Abstract
Since ancient times, seafood has been considered a healthy food for humans. Seafood by-products also play a major role in the daily life of humans. Focus has been increased to develop new seafood by-products from marine resources. In this opening chapter, we discuss fish-derived by-products such as proteins, peptides, and oil for nutriceutical and pharmaceutical uses. In addition, crustaceans and seaweed by-products and their applications are also discussed in detail. Overall, this book gives a good grounding in view of the processing, methods, and applications of seafood by-products.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
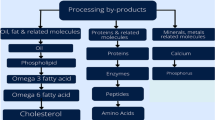
Since ancient times, seafood has been considered a healthy food for humans. Seafood by-products also play a major role in the daily lives of humans. Focus has been increased to develop new seafood by-products of marine origin. There could be environmental and economic advantages in utilizing the by-products of marine animals or plants for human consumption, rather than disposing of such materials at sea. Figure 1.1 Gives an overview of the utilization of marine by-products.
2 Fish By-products
Historically, fish by-products were considered to be of low value and were disposed of in the most convenient way. In many countries, importance is placed on the possibilities for further utilization of by-products, from aquaculture as well as traditional fisheries, rather than the problem of their disposal.
In the past, fish deaths, which can occur for a variety of reasons, have been disposed of in various ways, including by burning, discarding in the land, biogas production, production of fish meal, and incorporation into pet food or food for animals. Finfish and shellfish waste processing is confined to the manufacture of meal and oil. The meal is used mainly for animal feeds, while the oil is used in edible oils and oils for industrial applications. Recent studies have identified a number of biologically active compounds from fish muscle proteins, bones, fins, internal organs, collagen, gelatin, and oyster and crustaceans shells (Je et al. 2005; Jung et al. 2006; Kim and Mendis 2006; Kristinsson and Rasco 2000; Morimura et al. 2002; Nagai and Suzuki 2000; Ozawa and Suzuki 2002; Toppe et al. 2006).
2.1 Protein
A huge amount of protein-rich by-products from the fish processing industries is discarded every day (e.g., skin, bones, and fins). Recovery and alteration of the fish muscle proteins existing in the by-product materials and using them as functional ingredients in food systems is a very exciting and promising alternative. By developing enzyme technologies for protein recovery and modification, the production of a broad spectrum of food ingredients and industrial products will be possible. There are few hydrolyzed fish protein foods, with the exception of East Asian condiments and sauces. Fish protein hydrolysates using acid, base, endogenous enzymes, and added bacterial or digestive proteases are manufacturing techniques. The functional properties of fish protein hydrolysates are described in this book, including solubility, water-holding capacity, emulsification, and foam-forming ability (Fig. 1.2) (Kristinsson and Rasco 2000).
Discarded wastes from fish usually consist of a considerable amount of protein. These fish wastes are nutritionally important to isolate value-added products, Enzymatic and chemical methods are widely used to isolate the protein (Dyer et al. 1950; Shahidi et al. 1995). Fish hydrolysate is the main form of seafood by-products, which can be used as a fish-based fertilizer, animal food, and human food applications. Fish protein hydrolysates possess a number of biological activities, such as antiproliferative, antioxidant, and inhibitory activity of angiotensin-I-converting enzyme (ACE), as reported in the literature (Amarowicz and Shahidi 1997; Jun et al. 2004; Samaranayaka and Li-Chan 2008; Wu et al. 2003). Fish protein hydrolysates with multiple bioactivities could be useful in formulating functional food products that target the reduction of the symptoms of oxidative stress, hypertension, and possibly dyslipidemia, all of which are common to coronary heart disease (Samaranayaka et al. 2010).
2.2 Peptides
Peptides from marine sources have been proved to have significant antioxidant activity with no cytotoxicity (Kim et al. 2007; Liu et al. 2010; Qian et al. 2008; Rajapakse et al. 2005; Sampath Kumar et al. 2011). The in vitro antioxidant activity of bioactive peptides purified from different body parts of marine fishes have been explored in recent years (Naqash and Nazeer 2010, 2011). Several reviews have been completed related to fish peptides for antioxidant, antihypertensive, antiproliferative, and anticoagulant properties (Kim and Wijesekara 2010; Kim et al. 2007; Najafian and Babji 2012; Rajanbabu and Chen 2011). Fish peptides can be isolated using the following techniques: liquid extraction (Urakova et al. 2012) and microbial fermentation of proteins (Kim and Wijesekara 2010).
2.3 Collagen and Gelatin
Food and pharmaceutical industries all over the world are witnessing an increasing demand for collagen and gelatin. Mammalian gelatins (porcine and bovine) are the most popular and widely used. However, fish gelatin reportedly possesses similar characteristics to porcine gelatin, and may, thus, be considered as an alternative to mammalian gelatin for use in food products. The production and utilization of fish gelatin not only satisfies the needs of consumers, but also serves as a means to utilize some of the by-products of the fishing industry (Karim and Bhat 2009). The major protein constituent of the skin, bone, swim bladder, and scales of fishes resembles, in many ways, the more widely studied collagen of mammals (Eastoe 1957; Giraud-Guille et al. 2000). Type I collagen has also been extracted from the skin, bones, fins, and scales of freshwater and marine fishes.
Marine organisms such as fish and shellfish are rich sources of structurally diverse bioactive nitrogenous components. Based on the emerging evidence of potential health benefits, fish-derived peptides show significant promise as functional food ingredients. Bioactivities including antihypertensive, antioxidant, antimicrobial, anticoagulant, antidiabetic, anticancer, immunostimulatory, calcium binding, hypocholesteremic, and appetite suppression have been reported. Fish and shellfish waste components contain significant levels of high quality protein [10–23 % (w/w)], which represents a source for biofunctional peptide mining (Harnedy and FitzGerald 2012).
2.4 Fish Oil
On the basis of increasing global fish meal and fish oil costs, it is predicted that dietary fish meal and fish oil inclusion levels within compound aquafeeds will decrease in the long term, with fish meal and fish oil usage increasingly being targeted for use as a high value specialty feed ingredient for use within higher value starter, finisher, and broodstock feeds, and by so doing, extending the supply of these much sought after and limited feed ingredient commodities (Fig. 1.3) (Tacon and Metian 2008).
Fish oils are used in a variety of markets, including industrial uses, food, feed, and aquaculture and nutraceutical applications. The aquaculture market requires oils with low levels of oxidation, low levels of contaminants, and consistent quality. The nutraceutical market wants oils low in oxidation and contaminants, but also with high levels of omega-3 fatty acids. This market pays premiums for high levels of omega-3 and wild salmon oil might be able to command a premium in that market if only because of the name. However, the omega-3 content of salmon oil might present problems. Recently, the nutritional oil companies have revised their definition of omega-3 to include the sum of C18:3, C18:4, C20:4, C20:5, C21:5, C22:5, and C22:6 all n-3 fatty acids.
3 Crustaceans By-products
3.1 Chitin and Chitosan
Chitin is the second most important natural polymer in the world. The main sources exploited are two marine crustaceans, shrimp and crabs. Chitin, poly (β-(1 → 4)-N-acetyl-d-glucosamine), is a natural polysaccharide of major importance, first identified in 1884. This biopolymer is synthesized by an enormous number of living organisms, and considering the amount of chitin produced annually over the world, it is the most abundant polymer after cellulose. Chitin occurs in nature as ordered crystalline microfibrils forming structural components in the exoskeleton of arthropods or in the cell walls of fungi and yeast (Alishahi and Aïder 2012; Andres et al. 2007; Cheng et al. 2005; Gérente et al. 2010, 2007; Goosey and Kellner 2012; Kim et al. 2008; Krajewska 2005; Kwok and McKay 2010; No et al. 2007; Rinaudo 2006; Rong et al. 2010; Senevirathne and Kim 2012; Shahidi and Abuzaytoun 2005; Wang et al. 2011). Chitin and chitosan produced have also been characterized with Fourier transform infrared spectroscopy (FTIR) (Limam et al. 2011).
3.2 Application of Chitin and Chitosan
Chitin and its deacetylated derivative, chitosan, are non-toxic, antibacterial, biodegradable, and biocompatible biopolymers. Due to these properties, they are widely used for biomedical applications, such as tissue engineering scaffolds, drug delivery, wound dressings, separation membranes and antibacterial coatings, stent coatings, and sensors. Wound dressing is one of the most promising medical applications for chitin and chitosan. The adhesive nature of chitin and chitosan, together with their antifungal and bactericidal character, and their permeability to oxygen, is a very important property associated with the treatment of wounds and burns. Different derivatives of chitin and chitosan have been prepared for this purpose in the form of hydrogels, fibers, membranes, scaffolds, and sponges (Jayakumar et al. 2011). Electrospinning has been found to be a novel technique to produce chitin and chitosan nanofibers (Jayakumar et al. 2010a, b.
4 Seaweed By-products and Their Applications
Seaweeds are consumed by coastal populations, particularly Asians (Japan, China, Korea, Taiwan, Singapore, Thailand, Cambodia, Vietnam, Indonesia, and Malaysia). Seaweeds are also harvested or cultivated for the extraction of alginate, agar, and carrageenan, which are gelatinous substances (hydrocolloids). Alginates are commonly used in wound dressings, tissue engineering, and drug delivery (Li et al. 2005; Paul and Sharma 2004; Tønnesen and Karlsen 2002).
5 Organization of This Book’s Chapters
This book exposes knowledge on the view of processing, methods, and application of seafood by-products. The first part of the book includes an introduction to seafood by-products. The second part introduces statistics and methods involved in seafood by-products and illustrates trends in the use of seafood processing by-products in Europe and South Asia. The third part of the book explains in detail fisheries by-products, starting from protein, gelatin, soy sauce, enzymes, and lipids. The fourth part of the book deals with by-products of marine animals, bacteria, crustaceans, and algae. In this part, uses of seafood by-product in the animal seed industry, uses of lactic acid bacteria, chitosan products, chitosan from crustaceans, microalgae for biofuel production, and bioactive compounds from marine algae are explained. The fifth part of the book deals with the biological and biomedical applications, including the application of fish collagen in dental and hard tissue regeneration, food industry and human nutrition, antidiabetic and obesity effects of seafood by-products, bioluminescence, and bioactive marine algal and their therapeutics, and, finally, the functional properties of ascidians by-products for medicinal and nutritional value.
References
Alishahi A, Aïder M (2012) Applications of chitosan in the seafood industry and aquaculture: a review. Food Bioprocess Technol 5(3):817–830
Amarowicz R, Shahidi F (1997) Antioxidant activity of peptide fractions of capelin protein hydrolysates. Food Chem 58(4):355–359
Andres Y, Giraud L, Gerente C, Le Cloirec P (2007) Antibacterial effects of chitosan powder: mechanisms of action. Environ Technol 28(12):1357–1363
Cheng WP, Chi FH, Yu RF, Lee YC (2005) Using chitosan as a coagulant in recovery of organic matters from the mash and lauter wastewater of brewery. J Polym Environ 13(4):383–388
Dyer WJ, French HV, Snow JM (1950) Proteins in fish muscle. I. Extraction of protein fractions in fresh fish. J Fish Board Can 7(10):585–593
Eastoe JE (1957) The amino acid composition of fish collagen and gelatin. Biochem J 65(2):363
Gerente C, Lee VKC, Le Cloirec P, McKay G (2007) Application of chitosan for the removal of metals from wastewaters by adsorption—mechanisms and models review. Crit Rev Environ Sci Technol 37(1):41–127
Gérente C, Andrès Y, McKay G, Le Cloirec P (2010) Removal of arsenic(V) onto chitosan: from sorption mechanism explanation to dynamic water treatment process. Chem Eng J 158(3): 593–598
Giraud-Guille M-M, Besseau L, Chopin C, Durand P, Herbage D (2000) Structural aspects of fish skin collagen which forms ordered arrays via liquid crystalline states. Biomaterials 21(9):899–906
Goosey M, Kellner R (2012) Recovery of copper from PCB manufacturing effluent using chitin and chitosan. Circ World 38(1):16–20
Harnedy PA, FitzGerald RJ (2012) Bioactive peptides from marine processing waste and shellfish: a review. J Funct Food 4(1):6–24
Jayakumar R, Prabaharan M, Nair SV, Tamura H (2010a) Novel chitin and chitosan nanofibers in biomedical applications. Biotechnol Adv 28(1):142–150
Jayakumar R, Prabaharan M, Nair SV, Tokura S, Tamura H, Selvamurugan N (2010b) Novel carboxymethyl derivatives of chitin and chitosan materials and their biomedical applications. Prog Mater Sci 55(7):675–709
Jayakumar R, Prabaharan M, Sudheesh Kumar PT, Nair SV, Tamura H (2011) Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol Adv 29(3):322–337
Je J-Y, Park J-Y, Jung W-K, Park P-J, Kim S-K (2005) Isolation of angiotensin I converting enzyme (ACE) inhibitor from fermented oyster sauce, Crassostrea gigas. Food Chem 90(4):809–814
Jun S-Y, Park P-J, Jung W-K, Kim S-K (2004) Purification and characterization of an antioxidative peptide from enzymatic hydrolysate of yellowfin sole (Limanda aspera) frame protein. Eur Food Res Technol 219(1):20–26
Jung W-K, Lee B-J, Kim S-K (2006) Fish-bone peptide increases calcium solubility and bioavailability in ovariectomised rats. Br J Nutr 95(1):124–128
Karim AA, Bhat R (2009) Fish gelatin: properties, challenges, and prospects as an alternative to mammalian gelatins. Food Hydrocoll 23(3):563–576
Kim S-K, Mendis E (2006) Bioactive compounds from marine processing byproducts—a review. Food Res Int 39(4):383–393
Kim S-K, Wijesekara I (2010) Development and biological activities of marine-derived bioactive peptides: a review. J Funct Food 2(1):1–9
Kim S-Y, Je J-Y, Kim S-K (2007) Purification and characterization of antioxidant peptide from hoki (Johnius belengerii) frame protein by gastrointestinal digestion. J Nutr Biochem 18(1):31–38
Kim I-Y, Seo S-J, Moon H-S, Yoo M-K, Park I-Y, Kim B-C, Cho C-S (2008) Chitosan and its derivatives for tissue engineering applications. Biotechnol Adv 26(1):1–21
Krajewska B (2005) Membrane-based processes performed with use of chitin/chitosan materials. Sep Purif Technol 41(3):305–312
Kristinsson HG, Rasco BA (2000) Fish protein hydrolysates: production, biochemical, and functional properties. Crit Rev Food Sci Nutr 40(1):43–81
Kwok KCM, McKay G (2010) Novel batch reactor design for the adsorption of arsenate on chitosan. J Chem Technol Biotechnol 85(12):1561–1568
Li Z, Ramay HR, Hauch KD, Xiao D, Zhang M (2005) Chitosan–alginate hybrid scaffolds for bone tissue engineering. Biomaterials 26(18):3919–3928
Limam Z, Selmi S, Sadok S, El Abed A (2011) Extraction and characterization of chitin and chitosan from crustacean by-products: biological and physicochemical properties. Afr J Biotechnol 10(4):640–647
Liu Q, Kong B, Xiong YL, Xia X (2010) Antioxidant activity and functional properties of porcine plasma protein hydrolysate as influenced by the degree of hydrolysis. Food Chem 118(2):403–410
Morimura S, Nagata H, Uemura Y, Fahmi A, Shigematsu T, Kida K (2002) Development of an effective process for utilization of collagen from livestock and fish waste. Process Biochem 37(12):1403–1412
Nagai T, Suzuki N (2000) Preparation and characterization of several fish bone collagens. J Food Biochem 24(5):427–436
Najafian L, Babji AS (2012) A review of fish-derived antioxidant and antimicrobial peptides: their production, assessment, and applications. Peptides 33(1):178–185
Naqash SY, Nazeer RA (2010) Antioxidant activity of hydrolysates and peptide fractions of Nemipterus japonicus and Exocoetus volitans muscle. J Aquat Food Prod Technol 19(3–4):180–192
Naqash SY, Nazeer RA (2011) Evaluation of bioactive properties of peptide isolated from Exocoetus volitans backbone. Int J Food Sci Technol 46(1):37–43
No HK, Meyers SP, Prinyawiwatkul W, Xu Z (2007) Applications of chitosan for improvement of quality and shelf life of foods: a review. J Food Sci 72(5):R87–R100
Ozawa M, Suzuki S (2002) Microstructural development of natural hydroxyapatite originated from fish-bone waste through heat treatment. J Am Ceram Soc 85(5):1315–1317
Paul W, Sharma CP (2004) Chitosan and alginate wound dressings: a short review. Trends Biomater Artif Org 18(1):18–23
Qian Z-J, Jung W-K, Byun H-G, Kim S-K (2008) Protective effect of an antioxidative peptide purified from gastrointestinal digests of oyster, Crassostrea gigas against free radical induced DNA damage. Bioresour Technol 99(9):3365–3371
Rajanbabu V, Chen J-Y (2011) Applications of antimicrobial peptides from fish and perspectives for the future. Peptides 32(2):415–420
Rajapakse N, Mendis E, Byun H-G, Kim S-K (2005) Purification and in vitro antioxidative effects of giant squid muscle peptides on free radical-mediated oxidative systems. J Nutr Biochem 16(9):562–569
Rinaudo M (2006) Chitin and chitosan: properties and applications. Prog Polym Sci 31(7):603–632
Rong C, Qi L, Bang-zhong Y, Lan-lan Z (2010) Combined effect of ozonated water and chitosan on the shelf-life of Pacific oyster (Crassostrea gigas). Innov Food Sci Emerg Technol 11(1):108–112
Samaranayaka AGP, Li-Chan ECY (2008) Autolysis-assisted production of fish protein hydrolysates with antioxidant properties from Pacific hake (Merluccius productus). Food Chem 107(2):768–776
Samaranayaka AGP, Kitts DD, Li-Chan ECY (2010) Antioxidative and angiotensin-I-converting enzyme inhibitory potential of a Pacific hake (Merluccius productus) fish protein hydrolysate subjected to simulated gastrointestinal digestion and Caco-2 cell permeation. J Agric Food Chem 58(3):1535–1542
Sampath Kumar NS, Nazeer RA, Jaiganesh R (2011) Purification and biochemical characterization of antioxidant peptide from horse mackerel (Magalaspis cordyla) viscera protein. Peptides 32(7):1496–1501
Senevirathne M, Kim SK (2012) Utilization of seafood processing by-products: medicinal applications. Adv Food Nutr Res 65:495–512
Shahidi F, Abuzaytoun R (2005) Chitin, chitosan, and co-products: chemistry, production, applications, and health effects. Adv Food Nutr Res 49:93–135
Shahidi F, Han X-Q, Synowiecki J (1995) Production and characteristics of protein hydrolysates from capelin (Mallotus villosus). Food Chem 53(3):285–293
Tacon AG, Metian M (2008) Global overview on the use of fish meal and fish oil in industrially compounded aquafeeds: trends and future prospects. Aquaculture 285(1):146–158
Tønnesen HH, Karlsen J (2002) Alginate in drug delivery systems. Drug Dev Ind Pharm 28(6):621–630
Toppe J, Aksnes A, Hope B, Albrektsen S (2006) Inclusion of fish bone and crab by-products in diets for Atlantic cod, Gadus morhua. Aquaculture 253(1–4):636–645
Urakova IN, Pozharitskaya ON, Demchenko DV, Shikov AN, Makarov VG (2012) The biological activities of fish peptides and methods of their isolation. Russ J Mar Biol 38(6):417–422
Wang SL, Liang TW, Yen YH (2011) Bioconversion of chitin-containing wastes for the production of enzymes and bioactive materials. Carbohydr Polym 84(2):732–742
Wu H-C, Chen H-M, Shiau C-Y (2003) Free amino acids and peptides as related to antioxidant properties in protein hydrolysates of mackerel (Scomber austriasicus). Food Res Int 36(9–10):949–957
Acknowledgments
This research was supported by a grant from the Marine Bioprocess Research Center of the Marine Biotechnology Program funded by the Ministry of Oceans and Fisheries, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Kim, SK., Venkatesan, J. (2014). Introduction to Seafood Processing By-products. In: Kim, SK. (eds) Seafood Processing By-Products. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-9590-1_1
Download citation
DOI: https://doi.org/10.1007/978-1-4614-9590-1_1
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-9589-5
Online ISBN: 978-1-4614-9590-1
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)