Abstract
Although poliomyelitis has been eradicated from much of the globe, during the past 15 years countries in the Asia-Pacific region have been afflicted by large outbreaks of infection with the related human enterovirus A71 (EV71). This infection affects mostly children, causing hand-foot-and-mouth disease, aseptic meningitis, poliomyelitis-like acute flaccid paralysis, brainstem encephalitis, and other severe systemic manifestations, particularly pulmonary edema and cardiorespiratory collapse. Being an RNA virus, EV71 lacks a proofreading mechanism and is evolving rapidly, with new outbreaks occurring across Asia in regular cycles. The subgenogroups appear to differ in their clinical epidemiological properties, with some of the more recently evolved subgenogroups having potential to cause explosive outbreaks. There is controversy over the pathogenesis of the severe cardiopulmonary manifestations and the relative contributions of neurogenic pulmonary edema, cardiac dysfunction, increased vascular permeability, and “cytokine storm.” Clinical predictors of the development of severe disease include high temperature and lethargy, and a lumbar puncture may reveal pleocytosis. Of the many alternative biological samples and laboratory tests with diagnostic utility, PCR applied to swabs from the throat or vesicle fluid, if available, is one of the most efficient. Magnetic resonance imaging reveals features of inflammation, particularly in the anterior horns of the spinal cord, the dorsal pons, and the medulla. There is no established antiviral treatment. Although untested in formal trials, intravenous immunoglobulin appears to be beneficial in severe disease, perhaps through nonspecific anti-inflammatory mechanisms. Milrinone appears helpful in treatment of cardiac dysfunction. To control outbreaks, social distancing (e.g., closing nurseries) has been attempted as a public health measure, but its effectiveness has not been fully examined. An inactivated whole virus vaccine has been found to be safe and effective in a phase 3 randomized double-blind placebo-controlled trial.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Pulmonary Edema
- Central Nervous System Involvement
- Aseptic Meningitis
- Central Nervous System Infection
- EV71 Infection
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction and Historical Background
The global polio eradication campaign has eliminated this devastating enteroviral disease from Europe, the Americas, and most of Africa and Asia. However, in the past 15 years, a related virus, human enterovirus 71 (EV71), has emerged across Asia, where it threatens to become the “new polio” [120]. EV71 first appeared in California in the 1960s and subsequently caused sporadic cases or small outbreaks of hand-foot-and-mouth disease (HFMD) [120], neurological disease, or both. In 1997, the virus caused an unexpectedly large and severe outbreak with many fatalities in Sarawak, Malaysia. Since then, countries of the Asia-Pacific region have been hit by regular epidemics of EV71, including an epidemic in Taiwan in 1998, in which millions of people were thought to have been infected, and an outbreak of HFMD in China in 2009, during which nearly 500,000 cases were reported [13, 16, 33, 55, 56, 76, 98, 112, 146, 171, 177]. Although the virus circulates worldwide, the largest outbreaks of disease have so far been largely confined to the Asia-Pacific region, for reasons that are incompletely understood [12, 13, 16, 17, 33, 56, 70, 76, 98, 136, 146, 176, 178]. The neurological manifestations range from aseptic meningitis to acute flaccid paralysis and brainstem encephalitis. They are often accompanied by systemic complications such as severe pulmonary edema and shock, which are thought to be neurogenic in origin [22, 63].
2 Descriptive Epidemiology
2.1 Initial Epidemiological Observations
Although EV71 was first isolated from the stool of a 9-month-old infant with encephalitis in California in 1969, a retrospective analysis of samples from the Netherlands showed that EV71 was circulating as early as 1963 [128, 147]. Before long small outbreaks of neurological infection including encephalitis and aseptic meningitis attributed to the newly identified neurotrophic virus were reported in New York, Melbourne, and Sweden in the early 1970s [9, 43, 74]. The dermotropic properties of EV71 were first recognized during epidemics of HFMD in Japan in 1973 [54, 67]. In the 1970s, two large and severe EV71 epidemics occurred in Europe. The first was in Bulgaria in 1975, initially attributed to polioviruses because its epidemiological, clinical, and pathological characteristics mimicked those of poliomyelitis [40, 137]. In fact, at the height of the epidemic, nationwide administration of live-attenuated poliovirus vaccine was instituted. Isolation of EV71 later confirmed this virus as the etiological agent in 347 (77 %) of 451 children who presented with nonspecific febrile illness or neurological disease, of whom 44 died. The second major European epidemic occurred in Hungary in 1978, with 1,550 cases (826 aseptic meningitis and 724 encephalitis) and 47 deaths. Unlike the Bulgarian epidemic, the Hungarian one included a small number of patients with HFMD [103].
2.2 EV71 Activity in the Asia-Pacific Region
After the Australian and Japanese EV71 epidemics of the 1970s, further small epidemics and sporadic clusters occurred in Hong Kong (1985) and Australia (1986) [51, 126]. Then in 1997, a large outbreak in Sarawak, Malaysia, heralded the beginning of a series of large outbreaks across the Asia-Pacific region. These continuing epidemics have established EV71 as a major public health threat across the region (Table 12.1) [11, 12, 27, 39, 64, 66, 71, 79, 82, 97, 136, 146, 177, 178].
During the Sarawak outbreak, between May and July 1997, a total of 2,618 HFMD cases and 34 deaths were recorded. EV71 also caused four deaths in peninsular Malaysia and a number of cases of severe neurological disease in Japan [13, 76, 92]. Another large epidemic occurred in Taiwan in 1998 when 405 children were hospitalized for serious neurological complications and 78 died. Epidemiological studies estimated that almost 1.5 million people were infected with the virus [56]. The largest epidemic to date occurred in China in 2008; approximately 490,000 children including 126 deaths were reported nationwide. At the epicenter in Anhui Province, more than 6,000 HFMD cases and 22 deaths were reported [178]. Surveillance improved following designation of HFMD as a notifiable disease in China in May 2008, and regular massive epidemics with alarming number of deaths were recorded. There were 1.1 million cases (353 deaths), 1.8 million cases (905 deaths), 1.6 million cases (509 deaths), and 2.2 million cases (567 deaths) reported in 2009, 2010, 2011, and 2012, respectively (http://www.chinacdc.cn/tjsj/fdcrbbg/index_1.html, accessed 15/7/2013). In addition to these very large outbreaks, many countries, including Sarawak, Taiwan, Singapore, Vietnam, and Japan, have experienced cyclical epidemics at 2- to 3-year intervals [59, 101, 119]. The most recent EV71 epidemic occurred in Cambodia in 2012. The outbreak, first labeled as a mysterious illness by local media, was associated with more than 50 sudden deaths within 24 h of hospitalization in children who were 3 years of age and below [129].
The clinico-epidemiological features of the EV71 epidemics in Asia in recent years differ considerably from earlier epidemics in that in addition to HFMD, aseptic meningitis, and flaccid paralysis, brainstem encephalitis associated with cardiopulmonary dysfunction also occurs, which has been the principal cause of death in most fatal cases in Asia [13, 63, 120, 146, 178]. The affected children have typically presented with a short febrile illness accompanied by subtle neurological signs, following which they develop signs of tachycardia, poor perfusion, and tachypnea, which rapidly develops into acute intractable cardiac impairment and fulminant, often fatal, pulmonary edema or hemorrhage [111]. Diagnostic imaging and autopsy examination indicate that this is associated with encephalitis in the brainstem, especially the medulla, and neurogenic pulmonary edema is thought to be the primary pathogenic process [22, 63, 91, 92]. Such rapidly fatal HFMD was not observed in epidemics caused by EV71 in the 1970s and 1980s, where aseptic meningitis was the predominant clinical manifestation [9, 74].
2.3 EV71 Circulation Outside the Asia-Pacific Region
Outside the Asia-Pacific region, EV71 has continued to circulate at low levels of activity in America, Europe, and Africa, producing sporadic cases or small outbreaks. The outbreaks involve mainly young children who develop aseptic meningitis, hand-foot-and-mouth disease, and other complications (Table 12.2).
Clearly, EV71 has spread across the world. Why the huge epidemics have been confined to the Asia-Pacific region, and whether they might be seen elsewhere in the future is not fully understood, though recent molecular epidemiological work on of the evolution of the different genotypes of virus has offered some key insights into this important question.
3 Molecular Epidemiology
3.1 Virus Genogroup, Evolution, and Geographical Distribution
Before 1999, data about the molecular epidemiology of EV71 were sparse. However, systematic laboratory surveillance established in several Asian countries following the first epidemics in the late 1990s has provided invaluable information on the geographical distribution, spread, and evolution of the virus.
The first complete phylogenetic analysis of EV71 based on the structural VP1 gene identified three independent lineages of EV71 and designated them as genogroups A, B, and C [11]. A sequence diversity of at least 15 % in the VP1 gene was used to distinguish genogroups. Genogroup A consists of a single member, the prototype BrCr strain, which was first identified in California in 1970 and until very recently had never been reported outside the USA (see below). The genogroup B viruses, subdivided into subgenogroups B1 and B2 with divergence of 12 % at the nucleotide level, were the predominant circulating strains in the 1970s and 1980s. The genogroup C viruses, also similarly subdivided into C1 and C2, were only identified later in the mid-1980s (Fig. 12.1). A recent phylogenetic analysis suggests that EV71 may have emerged from the genetically closely related coxsackie virus (CV) A16 as recently as 1940 [144].
Phylogenetic analysis of EV71 VP1 gene sequences. A neighbor-joining tree constructed using the Kimura-2 parameter as a model for nucleotide substitution. The robustness of the tree was determined by bootstrapping using 1,000 pseudoreplicates. Sequences are labeled according to the following convention: “GenBank accession number” – “Country of origin” – “Year of isolation.” The scale bar represents nucleotide changes per site per year
A number of new subgenogroups within genogroups B and C have emerged in the Asia-Pacific region in recent years. Subgenogroup B3 and B4 viruses are thought to have co-circulated in the region since 1997 [12, 97, 136]. Subgenogroup B5 was first isolated in Sarawak and Japan in 2003 and was responsible for epidemics in Sarawak, Taiwan, and Brunei in 2006 [1, 66, 101, 119]. Except for the major community outbreak in Sydney in 1986, subgenogroup C1 viruses have mostly been isolated from sporadic cases since the mid-1980s, suggesting a low level of endemic circulation across the globe [11, 127]. Subgenogroup C2 viruses were responsible for the large EV71 epidemic in Taiwan in 1998 and the Perth outbreak in 1999 [12, 82, 96, 98]. Subgenogroup C3 was isolated in Japan in 1994 and Korea in 2000 [12, 68, 70]. Since 2000, subgenogroup C4 has been the predominant circulating genogroup in mainland China, through the most recent epidemic in 2008, and has occurred in Japan, Vietnam, and Taiwan [82, 136, 146, 178]. Subgenogroup C5 has been reported from Southern Vietnam and Taiwan [66, 146]. A genetically distinct EV71 strain (R13223, GenBank accession no. AY179600 to AY179602), with no genetic relationship to other strains recently isolated in the Asia-Pacific region, was isolated from a child with acute flaccid paralysis in India in 2001; it has been assigned as the only member of genogroup D [44]. In short, two major EV71 genogroups (B and C) have co-circulated and coevolved into subgenogroups in the Asia-Pacific region over the past 15 years. In Malaysia and Singapore, the genogroup B viruses have dominated, whereas in East Asia, particularly in mainland China and Vietnam, genogroup C viruses have dominated.
Interestingly, EV71 isolates of genogroup A were reported from 5 of 22 children presenting with HFMD in Anhui province of Central China during the 2008 outbreak that was the first ever reported detection of genogroup A viruses since the original prototype was isolated in America [174]. The nucleotide sequence of the complete VP1 gene of the isolates clusters very closely with that of the prototype genogroup A virus, with minimal divergence. This highly unexpected occurrence could indicate that the genogroup A virus has been circulating undetected in central China with very little evolutionary change for four decades or it could raise doubts about the source of the virus templates that were sequenced. Surveillance from the same outbreak by the Chinese Center for Disease Control does not appear to have identified any genogroup A viruses [178]. Hence, it is important to await confirmation by other laboratories before concluding that genogroup A viruses have reemerged. Another genetically distinct EV71 strain (NMA-03-008, GenBank accession no. JN255590) belonged to a previously unknown novel genogroup has been reported in Central African Republic; this indicates the existence of an additional African genogroup, which may have restricted geographical distribution so far [7]. Clearly, good surveillance programs are required in many different geographical regions in order to gather accurate and relevant information about EV71 transmission and evolution.
3.2 Transmission and Epidemic Potential of Genogroups
Following the initial outbreaks in Sarawak and Taiwan in the late 1990s, surveillance systems for EV71 were established in a number of countries in the Asia-Pacific region; while primarily aimed at monitoring transmission and spread, they have also provided invaluable insights into how the virus evolves in the community during outbreaks. For example, a virological surveillance system in Sarawak, Malaysia, has shown that increased EV71 circulation occurred every 3 years (1997, 2000, 2003, 2006, and 2008/09), closely paralleling increased reports of HFMD in the community [119]. The phenomenon of regular cyclical epidemics has also been observed in Fukushima Prefecture, Japan [59]. Such cyclical activity is presumed to relate to the availability of new birth cohorts of susceptible children who have not been exposed to the virus during the earlier epidemics [23, 89]. Trying to make predictions about the epidemic potential of specific subgenogroups has proved difficult, beyond the observation that some subgenogroups, such as C1, appear to be less virulent and cause endemic disease rather than large epidemics, whereas other subgenogroups, particularly those that appear to have emerged in Asia in recent years, are associated with large epidemics.
More than one subgenogroup may co-circulate, and a shift from one dominant genogroup to another was described during outbreaks in Sarawak and Vietnam [12, 119, 146]. In Japan and Taiwan, subgenogroup B and C viruses have caused epidemics at different times (Table 12.1) [66, 82, 149]. On the contrary in the Netherlands, a permanent shift appears to have occurred, from genogroup B viruses prior to 1986 to genogroup C viruses since 1987; cross-neutralization among the genogroup B viruses, but not with genogroup C viruses, is postulated as one explanation for this, although other experimental cross-neutralization data did not support it [77, 102, 147, 148]. While the older subgenogroups of virus have been circulating and causing low levels of disease for many years, some of the more recently evolved subgenogroups such as genogroup B5, which possess distinctive antigenicity from other viruses may have the potential to cause massive epidemics [64, 148]. Although these have been confined to the Asia-Pacific region so far, the fact that humans are the natural reservoir for the virus and that international air travel is increasing means that every region is at risk of EV71 epidemic.
4 Biological Characteristics
EV71 is a small, non-enveloped virus with a positive-stranded RNA genome about 7.4 kb in size [169]. The genome has a single open reading frame encoding a polyprotein that is cleaved into 11 proteins: the four capsid proteins (P1 – VP1, VP2, VP3, VP4) and seven nonstructural proteins (P2 – 2A, 2B, 2C; P3 – 3A, 3B, 3C, 3D) [99]. The VP1 open reading frame has considerable genetic variability, which confers antigenic variability and enables investigators to differentiate EV71 strains [53, 100]. VP1 is also important for attachment to cellular receptors and for viral virulence [169]. Other details regarding the biological characteristics of EV71 were provided earlier in the molecular epidemiology section; additional general information regarding the characteristics of enteroviruses can be found in Chap. 11.
5 Clinical Features
EV71 infection may present with a wide spectrum of clinical features, although central nervous system (CNS) infection and HFMD are the two most commonly recognized disease manifestations [114].
5.1 Mucocutaneous Manifestations
HFMD is a common childhood exanthema characterized by a short, usually mild, febrile illness with papulovesicular rashes over the palms and soles and multiple mouth ulcers (Fig. 12.2).
Mucocutaneous lesions in hand-foot-and-mouth disease. This Malaysian child has ulcers that are seen inside the upper lip (top) and has vesicular and macular lesions on the wrists (middle) and soles (bottom) (Photos from T. Solomon) (Reproduced from Ooi et al. [111])
Herpangina, a closely related childhood enanthema, is characterized by a febrile illness associated with multiple mouth ulcers at the anterior pharyngeal folds, uvula, tonsils, and soft palate. Although the classical HFMD picture is typically seen in older children with EV71, widespread and atypical rashes may occur in children aged 2 years and below. Besides EV71, another picornavirus, CVA16, is also a principal causative agent of HFMD in both sporadic and epidemic forms. The virus is not normally associated with neurological complication [114], but the rash it causes is indistinguishable from that caused by EV71. There may be other clues that HFMD is due to EV71 rather than CVA16; for example, studies from Sarawak and Taiwan show that children with EV71 are more likely to have a longer duration of fever (≥3 days), a higher peak temperature (≥38.5 °C), lethargy, and myoclonus [108, 152]. A household contact study in Taiwan during the 2001–2002 epidemic showed that, in addition to HFMD and herpangina, there is a broad range of mild clinical manifestations, including upper respiratory tract infection, gastroenteritis, and nonspecific rashes [24]. Other respiratory manifestations in young children include acute exacerbation of bronchial asthma, bronchiolitis, and pneumonia [100]. More than 20 % of adult contacts during one Taiwanese outbreak had symptoms of an upper respiratory tract infection, while asymptomatic infection occurred in more than 50 %, indicating that adults may be important source of infection for younger children [24].
5.2 Neurological, Respiratory, and Systemic Manifestations
Similar to other enteroviruses, EV71 can cause aseptic meningitis, acute flaccid paralysis, encephalitis, and other rarer manifestations (Table 12.3) [96].
Encephalitis typically affects the brainstem, and unlike most other enteroviruses, it is often accompanied by marked cardiorespiratory dysfunction. This feature is also seen in poliomyelitis and has been attributed to neurogenic pulmonary edema [6], although the mechanism of disease remains controversial.
During a 7-year prospective clinical study of HFMD in Sarawak that covered several epidemics, 10–30 % of hospitalized children with HFMD due to EV71 had CNS complications [108, 112]. Brainstem encephalitis was the most common presentation, accounting for about 60 % of neurological syndromes, followed by aseptic meningitis (36 %) and brainstem encephalitis with cardiorespiratory dysfunction (4 %). Most children with CNS disease will also have features of HFMD; however, a small proportion may present with neurological disease only. Myoclonic jerks are seen more often with EV71 than with other enterovirus infections. This sign may be an early indicator of neurological involvement, particularly in the brainstem [90]. However, it is not pathognomonic for enterovirus infection; myoclonus has also been reported in other virus infections of the CNS including Japanese B encephalitis, subacute sclerosing panencephalitis, and Nipah virus, herpes simplex virus, varicella-zoster virus, and HIV infection. Myoclonic jerks are also often seen in otherwise healthy young infants, particularly when they are asleep and may occur spontaneously or be provoked by a loud noise.
Seizures, if they occur at all in EV71 infection, tend to be seen in younger children and are short-lived with rapid recovery of consciousness, suggesting that they are febrile convulsions, rather than due to CNS infection itself. Unlike those seen in other viral encephalitides, recurrent and prolonged seizures are very rare with EV71 infection, a distinction probably reflecting predominantly brainstem rather than cortical involvement.
Brainstem encephalitis with associated pulmonary edema has been the hallmark of EV71 CNS infection in Asia since the late 1990s. This distinctive clinical syndrome has a stereotypic clinical course characterized by a prodromal illness of HFMD followed by a sudden deterioration that typically occurs after 3–5 days of fever. Children then develop acute rapidly progressing cardiorespiratory failure presenting as shock and pulmonary edema or hemorrhages. Without critical care support, most of these children die within 24 h of hospital admission and in some even before arriving at the hospital. A system of clinical staging (stage 1 through stage 4) has been used to help monitor the progress of the affected children during the clinical course of EV71 infection and to guide management, from uncomplicated febrile illness to CNS involvement to cardiorespiratory failure and development of neurological sequelae [20, 85, 86]. Such staging systems have not been adopted widely, possibly because they are not always easy to remember and they imply sequential progression through stages that do not always occur. In 2010, a WHO Informal Consultation on Hand Foot and Mouth Disease proposed the use of a simple clinical description of disease manifestation to assess the disease severity. It included uncomplicated HFMD/herpangina, HFMD with CNS involvement, HFMD with autonomic system dysregulation, and HFMD with cardiopulmonary failure [160].
Findings on magnetic resonance imaging (MRI) of children with brainstem encephalitis correlated well with those of autopsy examination; both procedures demonstrate frequent involvement of the medulla oblongata, reticular formation, pons, and midbrain in several studies (Fig. 12.3) [29, 131, 162].
MRI changes in EV71 encephalomyelitis (Modified from Shen et al. [131]). T2-weighted images of a 10-month-old female who presented 3 months earlier with somnolence tachycardia, tachypnea, and coma and who recovered consciousness but remained ventilator dependent. (a) Sagittal section showing high signal intensity in the posterior portion of the pons and medulla (black arrows) and anterior cervical cord (white arrows). (b) Axial section showing the high signal intensity in the two anterior horns of the cervical cords (black arrows)
Acute flaccid paralysis is the primary presenting feature of a number of neurological syndromes caused by EV71 including poliomyelitis-like paralysis, Guillain-Barré syndrome, and transverse myelitis. Poliomyelitis-like paralysis is probably the most common of these, though it may be less severe than that caused by polioviruses, with a higher recovery rate [96]. Other respiratory manifestations in young children include acute exacerbation of bronchial asthma, bronchiolitis, and pneumonia [100]. More than 20 % of adult contacts during one Taiwanese outbreak had symptoms of an upper respiratory tract infection, while asymptomatic infection occurred in more than 50 %, indicating that adults may be important source of infection for younger children [24].
6 Clinical Management
During outbreaks of EV71, tens of thousands of children develop symptoms, and while most of them have mild self-limiting illness, in a small proportion of apparently well children, the condition can rapidly deteriorate to severe and fatal neurological and systemic complications over days or even hours. Whereas in the past children with mild HFMD tended to be managed at home, with increasing parental awareness about the risk of fatal complications, many are now brought directly to hospital, and health services can easily become overwhelmed. The challenges faced by primary care clinicians are recognizing which patients are likely to deteriorate, knowing which investigations yield the best diagnostic information, and deciding which treatments might be appropriate, without the benefit of guidance from controlled clinical trials.
6.1 Laboratory Tests
In mild disease, the blood count is usually normal, but in severe disease, the white blood cell count is often high with a neutrophilia [25]. Blood urea and electrolytes are typically unaffected, but there may be hyperglycemia in severe disease [25]. Creatine kinase is sometimes elevated in patients with cardiac involvement [46] and elevated cardiac troponin I has been reported as a predictor of imminent cardiopulmonary failure in children with brainstem encephalitis [65]. Chest X-ray characteristically shows a normal heart size, even in the presence of marked pulmonary congestion, indicating that neither acute viral myocarditis nor congenital heart disease is causing the illness. There are often nonspecific ECG changes [46], and continuous monitoring may show abnormal beat-to-beat variability, which may predict imminent cardiovascular collapse [83]. Echocardiography shows generalized left ventricular hypokinesia, occasionally accompanied by mitral regurgitation, in children who are hemodynamically unstable with tachycardia, hypotension, or pulmonary edema [46]. Pericardial effusion is rare.
Lumbar puncture is essential in children who are unwell with suspected CNS involvement. In some patients, the clinical features, such as meningismus or myoclonic jerks, may clearly point to CNS involvement. However in other children particularly those younger than 2 years of age, there may just be high fever, vomiting or lethargy, but a lumbar puncture reveals CNS disease. There is typically a mild CSF lymphocytic pleocytosis of 10–100 cells per mm3, but not always [116]. The CSF-to-plasma glucose ratio is usually normal but it can be low.
6.2 Virological Diagnosis
Laboratory diagnosis of EV71 is established primarily through virus isolation or molecular detection of the virus nucleic acid in appropriate clinical specimens.
6.2.1 Choice of Sample
A wide range of samples may be available, depending on the disease manifestations; these include throat and rectal swabs, serum, urine, and, when taken, cerebrospinal fluid (CSF), as well as fluid from vesicles and swabs from ulcers, if they are present. The sensitivity, specificity, and usefulness vary according to the sample [109]. In particular, virus detection in sterile sites such as vesicular fluid, CSF, serum, urine, serum, or autopsy material more reliably indicates a causative organism than does detection from non-sterile sites such as throat or rectum, which may indicate coincidental carriage. However, many of the sterile sites only occasionally yield virus. For example, virus is isolated from only 0–5 % of the CSF of patients with neurological disease [25, 40, 51, 67, 74, 103, 108, 112], because, as for poliomyelitis, the viral load in the CSF is very low [48]. The yield for serum is similarly low [112, 159]. Vesicular fluid, when present, is more useful. Although throat and rectal swabs are more likely to have an enterovirus detected, one study from Malaysia found that this was not always the same enterovirus as that isolated from a sterile site: using the isolate from vesicle swabs as a reference, 10 % of positive throat swabs gave a different isolate, and for rectal swabs the figure was 20 % [109]. Presumably, the isolate from the non-sterile site represented coincidental carriage, whereas that from the vesicles was actually pathogenic.
Prolonged viral shedding from the gastrointestinal tract (throat, rectum, or stool) may occur after complete resolution of EV71 infection, as it does for other enteroviruses; a study in Taiwan showed that EV71 may be detected in the throat up to 2 weeks after recovery from HFMD or herpangina; in the stool, it can be detected up to 11 weeks later [41]. During an outbreak, so many samples could potentially be positive that laboratories can soon become overwhelmed. In one study, the most efficient approach was to examine throat swabs for all patients plus swabs either from at least two vesicles when present or from the rectum when vesicles are absent [109].
6.2.2 Virus Isolation, Serotyping, and Nucleic Acid Detection
The gold standard for diagnosis of enterovirus infection is virus isolation. Several human and nonhuman primate cell lines may be used: rhabdomyosarcoma (RD), which is most efficient; human lung fibroblast cells (MRC5); and African green monkey kidney cells (Vero) [114]. In RD cells, a characteristic cytopathic effect is typically observed 7–10 days after inoculation. However, to improve the yield, blind passage may be necessary before cytopathic effects become apparent. Once a cytopathic effect is observed, the virus is identified by neutralization tests using intersecting pools of type-specific antisera, by EV71-specific antisera, or by an indirect immunofluorescence assay using EV71-specific monoclonal antibodies [114]. More recently, a molecular “serotyping” approach has been devised. It involves amplifying part of the VP1 gene of the cultured virus, using polymerase chain reaction (PCR) and pan-enterovirus EV71-specific primers, and then sequencing the product [104]. To this end, several sets of primers directed at different regions of the VP1 gene of human enterovirus have been developed [10, 104, 115].
EV71-specific primers are now also being used to perform PCR directly on clinical samples. The advantage of this approach over virus culture is that it can provide rapid diagnosis in the midst of explosive EV71 outbreaks where urgent public health intervention is needed. Several sets of real-time RT-PCR protocols directed to detect EV71 and CVA16 in primary clinical samples have been published recently; however, their disadvantage is that the technique detects only the suspected virus for which primers are available, but they will miss any agents that are unexpected or for which no primers have been generated [34, 115, 142, 167]. DNA microarray technology is a powerful, though expensive, new tool designed to detect multiple pathogen targets by hybridization of pathogen-specific probes. Two groups have recently reported using such an approach to distinguish EV71 and CVA16 infection in primary clinical specimens [34, 134].
6.3 Serology
Serological diagnosis of an acute virus infection is classically established by demonstrating a fourfold rise in specific neutralizing antibody between the acute and convalescent samples [114]. However, in the case of EV71, very high levels of neutralizing antibodies are often detectable within the first few days of illness, and thus a fourfold rise cannot be demonstrated [40, 103]. Furthermore, although homologous antibody is produced when young children encounter their first enterovirus infection, heterologous cross-reacting IgG and IgM antibodies are produced by older children and adults following repeated infection with different enterovirus serotypes; this reduces their diagnostic usefulness [87]. Several rapid IgM ELISA tests for EV71 have recently been developed to try to overcome some of these limitations [153]; however, cross-reactivity remains an issue [168], and the duration of detectable EV71-specific IgM following an infection is also uncertain.
6.4 Diagnostic Imaging
Computer tomography scans are almost always normal in EV71 encephalitis, where the pathology is mostly in the brainstem. Conventional MRI may be normal in the early phase of EV71 encephalitis. Conversely, it may show characteristic high signal intensities on T2-weighted and fluid-attenuation inversion recovery (FLAIR) images in the dorsal pons and medulla, most of the midbrain, and the dentate nuclei of the cerebellum. Similar high-signal lesions may also be found in the anterior horn cells of cervical spinal cord (Fig. 12.3) [63, 131, 175]. Gadolinium-enhanced MRI examination improves the results [175]. Diffuse-weighted imaging (DWI), a sensitive tool for detecting of early changes in brain cellular function, seems to be better at detecting EV71 encephalitis than conventional MRI [81]. However, the value of MRI screening has yet to be demonstrated. In children with acute flaccid paralysis, MRI typically shows unilateral high-signal lesions in the anterior horn cells of spinal cord on T2-wieghted images and contrast-enhancing ventral root on T1-weighted images [28, 63, 132].
6.5 Predictors of Severe Disease
Several clinical features and laboratory abnormalities have been associated with neurological and fatal EV71 disease, but few have been prospectively validated as prognostic indicators [25, 60, 65]. Younger age is associated with increased risk of severe disease [21]. A prospective clinical study of nearly 1,500 children presenting to one hospital in three EV71 outbreaks in Sarawak over 7 years suggested that peak temperature of 38.5 °C or higher, duration of fever for 3 or more days, and a history of lethargy were useful clinical predictors for neurological involvement. The presence of at least two of those three factors was strongly associated with the subsequent development of neurological disease [108]. The study corroborated the findings from early retrospective studies. However, it did not confirm other findings from earlier studies, such as the association between the absence of mouth ulcers and development of complicated or fatal disease [38, 112]. Hyperglycemia and leukocytosis have also been associated with fatal EV71 disease in a retrospective evaluation [25], and although these findings were confirmed in the prospective study, because they were late changes occurring about the same time as the fulminant disease, they were not helpful clinically in identifying children at high risk of complications and death [108].
Not all children with CNS involvement in EV71 infection will progress to cardiorespiratory collapse. A recent study in Southern Vietnam revealed that, in general, about 6 % of children with CNS involvement would develop autonomic system dysregulation. Children with frequent myoclonus observed by doctors and healthcare workers, with or without other focal neurological signs, were at higher risk of disease progression compared with those children with no history of myoclonus (22.2 % vs 4.4 %). A small study involving 46 patients showed that children developed abnormal heart rate variability, an index of autonomic nervous system disease, about 7 h before the clinical onset of cardiorespiratory instability [83]. The authors proposed that screening children with CNS involvement for abnormalities in heart rate variability may provide early warning of imminent cardiorespiratory failure and allow timely institution of appropriate interventions. Cardiac troponin I is a cardiac-specific biomarker for myocardial damage, used for early diagnosis of acute coronary syndrome in adults. Elevated cardiac troponin I has been observed in children with brainstem encephalitis and cardiopulmonary failure [65]; in some cases, it was elevated prior to the development of cardiopulmonary failure, suggesting that serial measurement of troponin may be helpful in identifying children at risk of left ventricular failure. However, neither the evaluation of heart rate variability nor the measurement of troponin I level has become routine clinical practice in the management of EV71 infection, probably because the former requires relatively sophisticated equipment not widely available and the latter is relatively expensive in developing countries. Overall simple clinical parameters such as length of illness, height of fever, and lethargy are probably more useful indicators of potentially severe disease.
6.6 Outcome
Follow-up for as long as 7 years after infection shows that children who present with aseptic meningitis generally have a favorable outcome, although a recent report documented the incidence of attention-deficit/hyperactivity disorder-related symptoms, as reported by parents and teachers, is higher (20 %) in children who had recovered from CNS infection compared with the matched controls (3 %) [49]. Approximately one-fifth of those with more severe neurological disease, including encephalitis, poliomyelitis-like paralysis, and encephalomyelitis, have sequelae, particularly focal limb weakness and atrophy [21, 110, 145]. Cerebellar dysfunction is observed in about 10 % of patients who had moderately severe brainstem encephalitis, including cranial neuropathies, myoclonus, tremor, and ataxia. However, only a quarter of those with more severe brainstem encephalitis associated with fulminant cardiorespiratory failure make a full neurological recovery. Common sequelae include focal limb weakness and atrophy, swallowing difficulties requiring nasogastric feeding, central hypoventilation, facial nerve palsies, seizures, and psychomotor retardation.
7 Pathogenesis
7.1 Viral Determinants of Virulence
The factors that determine whether EV71 infection will be asymptomatic or result in HFMD or severe neurological disease remain unknown. For poliovirus, the 5’UTR and VP1 genes are known to contain virulence determinants [72]. Several studies have therefore examined the nucleotide sequence of these genes, or the whole genome, comparing EV71 isolates from fatal and nonfatal cases, but for the most part the isolates have been nearly or entirely identical, and significant changes have not been found [135, 139]. The incidence of CNS disease and other severe complications of EV71 infection seems to have varied among the recent outbreaks in Asia, leading to the postulation that differences in the virulence of the various genotypes may have a role. However, comparisons between outbreaks have been hampered by the retrospective nature of many of the studies as well as differences in inclusion criteria, definitions of disease severity, and viral diagnostic approaches and capabilities. Perhaps the strongest data supporting the hypothesis that strain virulence determinants play an important role in the pathogenesis of severe neurological disease come from studies in Perth and Sarawak. During the Perth epidemic in 1999, two subgenogroups, B3 and C2, co-circulated, thus providing a unique opportunity to examine the role of virulence determinants in a single epidemic setting [96, 97]. In this outbreak, subgenogroup C2 viruses linked to the Taiwan epidemic of 1998 were almost exclusively isolated from children with severe neurological disease, and only a single isolate came from a case of uncomplicated HFMD. Conversely, subgenogroup B3 viruses, similar to those from the Sarawak 1997 epidemic, were isolated mainly from children with uncomplicated HFMD, aseptic meningitis, or post-infectious neurological disease, none of whom died [98]. A detailed prospective clinical study of EV71 disease in Sarawak, which included 277 children with EV71-associated HFMD, provided further clinical and epidemiological evidence for different biological behavior of EV71 subgenogroups, with regard to risk of CNS disease and transmissibility within families [112]. Two discrete EV71 epidemics, caused predominantly by subgenogroup B3 and B4, respectively, but with small numbers of cases caused by subgenogroup C1, occurred [112]. Children infected with subgenogroup B4 viruses were less likely to present with CNS infection than those infected with C1 or B5 viruses, and they were also less likely to be part of a family cluster. On the other hand, children infected with B5 were more likely to be part of a family cluster, and there was a trend toward a greater incidence of CNS disease in these patients. These results suggest that subgenogroups do indeed vary in their propensity to cause neurological disease.
7.2 Dual Infection
During the first of the Asian EV71 epidemics in Sarawak in 1997, which was due to subgenogroup B3, an adenovirus type 21 was also implicated in the fatal cases as well as in some cases with acute flaccid paralysis [13, 110]. The adenovirus was isolated from sterile sites such as CSF, brain, and heart in fatal cases and indeed was more frequently detected than EV71 itself; this led to the suggestion that the fatalities were due to dual infection, rather than EV71 alone [13]. However, subsequent detailed studies, including longitudinal studies from Sarawak, have not found evidence of adenovirus 21 infection in other HFMD or neurological cases, though dual infection of EV71 with other viruses, including dengue and Japanese B encephalitis, has been found [112]. Furthermore, adenovirus 21 has never been isolated in Sarawak since 1997.
7.3 Host Susceptibility
A range of host factors may affect pathogenesis, particularly partial cross-protective immunity from prior outbreaks. Lack of cross protection may partially explain why young age is a risk factor for severe disease [23, 24, 89]. The potential for host genetic variants to explain differential susceptibility, clinical severity, and outcome of EV71 has also been studied. One study in Taiwan reported that HLA-A33 is associated with increased susceptibility to EV71, although the role of the major histocompatibility complex in the virus infection is still unknown [19]. HLA-A33 is more frequently found in the populations of Asian ancestry than in those of European ancestry, and a causal association of this allele could help explain the higher frequency of EV71 epidemics in Asia. In the same study HLA-A2 was linked to the risk of cardiopulmonary failure often observed in fatal cases [19]. HLA-G, an important immunotolerant molecule, is involved in the suppression of T lymphocytes, NK cells, and antigen-presenting cells and in the induction of regulatory T cells and tolerant dendritic cells. Increased levels of cell surface bound HLA-G and plasma sHLA-G were found in EV71-infected children and in children with pulmonary edema [179]. Cytotoxic T lymphocyte antigen-4 (CTLA-4) is an important regulator of T cell cytotoxicity and is involved in the regulation of immune response. Studies of polymorphisms in the gene encoding CTLA-4 in children with meningoencephalitis have yielded conflicting results [19, 173]. Th17 cells are effector cells in human immune response, and its related cytokines, IL-17 and IL-23, are important mediators in proinflammatory response. Chinese children with the IL-17 F 7488C allele, which has been associated with a blunted proinflammatory response, were more likely to have milder EV71 infection, and patients who were homozygous for the T allele had significantly higher level of C-reactive protein, leukocytosis, and neutrophil counts when compared with patients with CC + CT genotypes [93]. Another Chinese study showed increased frequency of Th17 cells, as well as elevated serum IL-17 and IL-23, in peripheral blood of children infected with EV71 when compared to the healthy controls [32]. Interferon gamma, a Th1 antiviral cytokine, and IL-10, a potent anti-inflammatory cytokine that suppresses innate host defense, including interferon gamma production, have been implicated in the pathogenesis of severe EV71 infection. Susceptibility to EV71 encephalitis has been reported in children with the IFN-gamma + 874 T/A genotype, previously associated with reduced IFN-gamma production, and in children with the IL-10-1082G/A genotype, previously associated with reduced IL-10 production [172]. All of these associations require replication.
7.4 Viral Entry and Spread
EV71 is transmitted predominantly via the feco-oral route, with respiratory spread also implicated [114]. As for other enteroviruses, initial viral replication is presumed to occur in the lymphoid tissues of the oropharyngeal cavity (tonsils) and small bowel (Peyer’s patches), with further multiplication in the regional lymph nodes (deep cervical nodes, mesenteric nodes), giving rise to a mild viremia. The majority of infections are controlled at this point and remain asymptomatic. However, in vivo studies show that if enteroviruses disseminate further, they reach target organs, particularly the reticuloendothelial system (liver, spleen, bone marrow, and lymph nodes), heart, lung, pancreas, skin, mucous membranes, and central nervous system, coinciding with the onset of clinical features.
The mechanism by which enteroviruses enter the CNS is not completely understood. A number of epidemiological and experimental animal studies on polioviruses indicate that the virus can invade the CNS system by permeation through a disrupted blood-brain barrier or by retrograde axonal spread along cranial or peripheral nerves. For EV71, this latter route has been implicated both in mouse models and by examining the distribution of virus and inflammation in fatal human cases [31, 107, 163].
7.5 Pathological Findings
The topographical distribution of CNS inflammation has been stereotypic and is observed predominantly in the neuronal areas of spinal cord, the entire medulla oblongata except the pyramidal areas, the tegmentum and floor of fourth ventricles in the pons but not the anterior pons, and the whole midbrain sparing the cerebral peduncles. In addition, the hypothalamus, subthalamic and dentate nuclei, and to a lesser degree motor cortex of the cerebrum and meninges are involved (Fig. 12.4) [62, 63, 91, 92, 131, 133, 163].
Pathological findings in enterovirus 71 encephalitis (Modified from [163]). Parenchymal inflammation (arrows) and perivascular cuffing in the medulla (a); more severely inflamed areas (b), with edema and neuronophagia (c, arrows). More subtle inflammation in the motor cortex with mild perivascular cuffing (arrow) and parenchymal inflammatory cells (circle) (d). Numerous CD68-positive macrophages/microglial (e), CD8-positive lymphocyte adjacent to a neuron (f). Viral RNA in the anterior horn cells of the spinal cord (g). (a–d: hematoxylin and eosin stains; e, f: immunohistochemistry/peroxidase/DAB; g: ISH/nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl phosphate stains. Original magnification: (a) 4×; (b, d) 10×; (c, f) 40×; (g) 20×
Inflammatory changes were absent in the cerebellar cortex, thalamus, basal ganglia, mammillary body, hippocampus, temporal lobe, peripheral nerve, and autonomic ganglia. The histopathological changes, characterized by perivascular cuffing of macrophages, lymphocytes, neutrophils and plasma cells, variable edema and necrosis, focal neuronophagia, and microglia nodules, are similar to those in encephalitis caused by other viruses [50]. Neither virus inclusion nor vasculitis has been observed, and viral antigens and RNA can only be seen in a small number of neuronal processes and phagocytic cells [163, 164].
7.6 Pathogenesis of Severe Pulmonary Edema and Heart Failure
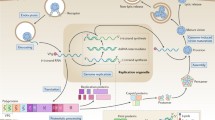
While it is clear that fulminant pulmonary edema is closely associated with, and preceded by CNS involvement, there is no consensus on its cause, especially the relative contributions of neurogenic pulmonary edema, cardiac dysfunction, increased vascular permeability, and a cytokine storm (Fig. 12.5).
The postulated pathogenesis of enterovirus 71-associated acute pulmonary edema Major postulated pathogenic pathways are shown with thick lines; lesser contributory pathways are shown with thinner lines. The solid boxes indicate strong supporting evidence from human clinical or pathological studies, while the dotted boxes indicate hypothetical but unproven steps or evidence from animal models only. EV71 human enterovirus 71, BBB blood-brain barrier, CNS central nervous system, ↑↑ markedly high, SVR systemic vascular resistance, SBP systemic blood pressure, HR heart rate, LV left ventricular (Reproduced from Solomon et al. [140])
Neurogenic pulmonary edema is classically seen following a head injury, where the associated raised intracranial pressure is thought to be important. Although the pathogenesis is not completely understood, studies from animal models suggest that the hypothalamus, and vasomotor centers of the medulla, and nuclei in the cervical spinal cord are important; lesions to various nuclei in these regions can increase activity along the sympathetic trunk, resulting in profound systemic and pulmonary hypertension and consequent pulmonary edema [57]. Pulmonary edema was also seen in poliomyelitis, and because it was associated with damage to brainstem nuclei, it was thought to be neurogenic in origin [6]. Thus when severe pulmonary edema was first seen in EV71 encephalitis, and brainstem inflammatory changes were observed too, the development of edema was attributed to neurogenic origin. Autopsy examination and magnetic resonance imaging studies of children with EV71 brainstem encephalitis showed that there was extensive inflammation of gray matter of the spinal cord and the whole medulla oblongata, as described above [62, 63, 91, 92, 131, 163]. The observations of hyperglycemia and leukocytosis were also postulated to be due to increased sympathetic discharges [25].
However, detailed hemodynamic observations of children with EV71 and pulmonary edema have not always shown the profound systemic and pulmonary hypertension that would be expected [46, 86, 89, 165] This may be because the changes in vascular pressures in neurogenic pulmonary edema are only transient, as was shown for one child with EV71 [165]. Others have argued that cardiac impairment is a major contributor to the pulmonary edema. Although there is no histological or virological evidence of a viral myocarditis, increased cardiac-specific troponin I level suggests some degree of cardiac injury [22, 46, 65, 91, 92]. A detailed echocardiographic study of 11 children with EV71 brainstem encephalitis shows that their cardiac function was impaired, with significantly reduced left ventricular ejection fraction [46]. Two children whose cardiac output was supported with a left ventricular assist device survived, whereas all the others died [45].
Although there is no myocardial inflammation, histological examination of heart ventricular tissue from six fatal cases and one survivor, obtained though a biopsy, revealed significant coagulative myocytolysis, myofibrillar degeneration, and cardiomyocyte apoptosis which are characteristic of catecholamine cardiotoxicity [46, 47]. Thus it is argued by some that the massive release of catecholamine caused by brainstem encephalitis may have a direct effect on cardiac function, as well as causing pulmonary edema through raised pulmonary pressures.
The other potential contributor to pulmonary edema is increased vascular permeability, which is postulated to have occurred following the systemic inflammatory response. Early studies that examined a narrow range of cytokines and chemokines have shown that interleukin (IL)-6, IL-1B, IL-10, IL-13, tumor necrosis factor (TNF)-α, and interferon (IFN)-γ are all significantly higher in EV71 patients with pulmonary edema than in those with uncomplicated encephalitis, and several of these, including IL-1, IL-6, IL-13, and IFN-γ, are known mediators of increased vascular permeability [84–86, 151]. In addition, increased plasma levels of several chemokines including interferon gamma-induced protein (IP-10), monocyte chemoattractant protein (MCP)-1, monokine induced by interferon gamma (MIG), and IL-8 have been found in children with pulmonary edema when compared to those with uncomplicated brainstem encephalitis [156]. A study of 30 cytokine and chemokine levels in EV71-infected Malaysian children confirmed that cardiorespiratory impairment was associated with a widespread elevation of both proinflammatory and anti-inflammatory mediators in the serum and CSF. In addition, the study also showed that serum IL-1Ra and G-CSF levels were significantly elevated in patients who died, with a serum G-CSF/IL-5 ratio of >100 being the most accurate prognostic marker [52]. Children with edema also had depleted lymphocyte population particularly in CD4, CD8, and natural killer cells [151, 173]. Thrombocytosis, neutrophilia, and hyperglycemia are all thought to be indicative of a systemic inflammatory response [84, 151]. Fewer studies have looked at cytokines in the CSF. Elevated CSF IL-1b was found in patients with encephalitis complicated by edema when compared to those with encephalitis alone [84]. It has previously been proposed that the CNS may be the source of the inflammatory cytokines detected in the serum of patients with EV71-associated cardiac dysfunction [156]. A study examining the relative abundance of inflammatory mediators in the serum and CSF samples from 88 Malaysian children showed two distinctive immune response patterns occurring independently in these two compartments [52]. Because the development of pulmonary edema in patients with EV71 encephalitis appears to be strongly associated with a dysregulated systemic and CNS inflammatory response, this observation has at least partly formed the basis for treatment with intravenous immunoglobulin, which appears to be effective [20, 85, 86, 108, 112, 154].
In summary, although still ill-defined, brainstem inflammation appears to be an important neurogenic mechanism for pulmonary edema in EV71 encephalitis. However, similar pathological changes are also observed in other encephalitides without pulmonary edema as a prominent feature. Cardiac compromise and the effects of the systemic inflammatory response on the vascular endothelium may also make an important contribution. In vivo mouse and nonhuman primate models have replicated some of the features of severe EV71 disease, such as neuroinvasiveness with marked inflammatory changes; however, no model has yet reproduced the severe systemic features such as pulmonary edema [5, 31, 36, 107, 158, 163].
8 Control and Prevention
8.1 Treatment
8.1.1 Antiviral Agents
There are no established antiviral treatments for EV71. Pleconaril is an antiviral drug that inhibits entry into cells for a number of enteroviruses by blocking viral attachment and uncoating. It has been used in clinical trials of aseptic meningitis [122–124]; however, it is not active against EV71 [35]. Several other newer capsid-function inhibitors have been investigated, and some have demonstrated promising anti-EV71 activities in preclinical studies [35]. Two human transmembrane proteins, P-selectin glycoprotein ligand-1 (PSGL-1) and scavenger receptor class B member 2 (SCARB2), have recently been identified to be functional receptors of EV71 [106, 170]. Analysis of the much-awaited crystal structure of EV71 revealed important structural differences from other enteroviruses. The mature virion of EV71 has a shallower canyon (believed to be the receptor binding site) on the viral surface and a relatively exposed “pocket factor” (which stabilizes the virus) compared to other enteroviruses [118, 157]. These discoveries are major steps forward and will guide rational design of antivirals against EV71.
8.1.2 Intravenous Immunoglobulin
Intravenous immunoglobulin (IVIG) was used during the initial epidemics of EV71 in Sarawak and Taiwan in the late 1990s on the basis that its anti-EV71 neutralizing antibodies and nonspecific anti-inflammatory properties might be therapeutic [112, 152]. Retrospective comparisons of patients who received IVIG with those who did not suggested possible benefit from IVIG if given early [20, 108]; for example, in Sarawak over 3 seasons, 204 (95 %) of 215 children who survived despite severe CNS complications had timely IVIG treatment, typically once severe disease occurred, whereas only one (11 %) of nine fatal cases received this treatment (OR 148.36, 95 % CI 16.34–6609.04, p < 0.0001) [108]. Proinflammatory cytokines measured before and after IVIG treatment were significantly lower in EV71 patients with autonomic dysfunction than patients with less severe disease [84–86, 151, 154]. IVIG has since become routine treatment for severe EV71 disease, and it has been recommended in the national treatment guidelines in Taiwan and Vietnam [20, 75, 85, 86, 108, 112, 150]. While remaining uncertainty over the efficacy of this treatment warrants randomized placebo-controlled trials, they would be logistically and ethically challenging to conduct in the face of the wide acceptance of IVIG as the current standard of care and the beliefs in their value strongly held by some. Carefully designed phase II trials would need surrogate end points for disease progression (e.g., failure of resolution of tachycardia).
8.1.3 Milrinone
Milrinone is a cyclic nucleotide phosphodiesterase (PDE) inhibitor currently used in the treatment of congestive heart failure. Inhibition of PDE subtype III by milrinone results in an increase in intracellular cyclic adenosine monophosphate (cAMP), which in turn leads to increased cardiac output and decreased peripheral vascular resistance. In a small nonrandomized retrospective comparison involving 24 children with EV71-induced pulmonary edema, those treated with milrinone had reduced tachycardia and lower mortality [150, 155]. Intriguingly, the peripheral white blood cell count, platelet count, and plasma IL-13 were also lower [155], suggesting an immunomodulatory as well as a cardiovascular effect of the drug. More recently a prospective, open-label, randomized controlled trial conducted by the same authors, involving 41 Vietnamese children 5 years of age and below, showed that milrinone significantly reduced the 1-week mortality from EV71-induced cardiovascular collapse without adverse effect. This encouraging finding has tentatively raised hope that earlier milrinone treatment might be useful in halting disease progression of cases with severe brainstem encephalitis [37].
8.1.4 Fluid Balance and Ionotrope Support
Hypovolemia and dehydration are the commonest causes of shock in children and are treated with rapid fluid resuscitation with good results. However, similar approaches used in the early EV71 epidemics in Asia precipitated massive pulmonary edema. Judicious use of intravenous fluid and early institution of inotrope support are critical in children with severe EV71 infection. Where fluid management could be guided by central venous pressure monitoring, in Taiwan, management algorithms based on this approach appear to have improved outcome [20].
8.1.5 Novel Treatment Approaches
Although better recognition of early signs of CNS involvement and the disease progression has helped improve the clinical outcome, many children continue to succumb to severe EV71 infection because of late presentation or disease progression despite intervention. A left ventricular assist device with extracorporeal membrane oxygenation was associated with a higher survival rate in a small number of recent cases than in historical controls, but with significant neurological sequelae [69]. Hemofiltration has been employed to treat children with cardiovascular collapse in Vietnam [117].
8.2 Prospects for Prevention
8.2.1 Surveillance
The only measures available currently for disease control at the population level are public health approaches. Countries in Asia (Singapore, Taiwan, Japan, and Vietnam) have implemented heightened surveillance for EV71 [2, 3, 33, 101, 119]. HFMD has now become a notifiable disease in many places, including Malaysia, Singapore, Thailand, Taiwan, Vietnam, and China [3, 4]. However, HFMD may be caused by a number of enteroviruses including CVA8, 10, and 16, and concurrent virological surveillance is necessary. Virological surveillance also provides invaluable molecular epidemiological data about the circulating EV71 genotype and may thus help to track the spread of the virus across the region.
Because humans are the only known natural hosts of human enteroviruses, outbreak control measures are targeted primarily at interrupting person-to-person virus transmission via contact with throat and nose secretions, saliva, stool, and vesicular fluid but also at minimizing contact with contaminated surfaces, toys, or fomites. Because of the lack of a lipid envelope, EV71 has considerable stability in the environment. It can remain viable at room temperature for several days and has been recovered from surface and ground water and hot spas [30, 61]. Hence, health education focuses on personal hygiene and good sanitation including frequent hand washing, proper disposal of soiled diapers, and disinfection of soiled surfaces with sodium hypochlorite [73]. Like other enteroviruses, EV71 is resistant to alcohol. Consequently, use of the widely available alcohol-based (70 % ethanol or isopropanol) disinfectants alone for hand hygiene is ineffective in preventing EV71 transmission [26]. A recent study showed that EV71 can be destroyed by virucidal disinfectants such as Virkon [18].
Transmission of enteroviruses including EV71 is most efficient in overcrowded settings, and most countries in the region including Malaysia, Singapore, Taiwan, Hong Kong, and China have adopted “social distancing” measures during epidemics. These measures include closures of childcare facilities and schools and cancelation of public functions involving children [2, 3]. Although there has been little systematic research to examine the effectiveness of public health interventions, a few studies of school closure or other approaches from Singapore and Hong Kong appeared to show some benefit [2, 95]. It nevertheless remains uncertain whether social distancing measures are effective or what the optimal timing for instituting them may be—at the onset of an HFMD outbreak or at the time of laboratory confirmation of an EV71 etiology. Although this widely adopted empirical measure has considerable socioeconomic impact, the cost of the disease and its control measures have not yet been studied. If experience with other directly transmissible viruses applies to EV71, then such measures as school closure could decrease the peak incidence but prolong the outbreak, with no reduction in the overall number of cases (Fig. 12.6) [140].
The effect of public health interventions on hand-foot-and-mouth disease outbreaks, comparing sentinel surveillance data from the 2003 outbreak in Sarawak, Malaysia, when the public health response was limited, and the 2006 outbreak, when more rigorous social distancing measures were encouraged. (Reproduced from Solomon et al. [140])
Further epidemiological work to elucidate the transmission dynamics of the virus will guide the formulation of evidence-based interventions to control the spread of EV71. Critical pieces of information such as precise estimates of the incubation period, ratio of asymptomatic to symptomatic cases, and time and duration of infectiousness are needed. The reproduction number (R0) for EV71 has been estimated to be higher than that of CVA16 (median 5.48, IQR 4.20–6.51 compared with 2.50 [1.96–3.67], respectively; p = 0.002) [94].
8.2.2 Vaccine Development
The success story of the control of poliomyelitis indicates that vaccines would be the best approach for future disease control, and the target population should be younger children, especially those less than 3 years of age, because they are at highest risk of severe disease. In fact, an inactivated EV71 whole virus vaccine was developed in Russia in 1976 after the Bulgaria epidemic. But it received no further evaluation because no further outbreaks of EV71 occurred [78]. More recent candidates for an EV71 vaccine include inactivated whole virus, live-attenuated, recombinant viral protein, virus-like particle, and DNA vaccines. These are at different stages of the development in China, Taiwan, and Singapore [78, 176]. Among these, inactivated whole virus vaccine candidates, the development of which modeled that of inactivated polio vaccine, are at the final stages of the clinical evaluation. A phase 3 randomized double-blinded, placebo-controlled trial of inactivated alum-adjuvant EV71 vaccine (Beijing Vigoo Biological, EV71 strain FY7VP5/AH/CHN/2008, genogroup C4) involving 10,245 Chinese children between the ages of 6 and 35 months has shown encouraging vaccine efficacy, immunogenicity, and safety [180]. Another alum-adjuvant inactivated whole virus candidate (EV71 strain H07, genogroup C4) produced by Sinovac Biotech Co., LTD) was reported to be well tolerated and highly immunogenic in a phase 1 trial in infant populations in China [80]. While an eagerly awaited vaccine will likely be available for routine use in the near future, it remains uncertain whether the vaccine developed from a specific genogroup would provide adequate cross protection against all genogroups. This concern is critical because genogroup B and C virus so far appear to have different geographical distributions in Asia, and the data to date on cross protection between genogroups are conflicting. Another important issue pertaining to manufacturing processes of inactivated whole virus vaccine is that no international reference standards for potency assays and quantification of EV71 vaccine antigens exist [78]. Establishment of such standards is urgent in Asia, where the vaccine will likely be used soonest and most extensively.
9 Concluding Remarks and Unsolved Problems
The emergence of EV71 in the Asia-Pacific region over the last 15 years has had a major public health impact. Molecular epidemiological studies suggest that some subgenogroups appear to have massive potential for explosive epidemics, while others circulate in a more indolent pattern. However, the biological determinants of these differences are poorly understood. The epidemiological differences observed between EV71 in the Asia-Pacific region and the strains found in Europe and USA also represent an unsolved puzzle. There are no reliable and easy-to-use clinical tools to predict who will develop neurological complications and which patients with CNS involvement are at risk of disease progression. The virological and host determinants of the wide-ranging clinical phenotypes in those infected remain unclear. There are relatively good animal models of neurological disease caused by EV71, but there is an urgent need for an animal model of cardiorespiratory dysfunction to advance understanding of its pathogenesis. Despite lack of solid evidence for its efficacy, the wide use of IVIG for severe EV71 infection in many Asian countries will make efficacy trials difficult. There is still no specific antiviral drug available for EV71 infection although the determination of its crystal structure and identification of several EV71 receptors should accelerate drug discovery. An inactivated whole virus vaccine is nearing clinical availability, but important steps must be taken to ensure that it ultimately reaches the populations in greatest need. Public health intervention and control measures of EV71 epidemics so far have been empirical and generic, not stringently evidence based. Because they have significant socioeconomic impact, further research is needed on the transmission dynamics of the virus and which of these public health intervention strategies most effectively limit the havoc wreaked by future EV71 outbreaks.
References
AbuBakar S, Sam IC, et al. Enterovirus 71 outbreak, Brunei. Emerg Infect Dis. 2009;15(1):79–82.
Ang LW, Koh BK, et al. Epidemiology and control of hand, foot and mouth disease in Singapore, 2001-2007. Ann Acad Med Singapore. 2009;38(2):106–12.
Anonymos. Forum on Hand Foot and Mouth Disease (HFMD) in Asia-Pacific Region: Epidemiological, Laboratory, Clinical, and Public Health aspects. Singapore, Regional Emerging Disease Intervention Center & The Ministry of Health Singapore: 17; 2009.
Anonymous. Forum on Hand Foot and Mouth Disease (HFMD) in Asia-Pacific Region: Epidemiological, Laboratory, Clinical, and Public Health aspects. Singapore, Regional Emerging Disease Intervention Center & The Ministry of Health Singapore: 17; 2009.
Arita M, Shimizu H, et al. Temperature-sensitive mutants of enterovirus 71 show attenuation in cynomolgus monkeys. J Gen Virol. 2005;86(5):1391–401.
Baker AB. Poliomyelitis. A study of pulmonary edema. Neurology. 1957;7(11):743–51.
Bessaud M, Pillet S, et al. Molecular characterization of human enteroviruses in the Central African Republic: uncovering wide diversity and identification of a New human enterovirus A71 genogroup. J Clin Microbiol. 2012;50(5):1650–8.
Bible JM, Iturriza-Gomara M, et al. Molecular epidemiology of human enterovirus 71 in the United Kingdom from 1998 to 2006. J Clin Microbiol. 2008;46(10):3192–200.
Blomberg J, Lycke E, et al. New enterovirus type associated with epidemic of aseptic meningitis and-or hand, foot, and mouth disease. Lancet. 1974;2(7872):112.
Brown B, Kilpatrick D, et al. Serotype-specific identification of enterovirus 71 by PCR. J Clin Virol. 2000;16:107–12.
Brown BA, Oberste MS, et al. Molecular epidemiology and evolution of enterovirus 71 strains isolated from 1970 to 1998. J Virol. 1999;73(12):9969–75.
Cardosa M, Perera D, et al. Molecular epidemiology of human enterovirus 71 strains and recent outbreaks in the Asia-Pacific region: comparative analysis of the VP1 and VP4 genes. Emerg Infect Dis. 2003;9(4):461–8.
Cardosa MJ, Krishnan S, et al. Isolation of subgenus B adenovirus during a fatal outbreak of enterovirus 71-associated hand, foot, and mouth disease in Sibu, Sarawak. Lancet. 1999;354(9183):987–91.
Castro CM, Cruz AC, et al. Molecular and seroepidemiologic studies of Enterovirus 71 infection in the State of Para, Brazil. Rev Inst Med Trop Sao Paulo. 2005;47(2):65–71.
Chakraborty R, Iturriza-Gomara M, et al. An epidemic of enterovirus 71 infection among HIV-1-infected orphans in Nairobi. AIDS. 2004;18(14):1968–70.
Chan K, Goh K, et al. Epidemic hand, foot and mouth disease caused by human enterovirus 71, Singapore. Emerg Infect Dis. 2003;9:78–85.
Chan L, Parashar U, et al. Deaths of children during an outbreak of hand, foot, and mouth disease in Sarawak, Malaysia: clinical and pathological characteristics of the disease. Clin Infect Dis. 2000;31:678–83.
Chan YF, Abu Bakar S. Virucidal activity of Virkon S on human enterovirus. Med J Malaysia. 2005;60(2):246–8.
Chang L-Y, Chang IS, et al. HLA-A33 is associated with susceptibility to enterovirus 71 infection. Pediatrics. 2008;122(6):1271–6.
Chang L-Y, Hsia S-H, et al. Outcome of enterovirus 71 infections with or without stage-based management: 1998 to 2002. Pediatr Infect Dis J. 2004;23(4):327–31.
Chang L-Y, Huang L-M, et al. Neurodevelopment and cognition in children after enterovirus 71 infection. N Engl J Med. 2007;356(12):1226–34.
Chang L-Y, Huang Y-C, et al. Fulminant neurogenic pulmonary oedema with hand, foot, and mouth disease. Lancet. 1998;352(9125):367–8.
Chang L-Y, King C-C, et al. Risk factors of enterovirus 71 infection and associated hand, foot, and mouth disease/herpangina in children during an epidemic in Taiwan. Pediatrics. 2002;109(6):e88.
Chang L-Y, Tsao K-C, et al. Transmission and clinical features of enterovirus 71 infections in household contacts in Taiwan. JAMA. 2004;291(2):222–7.
Chang L, Lin T, et al. Clinical features and risk factors of pulmonary oedema after enterovirus-71-related hand, foot, and mouth disease. Lancet. 1999;354(9191):1682–6.
Chang SC, Li WC, et al. Efficacy of alcohols and alcohol-based hand disinfectants against human enterovirus 71. J Hosp Infect. 2013;83(4):288–93.
Chatproedprai S, Theanboonlers A, et al. Clinical and molecular characterization of hand-foot-and-mouth disease in Thailand, 2008–2009. Jpn J Infect Dis. 2010;63(4):229–33.
Chen C-Y, Chang Y-C, et al. Acute flaccid paralysis in infants and young children with enterovirus 71 infection: MR imaging findings and clinical correlates. AJNR Am J Neuroradiol. 2001;22(1):200–5.
Chen C-Y, Chang Y-C, et al. MR imaging findings of epidemic enterovirus 71 encephalitis in infants and young children. Chin J Radiol. 2000;25(2):45–52.
Chen CH, Hsu BM, et al. Molecular detection and prevalence of enterovirus within environmental water in Taiwan. J Appl Microbiol. 2008;104(3):817–23.
Chen CS, Yao YC, et al. Retrograde axonal transport: a major transmission route of enterovirus 71 in mice. J Virol. 2007;81(17):8996–9003.
Chen J, Tong J, et al. Increased frequency of Th17 cells in the peripheral blood of children infected with enterovirus 71. J Med Virol. 2012;84(5):763–7.
Chen K, Chang H, et al. Epidemiologic features of hand-foot-mouth disease and herpangina caused by enterovirus 71 in Taiwan, 1998-2005. Pediatrics. 2007;120(2):244–52.
Chen T-C, Chen G-W, et al. Combining multiplex reverse transcription-PCR and a diagnostic microarray to detect and differentiate enterovirus 71 and coxsackievirus A16. J Clin Microbiol. 2006;44(6):2212–9.
Chen T-C, Weng K-F, et al. Development of antiviral agents for enteroviruses. J Antimicrob Chemother. 2008;62(6):1169–73.
Chen YC, Yu CK, et al. A murine oral enterovirus 71 infection model with central nervous system involvement. J Gen Virol. 2004;85:69–77.
Chi C-Y, Khanh TH, et al. Milrinone therapy for enterovirus 71-induced pulmonary edema and/or neurogenic shock in children: a randomized controlled trial*. Crit Care Med. 2013;41(7):1754–60. doi:10.1097/CCM.0b013e31828a2a85.
Chong CY, Chan KP, et al. Hand, foot and mouth disease in Singapore: a comparison of fatal and non-fatal cases. Acta Paediatr. 2003;92(10):1163–9.
Chua KB, Chua BH, et al. Genetic diversity of enterovirus 71 isolated from cases of hand, foot and mouth disease in the 1997, 2000 and 2005 outbreaks, Peninsular Malaysia. Malays J Pathol. 2007;29(2):69–78.
Chumakov M, Voroshilova M, et al. Enterovirus 71 isolated from cases of epidemic poliomyelitis-like disease in Bulgaria. Arch Virol. 1979;60(3–4):329–40.
Chung P-W, Huang Y-C, et al. Duration of enterovirus shedding in stool. J Microbiol Immunol Infect. 2001;34(3):167–70.
Da Silva EE, Winkler MT, et al. Role of enterovirus 71 in acute flaccid paralysis after the eradication of poliovirus in Brazil. MD Computing. 1996;13(4):231–3.
Deibel R, Flanagan TD. Central nervous system infections. Etiologic and epidemiologic observations in New York State, 1976-1977. N Y State J Med. 1979;79(5):689–95.
Deshpande JM, Nadkarni SS, et al. Enterovirus 71 isolated from a case of acute flaccid paralysis in India represents a new genotype. Curr Sci. 2003;84(10):1350–3.
Fu Y-C, Chi C-S, et al. Pulmonary edema of enterovirus 71 encephalomyelitis is associated with left ventricular failure: implications for treatment. Pediatr Pulmonol. 2003;35(4):263–8.
Fu YC, Chi CS, et al. Cardiac complications of enterovirus rhombencephalitis. Arch Dis Child. 2004;89(4):368–73.
Fu YC, Chi CS, et al. Comparison of heart failure in children with enterovirus 71 rhombencephalitis and cats with norepinephrine cardiotoxicity. Pediatr Cardiol. 2006;27(5):577–84.
Fujimoto T, Yoshida S, et al. Detection and quantification of enterovirus 71 genome from cerebrospinal fluid of an encephalitis patient by PCR applications. Jpn J Infect Dis. 2008;61(6):497–9.
Gau SS, Chang LY, et al. Attention-deficit/hyperactivity-related symptoms among children with enterovirus 71 infection of the central nervous system. Pediatrics. 2008;122(2):e452–8.
German AC, Myint KS, et al. A preliminary neuropathological study of Japanese encephalitis in humans and a mouse model. Trans R Soc Trop Med Hyg. 2006;100(12):1135–45.
Gilbert GLMFF, Dickson KEBAS, et al. Outbreak of enterovirus 71 infection in Victoria, Australia, with a high incidence of neurologic involvement. Pediatr Infect Dis J. 1988;7(7):484–7.
Griffiths MJ, Ooi MH, et al. In enterovirus 71 encephalitis with cardio-respiratory compromise, elevated interleukin 1β, interleukin 1 receptor antagonist, and granulocyte colony-stimulating factor levels are markers of poor prognosis. J Infect Dis. 2012;206(6):881–92.
Guan D, van der Sanden S, Zeng H, Li W, Zheng H, Ma C, Su J, Liu Z, Guo X, Zhang X, Liu L, Koopmans M, Ke C. Population dynamics and genetic diversity of C4 strains of human enterovirus 71 in Mainland China, 1998-2010. PLoS One. 2012;7(9):e44386. doi:10.1371/journal.pone.0044386. Epub 2012 Sep 12.
Hagiwara A, Tagaya I, et al. Epidemic of hand, foot and mouth disease associated with enterovirus 71 infection. Intervirology. 1978;9(1):60–3.
Han J, Ma X-J, et al. Long persistence of EV71 specific nucleotides in respiratory and feces samples of the patients with Hand-Foot-Mouth Disease after recovery. BMC Infect Dis. 2010;10(1):178.
Ho M, Chen ER, et al. An epidemic of enterovirus 71 infection in Taiwan. N Engl J Med. 1999;341(13):929–35.
Hoff JT, Nishimura M, et al. Experimental neurogenic pulmonary edema. Part 1: the role of systemic hypertension. J Neurosurg. 1981;54(5):627–31.
Honkanen H, Oikarinen S, et al. Human enterovirus 71 strains in the background population and in hospital patients in Finland. J Clin Virol. 2012;56(4):348–53.
Hosoya M, Kawasaki Y, et al. Genetic diversity of enterovirus 71 associated with hand, foot and mouth disease epidemics in Japan from 1983 to 2003. Pediatr Infect Dis J. 2006;25(8):691–4.
Hsia SH, Wu CT, et al. Predictors of unfavorable outcomes in enterovirus 71-related cardiopulmonary failure in children. Pediatr Infect Dis J. 2005;24(4):331–4.
Hsu BM, Chen CH, et al. Prevalence of enteroviruses in hot spring recreation areas of Taiwan. FEMS Immunol Med Microbiol. 2008;52(2):253–9.
Hsueh C, Jung SM, et al. Acute encephalomyelitis during an outbreak of enterovirus type 71 infection in Taiwan: report of an autopsy case with pathologic, immunofluorescence, and molecular studies. Mod Pathol. 2000;13(11):1200–5.
Huang C-C, Liu C-C, et al. Neurologic complications in children with enterovirus 71 infection. N Engl J Med. 1999;341(13):936–42.
Huang SW, Hsu YW, et al. Reemergence of enterovirus 71 in 2008 in Taiwan: dynamics of genetic and antigenic evolution from 1998 to 2008. J Clin Microbiol. 2009;47(11):3653–62.
Huang Y-F, Chiu P-C, et al. Cardiac troponin I: a reliable marker and early myocardial involvement with meningoencephalitis after fatal enterovirus-71 infection. J Infect. 2003;46(4):238–43.
Huang YP, Lin TL, et al. The circulation of subgenogroups B5 and C5 of enterovirus 71 in Taiwan from 2006 to 2007. Virus Res. 2008;137(2):206–12.
Ishimaru Y, Nakano S, et al. Outbreaks of hand, foot, and mouth disease by enterovirus 71. High incidence of complication disorders of central nervous system. Arch Dis Child. 1980;55(8):583–8.
Iwai M, Masaki A, et al. Genetic changes of coxsackievirus A16 and enterovirus 71 isolated from hand, foot, and mouth disease patients in Toyama, Japan between 1981 and 2007. Jpn J Infect Dis. 2009;62(4):254–9.
Jan SL, Lin SJ, et al. Extracorporeal life support for treatment of children with enterovirus 71 infection-related cardiopulmonary failure. Intensive Care Med. 2009;36(3):520–7.
Jee YM, Cheon D-S, et al. Genetic analysis of the VP1 region of human enterovirus 71 strains isolated in Korea during 2000. Arch Virol. 2003;148(9):1735–46.
Jeong EJ, Lee JH, et al. Molecular characterization of enteroviruses detected in Gyeong-Ju and Po-Hang provinces of Korea in 2003. Arch Virol. 2010;155(10):1707–12.
Jesus NHD. Epidemics to eradication: the modern history of poliomyelitis. Virol J. 2007;4:70.
Kadurugamuwa JL, Shaheen E. Inactivation of human enterovirus 71 and coxsackie virus A16 and hand, foot, and mouth disease. Am J Infect Control. 2011;39(9):788–9.
Kennett ML, Birch CJ, et al. Enterovirus type 71 infection in Melbourne. Bull World Health Organ. 1975;51(6):609–15.
Khanh TH, Sabanathan S, et al. Enterovirus 71–associated hand, foot, and mouth disease, southern Vietnam, 2011. Emerg Infect Dis. 2012;18(12).
Komatsu H, Shimizu Y, et al. Outbreak of severe neurologic involvement associated with Enterovirus 71 infection. Pediatr Neurol. 1999;20(1):17–23.
Kung SH, Wang SF, et al. Genetic and antigenic analyses of enterovirus 71 isolates in Taiwan during 1998-2005. Clin Microbiol Infect. 2007;13(8):782–7.
Lee MS, Tseng FC, et al. Challenges to licensure of enterovirus 71 vaccines. PLoS Negl Trop Dis. 2012;6(8):e1737.
Li L, He Y, et al. Genetic characteristics of human enterovirus 71 and coxsackievirus A16 circulating from 1999 to 2004 in Shenzhen, People’s Republic of China. J Clin Microbiol. 2005;43(8):3835–9.
Li Y-P, Liang Z-L, et al. Safety and immunogenicity of a novel human Enterovirus 71 (EV71) vaccine: a randomized, placebo-controlled, double-blind, Phase I clinical trial. Vaccine. 2012;30(22):3295–303.
Lian Z, Huang B, et al. Diffusion-weighted imaging in the diagnosis of enterovirus 71 encephalitis. Acta Radiol. 2012;53(2):208–13.
Lin K-H, Hwang K-P, et al. Evolution of EV71 genogroup in Taiwan from 1998 to 2005: an emerging of subgenogroup C4 of EV71. J Med Virol. 2006;78(2):254–62.
Lin M-T, Wang J-K, et al. Heart rate variability monitoring in the detection of central nervous system complications in children with enterovirus infection. J Crit Care. 2006;21(3):280–6.
Lin T-Y, Hsia S-H, et al. Proinflammatory cytokine reactions in enterovirus 71 infections of the central nervous system. Clin Infect Dis. 2003;36(3):269–74.
Lin TY, Chang LY, et al. The 1998 enterovirus 71 outbreak in Taiwan: pathogenesis and management. Clin Infect Dis. 2002;34 Suppl 2.
Lin TY, Chang LY, et al. Different proinflammatory reactions in fatal and non-fatal enterovirus 71 infections: implications for early recognition and therapy. Acta Paediatr. 2002;91(6):632–5 (Oslo, Norway: 1992).
Lin Y, Wen K, et al. Cross-reactivity of anti-Ev71 IgM and neutralizing antibody in series sera of patients infected with Enterovirus 71 and Coxsackievirus A 16. J Immunoassay Immunochem. 2011;32(3):233–43.
Liu MY, Liu W, et al. Characterization of an outbreak of hand, foot, and mouth disease in Nanchang, China in 2010. PLoS One. 2011;6(9):e25287.
Lu C-Y, Lee C-Y, et al. Incidence and case-fatality rates resulting from the 1998 enterovirus 71 outbreak in Taiwan. J Med Virol. 2002;67(2):217–23.
Lu H-K, Lin T-Y, et al. Prognostic implications of myoclonic jerk in children with enterovirus infection. J Microbiol Immunol Infect. 2004;37(2):82–7.
Lum LC, Wong KT, et al. Neurogenic pulmonary oedema and enterovirus 71 encephalomyelitis. Lancet. 1998;352(9137):1391.
Lum LC, Wong KT, et al. Fatal enterovirus 71 encephalomyelitis. J Pediatr. 1998;133(6):795–8.
Lv T, Li J, et al. Association of interleukin-17F gene polymorphism with enterovirus 71 encephalitis in patients with hand, foot, and mouth disease. Inflammation. 2013;36(4):977–81.
Ma E, Fung C, et al. Estimation of the basic reproduction number of enterovirus 71 and coxsackievirus A16 in hand, foot, and mouth disease outbreaks. Pediatr Infect Dis J. 2011;30(8):675–9. doi:10.1097/INF.0b013e3182116e95.
Ma E, Wong S, et al. Effects of public health interventions in reducing transmission of hand, foot, and mouth disease. Pediatr Infect Dis J. 2011;30(5):432–5. doi:10.1097/INF.0b013e3182127782.
McMinn P. An overview of the evolution of enterovirus 71 and its clinical and public health significance. FEMS Microbiol Rev. 2002;26(1):91–107.
McMinn P, Lindsay K, et al. Phylogenetic analysis of enterovirus 71 strains isolated during linked epidemics in Malaysia, Singapore, and Western Australia. J Virol. 2001;75:7732–8.
McMinn P, Stratov I, et al. Neurological manifestations of enterovirus 71 infection in children during an outbreak of hand, foot, and mouth disease in Western Australia. Clin Infect Dis. 2001;32(2):236–42.
McMinn PC. Recent advances in the molecular epidemiology and control of human enterovirus 71 infection. Curr Opin Virol. 2012;2(2):199–205.
Merovitz L, Demers A-M, et al. Enterovirus 71 infections at a canadian center. Pediatr Infect Dis J. 2000;19(8):755–7.
Mizuta K, Abiko C, et al. Frequent importation of enterovirus 71 from surrounding countries into the local community of Yamagata, Japan, between 1998 and 2003. J Clin Microbiol. 2005;43(12):6171–5.
Mizuta K, Aoki Y, et al. Cross-antigenicity among EV71 strains from different genogroups isolated in Yamagata, Japan, between 1990 and 2007. Vaccine. 2009;27(24):3153–8.
Nagy G, Takatsy S, et al. Virological diagnosis of enterovirus type 71 infections: experiences gained during an epidemic of acute CNS diseases in Hungary in 1978. Arch Virol. 1982;71(3):217–27.
Nasri D, Bouslama L, et al. Basic rationale, current methods and future directions for molecular typing of human enterovirus. Expert Rev Mol Diagn. 2007;7(4):419–34.
Ni H, Yi B, et al. Epidemiological and etiological characteristics of hand, foot, and mouth disease in Ningbo, China, 2008–2011. J Clin Virol. 2012;54(4):342–8.
Nishimura Y, Shimojima M, et al. Human P-selectin glycoprotein ligand-1 is a functional receptor for enterovirus 71. Nat Med. 2009;15(7):794–7.
Ong KC, Badmanathan M, et al. Pathologic characterization of a murine model of human enterovirus 71 encephalomyelitis. J Neuropathol Exp Neurol. 2008;67(6):532–42.
Ooi M, Wong S, et al. Identification and validation of clinical predictors for the risk of neurological involvement in children with hand, foot, and mouth disease in Sarawak. BMC Infect Dis. 2009;9(1):3.
Ooi MH, Solomon T, et al. Evaluation of different clinical sample types in diagnosis of human enterovirus 71-associated hand-foot-and-mouth disease. J Clin Microbiol. 2007;45(6):1858–66.
Ooi MH, Wong SC, et al. Adenovirus type 21-associated acute flaccid paralysis during an outbreak of hand-foot-and-mouth disease in Sarawak, Malaysia. Clin Infect Dis. 2003;36(5):550–9.
Ooi MH, Wong SC, et al. Clinical features, diagnosis and management of human enterovirus 71 infection. Lancet Neurol. 2010;9(11):1097–105.
Ooi MH, Wong SC, et al. Human enterovirus 71 disease in Sarawak, Malaysia: a prospective clinical, virological, and molecular epidemiological study. Clin Infect Dis. 2007;44(5):646–56.
Ortner B, Huang CW, et al. Epidemiology of enterovirus types causing neurological disease in Austria 1999-2007: detection of clusters of echovirus 30 and enterovirus 71 and analysis of prevalent genotypes. J Med Virol. 2009;81(2):317–24.
Pallansch MA, Ross RP. Enteroviruses: polioviruses, coxsackieviruses, echoviruses, and newer enteroviruses. Fields Virol. 2001;1:723–75.
Perera D, Podin Y, et al. Incorrect identification of recent Asian strains of coxsackievirus A16 as human enterovirus 71: Improved primers for the specific detection of human enterovirus 71 by RT-PCR. BMC Infect Dis. 2004;4:11.
Perez-Velez CM, Anderson MS, et al. Outbreak of neurologic enterovirus type 71 disease: a diagnostic challenge. Clin Infect Dis. 2007;45(8):950–7.
Phan P, Dau H, et al. Continuous veno-venous hemofiltration for treatment of enterovirus 71-induced fulminant cardiopulmonary failure: a case report. J Med Case Reports. 2012;6(1):159.
Plevka P, Perera R, et al. Crystal structure of human enterovirus 71. Science. 2012;336(6086):1274.
Podin Y, Gias E, et al. Sentinel surveillance for human enterovirus 71 in Sarawak, Malaysia: lessons from the first 7 years. BMC Public Health. 2006;6(1):180.
Prager P, Nolan M, et al. Neurogenic pulmonary edema in enterovirus 71 encephalitis is not uniformly fatal but causes severe morbidity in survivors. Pediatr Crit Care Med. 2003;4(3):377–81.
Puenpa J, Theamboonlers A, et al. Molecular characterization and complete genome analysis of human enterovirus 71 and coxsackievirus A16 from children with hand, foot and mouth disease in Thailand during 2008–2011. Arch Virol. 2011;156(11):2007–13.
Rotbart HA. Antiviral therapy for enteroviruses and rhinoviruses. Antivir Chem Chemother. 2000;11(4):261–71.
Rotbart HA, O’Connell JF, et al. Treatment of human enterovirus infections. Antiviral Res. 1998;38(1):1–14.
Rotbart HA, Webster AD. Treatment of potentially life-threatening enterovirus infections with pleconaril. Clin Infect Dis. 2001;32(2):228–35.
Ryu W-S, Kang B, et al. Clinical and etiological characteristics of enterovirus 71-related diseases during a recent 2-year period in Korea. J Clin Microbiol. 2010;48(7):2490–4.
Samuda GM, Chang WK, et al. Monoplegia caused by Enterovirus 71: an outbreak in hong Kong. Pediatr Infect Dis J. 1987;6(2):206–8.
Sanders SA, Herrero LJ, et al. Molecular epidemiology of enterovirus 71 over two decades in an Australian urban community. Arch Virol. 2006;151(5):1003–13.
Schmidt N, Lennette E, et al. An apparently new enterovirus isolated from patients with disease of the central nervous system. J Infect Dis. 1974;129:304–9.
Seiff A. Cambodia unravels cause of mystery illness. Lancet. 2012;380(9838):205.
Badran SA, Midgley S, et al. Clinical and virological features of enterovirus 71 infections in Denmark, 2005 to 2008. Scand J Infect Dis. 2011;43(8):642–8.
Shen W-C, Chiu H-H, et al. MR imaging findings of enteroviral encephalomyelitis: an outbreak in Taiwan. AJNR Am J Neuroradiol. 1999;20(10):1889–95.
Shen WC, Tsai C-H, et al. MRI of Enterovirus 71 myelitis with monoplegia. Neuroradiology. 2000;42(2):124–7.
Shieh W-J, Jung S-M, et al. Pathologic studies of fatal cases in outbreak of hand, foot, and mouth disease, Taiwan. Emerg Infect Dis. 2001;7(1):146–8.
Shih S-R, Wang Y-W, et al. Serotype-specific detection of enterovirus 71 in clinical specimens by DNA microchip array. J Virol Methods. 2003;111(1):55–60.
Shih S, Ho M, et al. Genetic analysis of enterovirus 71 isolated from fatal and non-fatal cases of hand, foot and mouth disease during an epidemic in Taiwan, 1998. Virus Res. 2000;68(2):127–36.
Shimizu H. Molecular epidemiology of enterovirus 71 infection in the Western Pacific Region. Pediatr Int. 2004;46:231–5.
Shindarov LM, Chumakov MP, et al. Epidemiological, clinical, and pathomorphological characteristics of epidemic poliomyelitis-like disease caused by enterovirus 71. J Hyg Epidemiol Microbiol Immunol. 1979;23(3):284–95.
Siafakas N, Attilakos A, et al. Molecular detection and identification of enteroviruses in children admitted to a university hospital in Greece. Mol Cell Probes. 2011;25(5–6):249–54.
Singh S, Poh C, et al. Complete sequence analyses of enterovirus 71 strains from fatal and non-fatal cases of the hand, foot and mouth disease outbreak in Singapore (2000). Microbiol Immunol. 2002;46(11):801–8.
Solomon T, Lewthwaite P, et al. Virology, epidemiology, pathogenesis, and control of enterovirus 71. Lancet Infect Dis. 2010;10(11):778–90.
Tan CY, Gonfrier G, et al. Screening and detection of human enterovirus 71 infection by a real-time RT-PCR assay in Marseille, France, 2009–2011. Clin Microbiol Infect. 2012;18(4):E77–80.
Tan EL, Chow VT, et al. Development of multiplex real-time hybridization probe reverse transcriptase polymerase chain reaction for specific detection and differentiation of Enterovirus 71 and Coxsackievirus A16. Diagn Microbiol Infect Dis. 2008;61(3):294–301.
Tao Z, Wang H, et al. Identification of a C2 subgenogroup strain of enterovirus 71 in a retrospective study in Shandong Province, China, from 1990 to 2010. J Clin Microbiol. 2012;50(5):1823–4.
Tee KK, Lam TT, et al. Evolutionary genetics of human enterovirus 71: origin, population dynamics, natural selection, and seasonal periodicity of the VP1 gene. J Virol. 2010;84(7):3339–50.
Tsou YA, Cheng YK, et al. Upper aerodigestive tract sequelae in severe enterovirus 71 infection: predictors and outcome. Int J Pediatr Otorhinolaryngol. 2008;72(1):41–7.
Tu PV, Thao NT, et al. Epidemiologic and virologic investigation of hand, foot, and mouth disease, southern Vietnam, 2005. Emerg Infect Dis. 2007;13(11):1733–41.
van der Sanden S, Koopmans M, et al. Epidemiology of enterovirus 71 in The Netherlands, 1963 to 2008. J Clin Microbiol. 2009;47(9):2826–33.
Van der Sanden S, Van der Avoort H, et al. Evolutionary trajectory of the VP1 gene of human enterovirus 71 genogroup B and C viruses. J Gen Virol. 2010;91:1949–58.
Wang J-R, Tuan Y-C, et al. Change of major genotype of enterovirus 71 in outbreaks of hand-foot-and-mouth disease in Taiwan between 1998 and 2000. J Clin Microbiol. 2002;40(1):10–5.
Wang JN, Yao CT, et al. Critical management in patients with severe enterovirus 71 infection. Pediatr Int. 2006;48(3):250–6.
Wang S-M, Lei H-Y, et al. Pathogenesis of enterovirus 71 brainstem encephalitis in pediatric patients: Roles of cytokines and cellular immune activation in patients with pulmonary edema. J Infect Dis. 2003;188(4):564–70.
Wang S-M, Liu C-C, et al. Clinical spectrum of enterovirus 71 infection in children in southern Taiwan, with an emphasis on neurological complications. Clin Infect Dis. 1999;29(1):184–90.
Wang S-Y, Lin T-L, et al. Early and rapid detection of enterovirus 71 infection by an IgM-capture ELISA. J Virol Methods. 2004;119(1):37–43.
Wang S, Lei H, et al. Modulation of cytokine production by intravenous immunoglobulin in patients with enterovirus 71-associated brainstem encephalitis. J Clin Virol. 2006;37(1):47–52.
Wang SM, Lei HY, et al. Therapeutic efficacy of milrinone in the management of enterovirus 71-induced pulmonary edema. Pediatr Pulmonol. 2005;39(3):219–23.
Wang SM, Lei HY, et al. Acute chemokine response in the blood and cerebrospinal fluid of children with enterovirus 71-associated brainstem encephalitis. J Infect Dis. 2008;198(7):1002–6.
Wang X, Peng W, et al. A sensor-adaptor mechanism for enterovirus uncoating from structures of EV71. Nat Struct Mol Biol. 2012;19(4):424–9.
Wang YF, Chou CT, et al. A mouse-adapted enterovirus 71 strain causes neurological disease in mice after oral infection. J Virol. 2004;78(15):7916–24.
Welch J, Maclaran K, et al. Frequency, viral loads, and serotype identification of enterovirus infections in Scottish blood donors. Transfusion. 2003;43(8):1060–6.
WHO. A guide to clinical management and public health response for Hand, Foot and Mouth Disease (HFMD, 2011). http://www.wpro.who.int/publications/docs/Guidanceforthe clinicalmanagementofHFMD.pdf. Accessed 6 Aug 2013.
Witso E, Palacios G, et al. Asymptomatic circulation of HEV71 in Norway. Virus Res. 2007;123(1):19–29.
Wong KT, Lum LC, et al. Enterovirus 71 infection and neurologic complications. N Engl J Med. 2000;342(5):356–8.
Wong KT, Munisamy B, et al. The distribution of inflammation and virus in human enterovirus 71 encephalomyelitis suggests possible viral spread by neural pathways. J Neuropathol Exp Neurol. 2008;67(2):162–9.
Wong KT, Ng KY, et al. Enterovirus 71 encephalomyelitis and Japanese encephalitis can be distinguished by topographic distribution of inflammation and specific intraneuronal detection of viral antigen and RNA. Neuropathol Appl Neurobiol. 2012;38(5):443–53.
Wu JM, Wang JN, et al. Cardiopulmonary manifestations of fulminant enterovirus 71 infection. Pediatrics. 2002;109(2).
Wu Y, Yeo A, et al. The largest outbreak of hand; foot and mouth disease in Singapore in 2008: The role of enterovirus 71 and coxsackievirus A strains. Int J Infect Dis. 2010;14(12):e1076–81.
Xiao XL, He YQ, et al. Simultaneous detection of human enterovirus 71 and coxsackievirus A16 in clinical specimens by multiplex real-time PCR with an internal amplification control. Arch Virol. 2009;154(1):121–5.
Xu F, Yan Q, et al. Performance of detecting IgM antibodies against enterovirus 71 for early diagnosis. PLoS One. 2010;5(6):e11388.
Xu J, Qian Y, Wang S, Serrano JM, Li W, Huang Z, Lu S. EV71: an emerging infectious disease vaccine target in the Far East? Vaccine. 2010;28(20):3516–21.
Yamayoshi S, Yamashita Y, et al. Scavenger receptor B2 is a cellular receptor for enterovirus 71. Nat Med. 2009;15(7):798–801.
Yang F, Ren L, et al. Enterovirus 71 outbreak in the People’s Republic of China in 2008. J Clin Microbiol. 2009;47(7):2351–2.
Yang J, Zhao N, et al. Association of interleukin 10 and interferon gamma gene polymorphisms with enterovirus 71 encephalitis in patients with hand, foot and mouth disease. Scand J Infect Dis. 2012;44(6):465–9.
Yang KD, Yang MY, et al. Altered cellular but not humoral reactions in children with complicated enterovirus 71 infections in Taiwan. J Infect Dis. 2001;183(6):850–6.
Yu H, Chen W, et al. Genetic analysis of the VP1 region of enterovirus 71 reveals the emergence of genotype A in central China in 2008. Virus Genes. 2010;41(41):1–4.
Zeng H, Wen F, Gan Y, Huang W. MRI and associated clinical characteristics of EV71-induced brainstem encephalitis in children with hand-foot-mouth disease. Neuroradiology. 2012;54(6):623–30. doi:10.1007/s00234-011-0979-3. Epub 2011 Dec 28.
Zhang D, Lu J, et al. Enterovirus 71 vaccine: close but still far. Int J Infect Dis. 2010;14(9):e739–43.
Zhang Y, Tan X-J, et al. An outbreak of hand, foot, and mouth disease associated with subgenotype C4 of human enterovirus 71 in Shandong, China. J Clin Virol. 2009;44(4):262–7.
Zhang Y, Zhu Z, et al. An emerging recombinant human enterovirus 71 responsible for the 2008 outbreak of Hand Foot and Mouth Disease in Fuyang city of China. Virol J. 2010;7(1):94.
Zheng X-Q, Chen X-Q, et al. Elevation of human leukocyte antigen-G expression is associated with the severe encephalitis associated with neurogenic pulmonary edema caused by Enterovirus 71. Clin Exp Med. 2013;1–7.
Zhu F-C, Meng F-Y, et al. Efficacy, safety, and immunology of an inactivated alum-adjuvant enterovirus 71 vaccine in children in China: a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2013;381(9882):2024–32.
Suggested Reading
Nishimura Y, Shimizu H. Cellular receptors for human enterovirus species A. Front Microbiol. 2012;3:105. doi:10.3389/fmicb.2012.00105.
Wong KT, Ng KY, et al. Enterovirus 71 encephalomyelitis and Japanese encephalitis can be distinguished by topographic distribution of inflammation and specific intraneuronal detection of viral antigen and RNA. Neuropathol Appl Neurobiol. 2012;38(5):443–53.
Wu K, Ng M, et al. Developments towards antiviral therapies against enterovirus 71. Drug Discov Today. 2010;15(23–24):1041–51.
Zhang D, Lu J, et al. Enterovirus 71 vaccine: close but still far. Int J Infect Dis. 2010;14(9):e739–43.
Acknowledgment
The authors are grateful to Associate Prof. David Perera, Institute of Health and Community Medicine, Universiti Malaysia Sarawak (UNIMAS), for his help in generating the phylogenetic tree of EV71 VP1 gene sequences (Fig. 12.1).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Ooi, M.H., Solomon, T. (2014). Enteroviruses: Enterovirus 71. In: Kaslow, R., Stanberry, L., Le Duc, J. (eds) Viral Infections of Humans. Springer, Boston, MA. https://doi.org/10.1007/978-1-4899-7448-8_12
Download citation
DOI: https://doi.org/10.1007/978-1-4899-7448-8_12
Published:
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4899-7447-1
Online ISBN: 978-1-4899-7448-8
eBook Packages: MedicineMedicine (R0)