organ transplantation did not result in decreased fractures [91]. Prospective randomized controlled trials are needed to evaluate the role of steroid free regimens or the timing of steroid free regimens withdrawal on fracture risk reduction.
Antiresorptive Therapy
Antiresorptive therapy may be indicated for patients with osteopenia or osteoporosis, who have no evidence of low bone turnover or refractory hyperparathyroidism, and preserved renal function (eGFR > 35 mL/min) in the first post-transplant year [56]. Consideration of antiresorptive therapy use is warranted if the patient is on a steroid-based immunosuppression protocol and/or has a history of fragility fractures [23].
Bisphosphonates
Bisphosphonates are synthetic analogues of pyrophosphates that bind to hydroxyapatite in the bone and directly inhibit osteoclastic bone resorption [92–94]. Bisphosphonates also reduce osteoclastic activity by decreasing osteoclast progenitor development and recruitment and by promoting osteoclast apoptosis [92–94]. As well bisphosphonates prevent osteocyte and osteoblast apoptosis [95]. Data from clinical trials show that both oral and intravenous bisphosphonates prevent bone loss in the lumbar spine and femoral neck in the first post-transplant year [33–43] and beyond [55]. There is a lack of evidence, however, that bisphosphonate use has any beneficial effect on bone strength or fracture prevention in the kidney transplant population. This lack of supporting data may be because of the underpowered nature of studies undertaken to examine this issue. When dosed appropriately in properly selected patients and used for limited periods of time, bisphosphonate therapy is well tolerated, safe, and effective for the prevention and management of post-transplant bone loss. Intravenous zoledronic acid or ibandronate can be an alternative to oral bisphosphonates in patients who cannot tolerate the oral formulation or have difficulty with the dosing requirements. Bisphosphonates should be used with caution in patients with reduced kidney function and their use should be avoided in patients with eGFR of less than 30–35 mL/min, given the prolonged duration of action and accumulation in bone resulting in the possibility of inducing or perpetuating adynamic bone disease [40, 96]. To reiterate, though bisphosphonates have been demonstrated to attenuate bone loss following transplantation, they have not been shown to reduce fractures. In fact, one study demonstrated increased fractures in patients receiving bisphosphonate therapy [55]. Bisphosphonate therapy is generally contraindicated in patients with evidence of low bone turnover [40], and obtaining a bone biopsy before institution of bisphosphonate therapy is the most accurate means of characterizing bone turnover and avoiding its use in adynamic bone states.
A Cochrane review of 24 trials, including 1,209 kidney transplant recipients, evaluating the risks and benefits of bisphosphonates, vitamin D analogues, and calcitonin versus no treatment found that all of the interventions provided clinically significant prevention of bone loss in both the femoral neck and lumbar spine, with the exception of calcitonin [38] which did not improve femoral neck BMD [33]. Bisphosphonates were superior to calcitriol in preserving BMD. No single therapy was found to prevent fractures; however, lack of demonstration does not necessarily mean they do not reduce fractures, because as noted earlier, none of these trials were powered to detect a reduction in fracture risk. However, combining the effect of all therapies versus no intervention, there was a 49 % reduction in the relative fracture risk (confidence interval 0.27–0.99). In another meta-analysis by Stein et al., 11 trials evaluated fracture incidence in 780 solid organ transplant recipients (4 of the 11 trials and 42 % of the patients had kidney transplants) that received either bisphosphonates or vitamin D receptor agonists (VDRAs) within the first-year post-transplant [97]. There were 134 reported fractures in these studies. The use of bisphosphonates or VDRAs was associated with a reduced number of subjects with any fracture and vertebral fractures specifically. The discordance in findings between the Cochrane review and the meta-analysis by Stein et al. may be attributable to discrepancies in the methodology of the analyses, differences between the study populations, the timing of initiation of therapy and most importantly that not all studies included reported on fractures. Specifically in the Palmer et al. review, only 11/24 studies (34/514 patients) had reported fracture data [33], while in the Stein et al. review, 134 incident fractures were reported in the 659 subjects who had follow-up information [97]. These data suggest that preventive therapy including bisphosphonates or VDRAs is likely better than no therapy or calcium alone in preventing post-transplant bone loss and perhaps even fractures. In summary, bisphosphonates may have a role in select patient populations: specifically those at risk for fracture but without evidence for low turnover bone disease. The appropriate duration of therapy is not known; however, a maximum of 12–18 months of therapy immediately post-transplantation may be sufficient. Prospective appropriately controlled studies are required to determine the optimal role of bisphosphonate therapy.
Vitamin D Receptor Agonists
VDRAs can preserve and improve BMD by several mechanisms. VDRAs promote differentiation of osteoblast precursors into mature cells, significantly suppress osteoclastic bone activity, and maintain trabecular bone volume and wall thickness [98, 99]. In addition VDRAs can reverse glucocorticoid-induced decreases in intestinal calcium absorption and mitigate against secondary hyperparathyroidism [99].
Some studies demonstrate that oral calcitriol lowers PTH in patients with persistent post-transplant hyperparathyroidism [100, 101]. Several placebo controlled clinical trials demonstrate that active vitamin D preserves and improves BMD at both the lumbar spine and femoral neck in renal allograft recipients [45–49]. None of these studies show that VDRAs have any deleterious effects on renal allograft function. Clinically important outcomes such as mortality, hospitalizations, or fractures were not evaluated in these studies. Although, bisphosphonates have been shown to be superior to VDRAs in preserving and improving BMD in clinical trials [33, 102, 103], there is emerging evidence that suggests the use of either bisphosphonates or VDRAs in solid organ transplant recipients prevents fractures in the first post-transplant year [97]. Table 24.2 summarizes the indications, risks, and contraindications of VDRA therapy.
Calcimimetics
The calcimimetic cinacalcet functions as an allosteric activator of the calcium-sensing receptor (CaSR), the molecular mechanism that mediates calcium-regulated PTH secretion by parathyroid glands. It has been shown to be effective in the treatment of persistent post-transplant hyperparathyroidism and hypercalcemia [104–109]. However, data regarding the influence of cinacalcet on BMD in renal allograft recipients are scarce. Small observational studies [110] and case reports [111] demonstrate that cinacalcet therapy for post-transplant hyperparathyroidism is associated with improved BMD, a finding that needs to be further evaluated in a well-designed prospective study. Table 24.2 summarizes the indications, risks, and contraindications of cinacalcet therapy.
Teriparatide
Teriparatide is a recombinant human PTH (1-34) that has anabolic bone effects in both glucocorticoid-induced [112] and postmenopausal osteoporosis [113]. Its use has been shown to reduce fracture rates in postmenopausal females. Teriparatide, however, did not improve BMD in a small randomized controlled trial in renal transplant recipients [114].
Denosumab and Odanacatib
Denosumab is a fully humanized monoclonal antibody that blocks the receptor activator of nuclear factor kappa-B (RANK) and subsequently inhibits the formation, function, and survival of osteoclasts. Odanacatib (ODN) is a selective, potent, and reversible inhibitor of cathepsin K (CatK). Both of these agents have been shown to improve BMD in postmenopausal women in clinical trials [115–117], and denosumab is currently FDA approved for postmenopausal osteoporosis. Clinical studies evaluating the potential effects of these agents in the prevention of post-transplant BMD loss and fracture reduction have not been performed.
Parathyroidectomy
The surgical correction of hyperparathyroidism remains indicated for persistent post-transplant hyperparathyroidism that cannot be controlled by medical management with VDRAs or cinacalcet. Approximately 0.5–5 % of kidney transplant recipients ultimately require surgical parathyroidectomy (PTX) [118, 119]. There is a paucity of data available on the effects of PTX on bone histology, BMD, and fracture risk reduction.
General indications for PTX for post-transplant hyperparathyroidism are listed in Table 24.2.
Summary of Take-Home Points
-
Post-transplant osteopenia and osteoporosis are very common in kidney transplant recipients.
-
Kidney allograft recipients are at an increased risk of fractures.
-
Post-transplant BMDs do not predict fracture risk.
-
The pathogenesis of post-transplant bone disease is multifactorial resulting from the interactions of complex abnormalities in pre-transplant CKD-MBD and renal osteodystrophy, the degree of kidney dysfunction post-transplant, peri-transplant disturbances in vitamin D-FGF-23-PTH access, and the deleterious skeletal effects of immunosuppressive therapy.
-
Bisphosphonate therapy in selected patients with high bone turnover and preserved eGFR (>35 mL/min/1.73 m2) is associated with improved BMDs and may reduce post-transplant fractures when given during the first-year post-transplantation.
-
VDRA therapy may be useful to preserve BMD as well as to treat patients with hyperparathyroidism without hypercalcemia.
-
Steroid-sparing and steroid withdrawal immunosuppressive protocols have been shown to improve BMD; however, any beneficial effect on post-transplant fractures is unproven.
-
Cinacalcet is the drug of choice for persistent post-transplant hyperparathyroidism with hypercalcemia.
-
PTX is indicated for patients who fail medical therapy and develop complications of hyperparathyroidism.
References
Brandenburg VM, Politt D, Ketteler M, Fassbender WJ, Heussen N, Westenfeld R, et al. Early rapid loss followed by long-term consolidation characterizes the development of lumbar bone mineral density after kidney transplantation. Transplantation. 2004;77(10):1566–71. Epub 2004/07/09.
Brandenburg VM, Westenfeld R, Ketteler M. The fate of bone after renal transplantation. J Nephrol. 2004;17(2):190–204. Epub 2004/08/06.
Stein E, Ebeling P, Shane E. Post-transplantation osteoporosis. Endocrinol Metab Clin North Am. 2007;36(4):937–63, viii. Epub 2007/11/07.
Vautour LM, Melton 3rd LJ, Clarke BL, Achenbach SJ, Oberg AL, McCarthy JT. Long-term fracture risk following renal transplantation: a population-based study. Osteoporos Int. 2004;15(2):160–7. Epub 2003/12/11.
Nisbeth U, Lindh E, Ljunghall S, Backman U, Fellstrom B. Fracture frequency after kidney transplantation. Transplant Proc. 1994;26(3):1764. Epub 1994/06/01.
Paydas S, Balal M, Demir E, Sertdemir Y, Erken U. Avascular osteonecrosis and accompanying anemia, leucocytosis, and decreased bone mineral density in renal transplant recipients. Transplant Proc. 2011;43(3):863–6. Epub 2011/04/14.
Abbott KC, Oglesby RJ, Hypolite IO, Kirk AD, Ko CW, Welch PG, et al. Hospitalizations for fractures after renal transplantation in the United States. Ann Epidemiol. 2001;11(7):450–7. Epub 2001/09/15.
Julian BA, Laskow DA, Dubovsky J, Dubovsky EV, Curtis JJ, Quarles LD. Rapid loss of vertebral mineral density after renal transplantation. N Engl J Med. 1991;325(8):544–50. Epub 1991/08/22.
Horber FF, Casez JP, Steiger U, Czerniak A, Montandon A, Jaeger P. Changes in bone mass early after kidney transplantation. J Bone Miner Res. 1994;9(1):1–9. Epub 1994/01/01.
Mikuls TR, Julian BA, Bartolucci A, Saag KG. Bone mineral density changes within six months of renal transplantation. Transplantation. 2003;75(1):49–54. Epub 2003/01/25.
Almond MK, Kwan JT, Evans K, Cunningham J. Loss of regional bone mineral density in the first 12 months following renal transplantation. Nephron. 1994;66(1):52–7. Epub 1994/01/01.
Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl. 2009;(113):S1–130. Epub 2009/08/01.
Ramsey-Goldman R, Dunn JE, Dunlop DD, Stuart FP, Abecassis MM, Kaufman DB, et al. Increased risk of fracture in patients receiving solid organ transplants. J Bone Miner Res. 1999;14(3):456–63. Epub 1999/02/23.
Van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C. Use of oral corticosteroids and risk of fractures. J Bone Miner Res. 2000;15(6):993–1000. Epub 2000/06/07.
Unal A, Kocyigit I, Sipahioglu MH, Tokgoz B, Kavuncuoglu F, Oymak O, et al. Loss of bone mineral density in renal transplantation recipients. Transplant Proc. 2010;42(9):3550–3. Epub 2010/11/26.
Ozel L, Ata P, Ozel MS, Toros AB, Kara M, Unal E, et al. Risk factors for osteoporosis after renal transplantation and effect of vitamin D receptor Bsm I polymorphism. Transplant Proc. 2011;43(3):858–62. Epub 2011/04/14.
Nikkel LE, Hollenbeak CS, Fox EJ, Uemura T, Ghahramani N. Risk of fractures after renal transplantation in the United States. Transplantation. 2009;87(12):1846–51. Epub 2009/06/23.
Kanaan N, Claes K, Devogelaer JP, Vanderschueren D, Depresseux G, Goffin E, et al. Fibroblast growth factor-23 and parathyroid hormone are associated with post-transplant bone mineral density loss. Clin J Am Soc Nephrol. 2010;5(10):1887–92. Epub 2010/07/17.
Dolgos S, Hartmann A, Bonsnes S, Isaksen GA, Godang K, Ueland T, et al. Early changes in bone mass, biochemical bone markers and fibroblast growth factor 23 after renal transplantation. Scand J Clin Lab Invest. 2009;69(2):161–7. Epub 2008/10/31.
Braga Junior JW, Neves RM, Pinheiro MM, Frisoli Junior A, Castro CH, Szejnfeld VL, et al. Prevalence of low trauma fractures in long-term kidney transplant patients with preserved renal function. Braz J Med Biol Res. 2006;39(1):137–47. Epub 2006/01/10.
Marcen R, Caballero C, Uriol O, Fernandez A, Villafruela JJ, Pascual J, et al. Prevalence of osteoporosis, osteopenia, and vertebral fractures in long-term renal transplant recipients. Transplant Proc. 2007;39(7):2256–8. Epub 2007/09/25.
Opelz G, Dohler B. Association of mismatches for HLA-DR with incidence of posttransplant hip fracture in kidney transplant recipients. Transplantation. 2011;91(1):65–9. Epub 2011/04/01.
Mainra R, Elder GJ. Individualized therapy to prevent bone mineral density loss after kidney and kidney-pancreas transplantation. Clin J Am Soc Nephrol. 2010;5(1):117–24. Epub 2009/12/08.
Rojas E, Carlini RG, Clesca P, Arminio A, Suniaga O, De Elguezabal K, et al. The pathogenesis of osteodystrophy after renal transplantation as detected by early alterations in bone remodeling. Kidney Int. 2003;63(5):1915–23. Epub 2003/04/05.
Lehmann G, Ott U, Stein G, Steiner T, Wolf G. Renal osteodystrophy after successful renal transplantation: a histomorphometric analysis in 57 patients. Transplant Proc. 2007;39(10):3153–8. Epub 2007/12/20.
Sprague SM, Belozeroff V, Danese MD, Martin LP, Olgaard K. Abnormal bone and mineral metabolism in kidney transplant patients—a review. Am J Nephrol. 2008;28(2):246–53. Epub 2007/11/09.
Maalouf NM, Shane E. Osteoporosis after solid organ transplantation. J Clin Endocrinol Metab. 2005;90(4):2456–65. Epub 2004/12/30.
Adachi JD, Bensen WG, Hodsman AB. Corticosteroid-induced osteoporosis. Semin Arthritis Rheum. 1993;22(6):375–84. Epub 1993/06/01.
Weinstein RS, Jilka RL, Parfitt AM, Manolagas SC. Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids. Potential mechanisms of their deleterious effects on bone. J Clin Investig. 1998;102(2):274–82. Epub 1998/07/17.
Schlosberg M, Movsowitz C, Epstein S, Ismail F, Fallon MD, Thomas S. The effect of cyclosporin A administration and its withdrawal on bone mineral metabolism in the rat. Endocrinology. 1989;124(5):2179–84. Epub 1989/05/01.
Moreira RO, Thiago LS, Oliveira FL, Balduino A, Borojevic R, Duarte ME, et al. Cyclosporine A, but not tacrolimus, is associated with impaired proliferation and differentiation of human osteoblast-like cells in vitro. Med Sci Monit. 2009;15(3):BR65–70. Epub 2009/02/28.
Stewart PJ, Stern PH. Cyclosporines: correlation of immunosuppressive activity and inhibition of bone resorption. Calcif Tissue Int. 1989;45(4):222–6. Epub 1989/10/01.
Palmer SC, McGregor DO, Strippoli GF. Interventions for preventing bone disease in kidney transplant recipients. Cochrane Database Syst Rev. 2007;(3):CD005015. Epub 2007/07/20.
Grotz W, Nagel C, Poeschel D, Cybulla M, Petersen KG, Uhl M, et al. Effect of ibandronate on bone loss and renal function after kidney transplantation. J Am Soc Nephrol. 2001;12(7):1530–7. Epub 2001/06/26.
Mitterbauer C, Schwarz C, Haas M, Oberbauer R. Effects of bisphosphonates on bone loss in the first year after renal transplantation—a meta-analysis of randomized controlled trials. Nephrol Dial Transplant. 2006;21(8):2275–81. Epub 2006/04/01.
Fan SL, Almond MK, Ball E, Evans K, Cunningham J. Pamidronate therapy as prevention of bone loss following renal transplantation. Kidney Int. 2000;57(2):684–90. Epub 2000/01/29.
Jeffery JR, Leslie WD, Karpinski ME, Nickerson PW, Rush DN. Prevalence and treatment of decreased bone density in renal transplant recipients: a randomized prospective trial of calcitriol versus alendronate. Transplantation. 2003;76(10):1498–502. Epub 2003/12/06.
El-Agroudy AE, El-Husseini AA, El-Sayed M, Ghoneim MA. Preventing bone loss in renal transplant recipients with vitamin D. J Am Soc Nephrol. 2003;14(11):2975–9. Epub 2003/10/22.
Nowacka-Cieciura E, Cieciura T, Baczkowska T, Kozinska-Przybyl O, Tronina O, Chudzinski W, et al. Bisphosphonates are effective prophylactic of early bone loss after renal transplantation. Transplant Proc. 2006;38(1):165–7. Epub 2006/03/01.
Coco M, Glicklich D, Faugere MC, Burris L, Bognar I, Durkin P, et al. Prevention of bone loss in renal transplant recipients: a prospective, randomized trial of intravenous pamidronate. J Am Soc Nephrol. 2003;14(10):2669–76. Epub 2003/09/30.
Haas M, Leko-Mohr Z, Roschger P, Kletzmayr J, Schwarz C, Mitterbauer C, et al. Zoledronic acid to prevent bone loss in the first 6 months after renal transplantation. Kidney Int. 2003;63(3):1130–6. Epub 2003/03/13.
Abediazar S, Nakhjavani MR. Effect of alendronate on early bone loss of renal transplant recipients. Transplant Proc. 2011;43(2):565–7. Epub 2011/03/29.
Torregrosa JV, Fuster D, Pedroso S, Diekmann F, Campistol JM, Rubi S, et al. Weekly risedronate in kidney transplant patients with osteopenia. Transpl Int. 2007;20(8):708–11. Epub 2007/06/09.
Torregrosa JV, Fuster D, Monegal A, Gentil MA, Bravo J, Guirado L, et al. Efficacy of low doses of pamidronate in osteopenic patients administered in the early post-renal transplant. Osteoporos Int. 2011;22(1):281–7. Epub 2010/03/17.
Josephson MA, Schumm LP, Chiu MY, Marshall C, Thistlethwaite JR, Sprague SM. Calcium and calcitriol prophylaxis attenuates posttransplant bone loss. Transplantation. 2004;78(8):1233–6. Epub 2004/10/27.
El-Agroudy AE, El-Husseini AA, El-Sayed M, Mohsen T, Ghoneim MA. A prospective randomized study for prevention of postrenal transplantation bone loss. Kidney Int. 2005;67(5):2039–45. Epub 2005/04/21.
El-Husseini AA, El-Agroudy AE, El-Sayed M, Sobh MA, Ghoneim MA. A prospective randomized study for the treatment of bone loss with vitamin d during kidney transplantation in children and adolescents. Am J Transplant. 2004;4(12):2052–7. Epub 2004/12/04.
Torres A, Garcia S, Gomez A, Gonzalez A, Barrios Y, Concepcion MT, et al. Treatment with intermittent calcitriol and calcium reduces bone loss after renal transplantation. Kidney Int. 2004;65(2):705–12. Epub 2004/01/14.
De Sevaux RG, Hoitsma AJ, Corstens FH, Wetzels JF. Treatment with vitamin D and calcium reduces bone loss after renal transplantation: a randomized study. J Am Soc Nephrol. 2002;13(6):1608–14. Epub 2002/06/01.
Kodras K, Haas M. Effect of kidney transplantation on bone. Eur J Clin Invest. 2006;36 Suppl 2:63–75. Epub 2006/08/04.
Wissing KM, Broeders N, Moreno-Reyes R, Gervy C, Stallenberg B, Abramowicz D. A controlled study of vitamin D3 to prevent bone loss in renal-transplant patients receiving low doses of steroids. Transplantation. 2005;79(1):108–15. Epub 2005/02/17.
Cruz DN, Wysolmerski JJ, Brickel HM, Gundberg CG, Simpson CA, Mitnick MA, et al. Parameters of high bone-turnover predict bone loss in renal transplant patients: a longitudinal study. Transplantation. 2001;72(1):83–8. Epub 2001/07/27.
Pichette V, Bonnardeaux A, Prudhomme L, Gagne M, Cardinal J, Ouimet D. Long-term bone loss in kidney transplant recipients: a cross-sectional and longitudinal study. Am J Kidney Dis. 1996;28(1):105–14. Epub 1996/07/01.
Ball AM, Gillen DL, Sherrard D, Weiss NS, Emerson SS, Seliger SL, et al. Risk of hip fracture among dialysis and renal transplant recipients. JAMA. 2002;288(23):3014–8. Epub 2002/12/17.
Conley E, Muth B, Samaniego M, Lotfi M, Voss B, Armbrust M, et al. Bisphosphonates and bone fractures in long-term kidney transplant recipients. Transplantation. 2008;86(2):231–7. Epub 2008/07/23.
Kidney Disease: Improving Global Outcomes Transplant Work Group. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplant. 2009;9 Suppl 3:S1–155. Epub 2009/10/23.
Tang S, Chan TM, Lui SL, Li FK, Lo WK, Lai KN. Risk factors for avascular bone necrosis after renal transplantation. Transplant Proc. 2000;32(7):1873–5. Epub 2000/12/20.
Celik A, Tekis D, Saglam F, Tunali S, Kabakci N, Ozaksoy D, et al. Association of corticosteroids and factor V, prothrombin, and MTHFR gene mutations with avascular osteonecrosis in renal allograft recipients. Transplant Proc. 2006;38(2):512–6. Epub 2006/03/22.
Hedri H, Cherif M, Zouaghi K, Abderrahim E, Goucha R, Ben Hamida F, et al. Avascular osteonecrosis after renal transplantation. Transplant Proc. 2007;39(4):1036–8. Epub 2007/05/26.
Lausten GS, Lemser T, Jensen PK, Egfjord M. Necrosis of the femoral head after kidney transplantation. Clin Transplant. 1998;12(6):572–4. Epub 1998/12/16.
Marston SB, Gillingham K, Bailey RF, Cheng EY. Osteonecrosis of the femoral head after solid organ transplantation: a prospective study. J Bone Joint Surg Am. 2002;84-A(12):2145–51. Epub 2002/12/11.
Fernandez-Fresnedo G, Rodrigo E, Ruiz JC, Martin de Francisco AL, Arias M. Bone metabolism according to chronic kidney disease stages in patients undergoing kidney transplantation: a 5-year database analysis. Transplant Proc. 2009;41(6):2403–5. Epub 2009/09/01.
Ambrus C, Molnar MZ, Czira ME, Rosivall L, Kiss I, Remport A, et al. Calcium, phosphate and parathyroid metabolism in kidney transplanted patients. Int Urol Nephrol. 2009;41(4):1029–38. Epub 2009/08/25.
Kasiske BL, Zeier MG, Chapman JR, Craig JC, Ekberg H, Garvey CA, et al. KDIGO clinical practice guideline for the care of kidney transplant recipients: a summary. Kidney Int. 2010;77(4):299–311. Epub 2009/10/23.
Cruz EA, Lugon JR, Jorgetti V, Draibe SA, Carvalho AB. Histologic evolution of bone disease 6 months after successful kidney transplantation. Am J Kidney Dis. 2004;44(4):747–56. Epub 2004/09/24.
Bates DW, Black DM, Cummings SR. Clinical use of bone densitometry: clinical applications. JAMA. 2002;288(15):1898–900. Epub 2002/10/17.
Grotz WH, Mundinger FA, Gugel B, Exner V, Kirste G, Schollmeyer PJ. Bone fracture and osteodensitometry with dual energy X-ray absorptiometry in kidney transplant recipients. Transplantation. 1994;58(8):912–5. Epub 1994/10/27.
Kanis JA. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: synopsis of a WHO report. WHO Study Group. Osteoporos Int. 1994;4(6):368–81. Epub 1994/11/01.
Huang M, Sprague SM. Bone disease in kidney transplant patients. Semin Nephrol. 2009;29(2):166–73. Epub 2009/04/18.
Sprague SM, Josephson MA. Bone disease after kidney transplantation. Semin Nephrol. 2004;24(1):82–90. Epub 2004/01/20.
Genant HK, Engelke K, Prevrhal S. Advanced CT bone imaging in osteoporosis. Rheumatology (Oxford). 2008;47 Suppl 4:iv9–16. Epub 2008/07/02.
Liu XS, Stein EM, Zhou B, Zhang CA, Nickolas TL, Cohen A, et al. Individual trabecula segmentation (ITS)-based morphological analyses and micro finite element analysis of HR-pQCT images discriminate postmenopausal fragility fractures independent of DXA measurements. J Bone Miner Res. 2012;27:263–72. Epub 2011/11/11.
Nickolas TL, Cremers S, Zhang A, Thomas V, Stein E, Cohen A, et al. Discriminants of prevalent fractures in chronic kidney disease. J Am Soc Nephrol. 2011;22(8):1560–72. Epub 2011/07/26.
Nickolas TL, Stein E, Cohen A, Thomas V, Staron RB, McMahon DJ, et al. Bone mass and microarchitecture in CKD patients with fracture. J Am Soc Nephrol. 2010;21(8):1371–80. Epub 2010/04/17.
Sugano N, Kubo T, Takaoka K, Ohzono K, Hotokebuchi T, Matsumoto T, et al. Diagnostic criteria for non-traumatic osteonecrosis of the femoral head. A multicentre study. J Bone Joint Surg Br. 1999;81(4):590–5. Epub 1999/08/27.
Wehrli FW, Saha PK, Gomberg BR, Song HK, Snyder PJ, Benito M, et al. Role of magnetic resonance for assessing structure and function of trabecular bone. Top Magn Reson Imaging. 2002;13(5):335–55. Epub 2002/12/05.
Wehrli FW, Leonard MB, Saha PK, Gomberg BR. Quantitative high-resolution magnetic resonance imaging reveals structural implications of renal osteodystrophy on trabecular and cortical bone. J Magn Reson Imaging. 2004;20(1):83–9. Epub 2004/06/29.
Link TM, Saborowski, Kisters K, Kempkes M, Kosch M, Newitt D, et al. Changes in calcaneal trabecular bone structure assessed with high-resolution MR imaging in patients with kidney transplantation. Osteoporos Int. 2002;13(2):119–29. Epub 2002/03/22.
Rajapakse CS, Leonard MB, Bhagat YA, Sun W, Magland JF, Wehrli FW. Micro-MR imaging-based computational biomechanics demonstrates reduction in cortical and trabecular bone strength after renal transplantation. Radiology. 2012;262(3):912–20. Epub 2012/02/24.
Sperschneider H, Stein G. Bone disease after renal transplantation. Nephrol Dial Transplant. 2003;18(5):874–7. Epub 2003/04/11.
Sahin G, Yasar NS, Sirmagul B, Bal C, Yalcin AU. The effect of low-dose cholecalciferol and calcium treatment on posttransplant bone loss in renal transplant patients: a prospective study. Ren Fail. 2008;30(10):992–9. Epub 2008/11/19.
Isoniemi H, Appelberg J, Nilsson CG, Makela P, Risteli J, Hockerstedt K. Transdermal oestrogen therapy protects postmenopausal liver transplant women from osteoporosis. A 2-year follow-up study. J Hepatol. 2001;34(2):299–305.
Evenepoel P, Lerut E, Naesens M, Bammens B, Claes K, Kuypers D, et al. Localization, etiology and impact of calcium phosphate deposits in renal allografts. Am J Transplant. 2009;9(11):2470–8. Epub 2009/08/18.
Caravaca F, Fernandez MA, Ruiz-Calero R, Cubero J, Aparicio A, Jimenez F, et al. Effects of oral phosphorus supplementation on mineral metabolism of renal transplant recipients. Nephrol Dial Transplant. 1998;13(10):2605–11. Epub 1998/10/30.
Kulak CA, Borba VZ, Kulak Junior J, Campos DJ, Shane E. Post-transplantation osteoporosis. Arq Bras Endocrinol Metabol. 2010;54(2):143–9. Epub 2010/05/21.
Weisinger JR, Carlini RG, Rojas E, Bellorin-Font E. Bone disease after renal transplantation. Clin J Am Soc Nephrol. 2006;1(6):1300–13. Epub 2007/08/21.
Pereira S, Pedroso S, Martins L, Santos P, Almeida M, Freitas C, et al. Bone mineral density after simultaneous kidney-pancreas transplantation: four years follow-up of 57 recipients. Transplant Proc. 2010;42(2):555–7. Epub 2010/03/23.
Nikkel LE, Mohan S, Zhang A, McMahon DJ, Boutroy S, Dube G, et al. Reduced fracture risk with early corticosteroid withdrawal after kidney transplant. Am J Transplant. 2012;12(3):649–59. Epub 2011/12/14.
Hazzan M, Glowacki F, Lionet A, Provot F, Noel C. [Weaning from corticosteroid therapy after kidney transplantation]. Nephrol Ther. 2009;5 Suppl 6:S355–8. Epub 2010/02/05. Sevrage de la corticotherapie apres transplantation renale.
Ing SW, Sinnott LT, Donepudi S, Davies EA, Pelletier RP, Lane NE. Change in bone mineral density at one year following glucocorticoid withdrawal in kidney transplant recipients. Clin Transplant. 2011;25(2):E113–23. Epub 2010/10/22.
Edwards BJ, Desai A, Tsai J, Du H, Edwards GR, Bunta AD, et al. Elevated incidence of fractures in solid-organ transplant recipients on glucocorticoid-sparing immunosuppressive regimens. J Osteoporos. 2011;2011:591793. Epub 2011/09/17.
Edwards BJ, Desai A, Tsai J, Du H, Edwards GR, Bunta AD, et al. Elevated incidence of fractures in solid-organ transplant recipients on glucocorticoid-sparing immunosuppressive regimens. J Osteoporos. 2011;2011:591793. Epub 2011/09/17.
Sato M, Grasser W, Endo N, Akins R, Simmons H, Thompson DD, et al. Bisphosphonate action. Alendronate localization in rat bone and effects on osteoclast ultrastructure. J Clin Invest. 1991;88(6):2095–105. Epub 1991/12/01.
Colucci S, Minielli V, Zambonin G, Cirulli N, Mori G, Serra M, et al. Alendronate reduces adhesion of human osteoclast-like cells to bone and bone protein-coated surfaces. Calcif Tissue Int. 1998;63(3):230–5. Epub 1998/08/14.
Plotkin LI, Lezcano V, Thostenson J, Weinstein RS, Manolagas SC, Bellido T. Connexin 43 is required for the anti-apoptotic effect of bisphosphonates on osteocytes and osteoblasts in vivo. J Bone Miner Res. 2008;23(11):1712–21. Epub 2008/07/04.
Coco M, Pullman J, Cohen HW, Lee S, Shapiro C, Solorzano C, et al. Effect of risedronate on bone in renal transplant recipients. J Am Soc Nephrol. 2012;23(8):1426–37. Epub 2012/07/17.
Stein EM, Ortiz D, Jin Z, McMahon DJ, Shane E. Prevention of fractures after solid organ transplantation: a meta-analysis. J Clin Endocrinol Metabol. 2011;96(11):3457–65. Epub 2011/08/19.
Cueto-Manzano AM, Konel S, Freemont AJ, Adams JE, Mawer B, Gokal R, et al. Effect of 1,25-dihydroxyvitamin D3 and calcium carbonate on bone loss associated with long-term renal transplantation. Am J Kidney Dis. 2000;35(2):227–36. Epub 2000/02/17.
Sambrook P. Alfacalcidol and calcitriol in the prevention of bone loss after organ transplantation. Calcif Tissue Int. 1999;65(4):341–3. Epub 1999/09/15.
Steiner RW, Ziegler M, Halasz NA, Catherwood BD, Manolagas S, Deftos LJ. Effect of daily oral vitamin D and calcium therapy, hypophosphatemia, and endogenous 1-25 dihydroxycholecalciferol on parathyroid hormone and phosphate wasting in renal transplant recipients. Transplantation. 1993;56(4):843–6. Epub 1993/10/01.
Lobo PI, Cortez MS, Stevenson W, Pruett TL. Normocalcemic hyperparathyroidism associated with relatively low 1:25 vitamin D levels post-renal transplant can be successfully treated with oral calcitriol. Clin Transplant. 1995;9(4):277–81. Epub 1995/08/01.
Koc M, Tuglular S, Arikan H, Ozener C, Akoglu E. Alendronate increases bone mineral density in long-term renal transplant recipients. Transplant Proc. 2002;34(6):2111–3. Epub 2002/09/25.
Nam JH, Moon JI, Chung SS, Kim SI, Park KI, Song YD, et al. Pamidronate and calcitriol trial for the prevention of early bone loss after renal transplantation. Transplant Proc. 2000;32(7):1876. Epub 2000/12/20.
Serra AL, Wuhrmann C, Wuthrich RP. Phosphatemic effect of cinacalcet in kidney transplant recipients with persistent hyperparathyroidism. Am J Kidney Dis. 2008;52(6):1151–7. Epub 2008/10/28.
Serra AL, Schwarz AA, Wick FH, Marti HP, Wuthrich RP. Successful treatment of hypercalcemia with cinacalcet in renal transplant recipients with persistent hyperparathyroidism. Nephrol Dial Transplant. 2005;20(7):1315–9. Epub 2005/06/09.
Copley JB, Germain M, Stern L, Pankewycz O, Katznelson S, Shah T, et al. Evaluation of cinacalcet HCl treatment after kidney transplantation. Transplant Proc. 2010;42(7):2503–8. Epub 2010/09/14.
Gomez Marques G, Obrador Mulet A, Vilar Gimeno A, Pascual Felip MJ, Alarcon Zurita A, Molina Guasch M, et al. Treatment with cinacalcet of secondary hyperparathyroidism after renal transplantation. Transplant Proc. 2009;41(6):2139–43. Epub 2009/09/01.
Serra AL, Savoca R, Huber AR, Hepp U, Delsignore A, Hersberger M, et al. Effective control of persistent hyperparathyroidism with cinacalcet in renal allograft recipients. Nephrol Dial Transplant. 2007;22(2):577–83. Epub 2006/09/29.
El-Amm JM, Doshi MD, Singh A, Migdal S, Morawski K, Sternbauer D, et al. Preliminary experience with cinacalcet use in persistent secondary hyperparathyroidism after kidney transplantation. Transplantation. 2007;83(5):546–9. Epub 2007/03/14.
Bergua C, Torregrosa JV, Fuster D, Gutierrez-Dalmau A, Oppenheimer F, Campistol JM. Effect of cinacalcet on hypercalcemia and bone mineral density in renal transplanted patients with secondary hyperparathyroidism. Transplantation. 2008;86(3):413–7. Epub 2008/08/14.
Decleire PY, Devogelaer JP, Goffin E. Cinacalcet improves bone mineral density in a renal transplant recipient with persistent hyperparathyroidism. Clin Nephrol. 2008;69(3):231–2. Epub 2008/04/10.
Saag KG, Shane E, Boonen S, Marin F, Donley DW, Taylor KA, et al. Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N Engl J Med. 2007;357(20):2028–39. Epub 2007/11/16.
Finkelstein JS, Wyland JJ, Lee H, Neer RM. Effects of teriparatide, alendronate, or both in women with postmenopausal osteoporosis. J Clin Endocrinol Metab. 2010;95(4):1838–45. Epub 2010/02/19.
Cejka D, Benesch T, Krestan C, Roschger P, Klaushofer K, Pietschmann P, et al. Effect of teriparatide on early bone loss after kidney transplantation. Am J Transplant. 2008;8(9):1864–70. Epub 2008/09/13.
Jamal SA, Ljunggren O, Stehman-Breen C, Cummings SR, McClung MR, Goemaere S, et al. Effects of denosumab on fracture and bone mineral density by level of kidney function. J Bone Miner Res. 2011;26(8):1829–35. Epub 2011/04/15.
Papapoulos S, Chapurlat R, Libanati C, Brandi M, Brown J, Czerwinski E, et al. Five years of denosumab exposure in women with postmenopausal osteoporosis: results from the first two years of the FREEDOM extension. J Bone Miner Res. 2012;27(3):694–701. Epub 2011/11/25.
Bone HG, McClung MR, Roux C, Recker RR, Eisman JA, Verbruggen N, et al. Odanacatib, a cathepsin-K inhibitor for osteoporosis: a two-year study in postmenopausal women with low bone density. J Bone Miner Res. 2010;25(5):937–47. Epub 2009/10/31.
Evenepoel P, Claes K, Kuypers D, Maes B, Vanrenterghem Y. Impact of parathyroidectomy on renal graft function, blood pressure and serum lipids in kidney transplant recipients: a single centre study. Nephrol Dial Transplant. 2005;20(8):1714–20. Epub 2005/05/28.
Drakopoulos S, Koukoulaki M, Apostolou T, Pistolas D, Balaska K, Gavriil S, et al. Total parathyroidectomy without autotransplantation in dialysis patients and renal transplant recipients, long-term follow-up evaluation. Am J Surg. 2009;198(2):178–83. Epub 2009/02/17.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Alshayeb, H.M., Sprague, S.M., Josephson, M.A. (2014). Management of Transplantation Renal Bone Disease: Interplay of Bone Mineral Density and Decisions Regarding Bisphosphonate Use. In: Weir, M., Lerma, E. (eds) Kidney Transplantation. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-0342-9_24
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0342-9_24
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-0341-2
Online ISBN: 978-1-4939-0342-9
eBook Packages: MedicineMedicine (R0)