Abstract
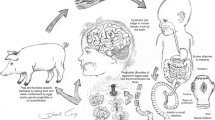
Neurocysticercosis, the most common parasitic brain disease worldwide, is due to the larvae infestation of Taenia solium. It is an endemic, neglected disease in poor countries with deprived sanitation, and is increasingly being reported in wealthy countries due to migration. Humans are the only definitive host of T. solium, while pigs are the intermediate hosts. Humans may become intermediate host by ingesting food or water contaminated by T. solium eggs.
Infection is associated with local and systemic immune-inflammatory responses modulated by the developmental stage of the parasite in the host (vesicular, colloidal. granular-nodular, and calcified stages) and by the central nervous system compartment where the parasites are located. Genetic diversity of cysticerci has been studied and the genome of T. solium is currently being sequenced.
The clinical manifestations are heterogeneous and depend mainly on the localization of cysts and immune response to the host. Seizures, headache, focal deficits and cognitive abnormalities are the most frequent manifestations. The prognosis is good; nevertheless, it may lead to long-term neurological sequels such as epilepsy and hydrocephalus.
Diagnosis is made mainly by neuroimaging, which is useful in the detection of evolutionary stage, number and localization of cysts. Immunological testing can be helpful; nonetheless, a negative test does not rule out the diagnosis. Treatment is complex and should be individualized, based on location and viability of the parasites. In most cases treatment is only symptomatic. Antihelminthic drugs are effective in approximately one-third of patients with parenchymal viable cysts. The most effective approach to Taeniasis/cysticercosis is prevention. This should be a primary public health focus for poor countries.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Seizures
- Epilepsy
- Imaging
- Albendazole
- Praziquantel
- Parasitic infection
- Taeniasis/cysticercosis
- Taenia solium
1 Introduction
Neurocysticercosis (NC), caused by the presence in the central nervous system (CNS) of the larval phase of Taenia solium is a neglected disease still endemic in most developing countries, mainly in the low-income ones, of Latin America, sub-Saharan Africa, the Indian subcontinent, and Southern Asia (Winkler et al. 2008; Blocher et al. 2011; Fleury et al. 2011). NC is currently an emergent disease in some high-income countries due to the increase of migration and tourism. It still causes high morbidity and mortality, the burden of which is difficult to state due to under-diagnosis (Bhattarai et al. 2012). The economic losses it entails are also important for the population and the society in general (Croker et al. 2012).
2 Life Cycle: Transmission
Cysticercosis is caused by ingestion of the eggs of T. solium shed in the feces of a human tapeworm carrier. Pigs, that are coprophages, become infected when they are reared under a free-range system, a condition still frequent in the rural communities of endemic countries, where disposal of human feces is deficient.
After ingestion, T. solium eggs hatch in the intestine, liberating motile oncospheres that invade the intestinal wall and migrate through the bloodstream to different tissues, where they develop into cysticerci. The parasite life cycle is completed when humans ingest undercooked pork containing cysticerci, resulting in human tapeworm infection. Cysts evaginate and attach to the small intestine by their scolex. Adult tapeworms develop, and can reside in the human small intestine for years.
Incidentally, infection of humans by cysticerci is possible mainly through the ingestion of food or water contaminated with T. solium eggs. Self-infection can also occur, mainly by orofaecal contamination and possibly by reverse peristalsis. In humans, at least in Latin America, the preferential localization of cysticerci is the central nervous system (CNS).
Infection may be prevented by proper disposal of human feces around pigs, use of latrines, and keeping pigs indoors (Goodman et al. 1999). In the case of infected pork meat, this must be cut into slices of 5 cm or less in thickness, and fried for at least 1 h or boiled for 2 or 3 h (Aluja et al. 1987). Freezing of the meat before consumption at −5 °C for 4 days or at −15 °C for 3 days is also efficient (Sotelo and Marin 1987).
3 Causative Agent
Adult T. solium, like other species of the gender Taenia is a plane worm. Its head (scolex) consists of four suckers and a rostellum with two rows of hooks, which are essential to fix the parasite in the human small intestine. The head thins to form a neck from which the proglottids are produced. These form a chain, named strobila (1.5–5 m in length). The proglottids nearest to the neck are immature, while those located at the end of the strobila are differentiated and contain about 50,000 eggs. After 2 or 3 months of infection, 4 or 5 gravid proglottids are released every day in the host’s faeces. Eggs contained in each proglottid are at different grades of maturation; almost 50 % of them contain an infective completely developed oncosphere. The immature ones can mature out of the host and can persist for weeks, viable and infective, in water, soil, and vegetation. Eggs are round and measure around 20–40 μm. They are morphologically indistinguishable from the eggs of other Taeniae and are protected from the environment by a rigid structure called the embryophore (Laclette et al. 1982). In the larval phase, there are two morphologically distinct types of cysticerci (Rabiela et al. 1989). The most common one is the “cellulosae”, a small vesicle (0.5–2 cm in diameter) containing a small invaginated scolex similar to the scolex of the adult taenia. This form of cysticerci is found in muscle, subcutaneous tissue, and cerebral parenchyma. The “racemosus” form constitutes a hydropic change that leads to a large parasite structure composed of a conglomeration of vesicles of different sizes. It has no evident scolex and is mainly located in the cerebral ventricles and in the basal subarachnoid space of the CNS. As cestodes lack a digestive track, they obtain their nutrients and excrete their waste through their tegument surface by absorption and diffusion.
Four developmental phases have been described in the life cycle of cysticerci (Escobar 1983). In the first one (vesicular phase), the parasite is alive, and the cyst contains the invaginated larva of 4–5 mm that lies in a transparent liquid (Figs. 1 and 4a). In the following one, (colloidal phase, Figs. 2 and 4b), the cyst is adherent and surrounded by a connective capsule. The content of the vesicle loses fluidity and becomes milky. In the third stage (nodular granular phase), the size of the cyst has diminished, and its membrane cannot be easily identified as it is completely attached to the collagen capsule. Finally, in the last stage (calcified phase, Figs. 3 and 5b) a hard, completely calcified nodule is identified, reduced to less than half of its original size. In adjacent tissue, astrogliosis and low-grade inflammatory reaction are present. It is interesting to note that this temporal sequence varies according to the localization of the parasites. In particular, when parasites are in the cisterns of the subarachnoid space or in the ventricular system, the vesicular phase is much longer than in other locations, facilitating parasite growth. It is also interesting to note that calcification of parasites in these locations is extremely rare.
Genetic diversity of cysticerci collected in pork has been investigated. The comparison of parasites from different continents shows significant genetic differentiation, suggesting diverse evolutionary paths (Vega et al. 2003; Maravilla et al. 2003). On the other hand, when cysticerci of the nearest geographical regions or from different tissues of the same pig were studied, DNA diversity was within the range expected from a recombination process (Vega et al. 2003; Bobes et al. 2010).
The genome of T. solium is currently being sequenced in Mexico and only partial results have been published at the time of this review. The information generated will provide a powerful resource for the study of taeniasis/cysticercosis helping to address fundamental questions such as the molecular basis of host-parasite specificity and mechanisms of parasite pathogenesis, among others (Aguilar-Díaz et al. 2006). Genome size has been estimated to be between 251 and 270 Mb and only about 24 % of the sequences are homologous to mammalian genes, in particular humans. The sequences lacking homologues in humans are strong candidates for investigation of treatment, diagnosis and vaccination (Aguilar-Díaz et al. 2006).
4 Immunological Response
In their larval phase, cysticerci express a complex and diverse set of antigens. A few of them are specific of T. solium, but many others cross-react with those of other helminths. Some of them are present in all the phases of the parasites, while others are phase-specific. Most patients have detectable antibodies, generally of the IgG class, against parasites in serum and cerebrospinal fluid (CSF). They can also be detected in the saliva (Bueno et al. 2000), The four subclasses of IgG are increased in symptomatic NC patients compared to the asymptomatic patients. The relevance of antibodies to parasite damage has not been extensively studied. Although some observations indicate that antibodies can have a protective action, the demonstration of a net beneficial effect is still lacking.
Characteristics of the local inflammatory reaction associated with the presence of the parasite depend on the CNS compartment where the parasites are located. When located in the parenchyma or in the subarachnoid space, inflammation is confined around the parasite. In these cases, the CSF is in general normal and immunohistochemical studies of the inflammatory infiltrate around the cysticerci reveals the presence of specific IgM and plasma cells, natural killer (NK) lymphocytes, macrophages, granulocytes, and T cells. Intensity of the inflammatory reaction varies, based upon parasite evolutionary phase, being almost absent in the vesicular phase and intense in the colloidal one (Alvarez et al. 2002). When the parasites are located in the basal subarachnoid space or in the ventricular system, inflammation is mainly seen in the CSF with the presence of lymphocytic pleocytosis, mild elevation of proteins, and hypoglycorrhachia, with increased levels of all IgG subclasses as well as interleukin (IL) -6, -5, and -10 (Chavarría et al. 2005). This reaction can affect the leptomeninges with the formation of a dense exudate containing collagen fibers, lymphocytes, and multinucleated giant cells resulting in meningeal thickening as well as occlusion of Luschka and Magendie foramina that can lead to chronic hydrocephalus (Escobar 1983). In these cases, inflammation can cause damage in vascular and nervous structures remote from the parasites.
Systemic immunological changes are also observed. In the blood of patients with inflammatory parasites located in the subarachnoid space or in the ventricular system, a decreased proliferation of specific cells without production of cytokines, and an increase of the proportion of T regulatory cells, significantly correlated with the increase of these cells in CSF, have been observed (Chavarria et al. 2006; Adalid-Peralta et al. 2012). On the other hand, patients with asymptomatic calcified NC present a specific proliferative reaction, with production of cytokines predominantly of the Th2 type (IL-4, IL-5, IL-13, IL-12) and a higher level of specific IgG4, compared to persons exposed to parasites but non-infected (Chavarría et al. 2003).
Parasites have developed different protective mechanisms to evade the immune surveillance of the host. Particularly, antigen B, the most frequent antigen recognized by the serum of patients, can bind factor C1q from the complement system, a property that can prevent the potential toxicity of antibody-mediated parasite damage (Laclette et al. 1992). The presence of a great amount of immunoglobulin on the surface of the cysticercus can mask its presence in the immunological system (Flisser et al. 1986).
5 Clinical Manifestations
One of the most intriguing aspects of NC is that presumably a high percentage of the individuals harboring NC remain asymptomatic. Among the symptomatic group, clinical manifestations of NC are determined mainly by the evolutive phase and location of the parasite within the CNS, as well as by the intensity of the immunological response of the patient (Loureiro das Chagas et al. 2003; Patel et al. 2006). The combination of these factors, among others, explains the great heterogeneity and the absence of a specific clinical picture. A systematic review was conducted to estimate the clinical manifestation frequencies of symptomatic NC (Carabin et al. 2011). Among NC patients seen in neurology clinics, about 79 % had seizures/epilepsy, 38 % severe headaches, 16 % focal deficits, and 12 % signs of increased intracranial pressure. Several other symptoms were also reported in less than 10 % of the patients.
5.1 Parenchymal Neurocysticercosis
The most common clinical manifestation of parenchymal NC is epileptic seizure, which occurs in 60–90 % of cases. The symptomatology of altered mental state and psychiatric manifestations remains poorly described in the literature (Carabin et al. 2011). In two studies (Forlenza et al. 1997; Carpio et al. 2008) which provided definitions of clinical manifestations, depression was reported in about 53 % and 15 % of the patients, respectively. A recent study reports a spectrum of cognitive abnormalities, including dementia (Rodrigues et al. 2012).
5.1.1 Neurocysticercosis and Epilepsy
Seizures associated with NC may be categorized as either acute symptomatic (Carpio et al. 1998; Murthy and Yangala 1999) or as unprovoked, remote symptomatic (epilepsy, if recurrent). This differentiation is very important, due to its implications concerning treatment and prognosis. The classification of seizure types in patients with NC varied among studies (Cukiert et al. 1994). It seems that either generalized seizures or partial seizures with secondary generalization are most commonly reported, while complex partial seizures are less frequent (Carpio et al. 2013). Seizures may occur at any evolutionary phase of the parasite. In a recent prospective cohort study (Kelvin et al. 2011), transitional cysts have been found to be associated with a significantly higher probability of seizure in the univariate analysis. However, this association was lost after adjusting for patient age and gender as well as for number and location of the cysts. Patients with cysts in the parietal and frontal lobes were also more likely to present seizures.
5.1.2 Epileptogenesis and Neurocysticercosis
So far, the mechanism by which the calcified neurocysticercal lesions (CNL) cause seizures or epilepsy is not known (Antoniuk et al. 2001; Carpio et al. 2013; Rathore et al. 2012). This has been attributed to residual perilesional gliosis that results in chronic epileptogenic foci (Leite et al. 2000). CNL are frequently encountered in computed tomography (CT) scans of asymptomatic individuals, and studies from Latin American countries report that the majority are incidental lesions (Fleury et al. 2010; Leite et al. 2000; Kowacs et al. 2006). These observations would question the epileptogenicity of CNL.
Edema associated with CNL has also been suggested to be implicated in epileptogenicity. In fact, episodic appearance of edema surrounding the CNL after seizures has been described (Nash et al. 2008). In this study, the authors argue that episodic release of cysticercal antigens from the calcified lesions can lead to inflammation, perilesional edema, and seizures. However, it is not clear whether this edema is the cause or the consequence of seizure (Leite et al. 2000).
5.1.3 Neurocysticercosis as Etiology of Epilepsy
Although it is clear that epilepsy is the main symptom of NC, the relevance of NC as a generator of epilepsy is debated. The percentage of patients with NC in epileptic patients of endemic countries varies among studies from 14 to 54 % (Ndimubanzi et al. 2010). It is interesting, however, to note that some observations may question the causal relationship between NC and epilepsy (Sakamoto et al. 1999). Parasite location may be remote from the apparent epileptogenic region, so that there is no correlation between the NC burden of lesions and the severity of epilepsy; patients with severe refractory seizures may have only one calcified lesion, while, on the other hand, there are patients with multiple cysts or calcifications but no seizures (Carpio et al. 1998; Ferreira et al. 2002).
It is important to note that both NC and epilepsy are common diseases in most developing countries, suggesting both causal and fortuitous relationships between these pathological conditions (Sakamoto et al. 1999; Terra-Bustamante et al. 2005). In particular, in cross-sectional studies investigating the etiology of intractable epilepsy in Brazil, NC turned out to mostly represent a coexistent pathology (Pal et al. 2000; Velasco et al. 2006).
5.1.4 Risk of Seizure Recurrence in Patients with Neurocysticercosis
Some studies have reported that NC patients with acute symptomatic seizures have a good prognosis in terms of remission of seizures (Carpio and Hauser 2009; Singhi et al. 2000; Manreza 2000; Ferreira et al. 2001; Goel et al. 2010). Other studies have reported that most patients have a high risk of seizure recurrence, and suggest that prognosis improves after antihelminthic treatment (Garcia et al. 2004). Prospective cohort studies have determined a risk between 17 and 56 % of seizure recurrence after a first seizure due to NC, depending on the viability of the parasite. The risk is greater in the transitional forms and diminishes in the viable or calcified forms (Carpio and Hauser 2002; De Souza et al. 2009; Sharma et al. 2011; Thussu et al. 2008).
5.2 Extraparenchymal Neurocysticercosis (Basal Subarachnoid Space and Ventricular System)
At these locations (around 15–30 % of cases) headache and signs of elevated intracranial pressure due to the obstruction of CSF circulation are the most frequent symptoms. These occur in 88 % of the cases, in comparison with 10 % of cases with parenchymal location (Carpio et al. 1994). Inflammation of meninges can also generate cranial nerve dysfunction, chiasmatic syndrome, and cerebral infarcts secondary to vasculitis (Agapejev et al. 2007; Cárdenas et al. 2010a, b). When hydrocephalus is present (Fig. 5a), the mortality rate is high (50 %), and most patients die within 2 years after CSF shunting (Cárdenas et al. 2010a, b; Sotelo et al. 1988). This is why ventricular and basal cisternal locations are considered to be malignant forms of NC (Estañol et al. 1986).
Spinal cord cysticercosis is rare. Patients experience nonspecific clinical manifestations, such as nerve root pain or spinal cord compression syndromes, according to the level of the lesion (Alsina et al. 2002). Severe forms of NC may exceptionally occur, including cysticercotic encephalitis, and result in permanent neurological sequels, such as amaurosis.
6 Host and Parasite Factors Modulating Clinical Presentation
6.1 Age
In addition to being less common in children, NC clinical manifestations clearly vary in different age groups (Kelvin et al. 2009a, b; Rosenfeld et al. 1996). Most cases of childhood NC present mild to moderate symptomatology and single lesions (Ruiz-Garcia et al. 1997; Kelvin et al. 2011). A study specifically carried out to compare the clinical manifestations between pediatric and adult NC patients (Sáenz et al. 2006) has reported statistically significant differences: seizures were more frequent in children (80.4 % vs. 56.1 %) and intracranial hypertension and headaches were more frequent in adults (27.2 % vs. 15.2 % and 35.1 % vs. 21.7 %, respectively). Although these age-related differences seem clear, a single effect of age is difficult to demonstrate, since various confounding factors are probably involved (Kelvin et al. 2009a, b).
Most paediatric cases show a single enhancing lesion, also named solitary cysticercus granuloma (Singh et al. 2010). This lesion is a common finding in patients with newly identified seizures in developing countries. These patients have some benign and transitory clinical manifestations, predominantly partial or partial secondary generalized seizures. Single enhancing lesions tend to resolve spontaneously, without anticysticercal treatment or surgery, since the parasite is already in the degenerative phase and will eventually disappear or become calcified.
6.2 Gender
Inflammation surrounding parenchymal cysticerci is more intense in women (Kelvin et al. 2009a, b), and multiple degenerating parasites localized in the CNS parenchyma are also more frequently reported in young women. Regardless of the localization of the parasite, the inflammatory response, as expressed by CSF cellularity, is more intense in women (Fleury et al. 2010). There are significant gender and age differences in the local immune response (Kelvin et al. 2009a, b). It has been suggested that both age and gender influence the strength of the host’s immune response. The odds of having transitional cysts are higher for female patients than for males (Kelvin et al. 2011).
6.3 Genes
Clinical heterogeneity across geographical areas is well documented. Most cases from the Indian subcontinent present single degenerative lesions, whereas those from Latin America present few viable cysts (Singh et al. 2010). These differences are probably due to complex interactions between the host, parasite, and environmental factors (Singh 1997; Fleury et al. 2010). Genetic differences in T. solium cysticerci have been reported from different countries (Maravilla et al. 2008; Vega et al. 2003) and may contribute to clinical variations among countries. Genetic susceptibility to NC has been suggested on the basis of positive association of HLA-DRBII 13 with single, contrast-enhancing CT lesions (Jain et al. 1999). However, neither familial aggregation of seizures in first degree relatives of NC patients with seizures (Kelvin et al. 2009a, b) nor significant aggregation of NC cases in families have been found (Fleury et al. 2006), arguing in favor of the involvement of multiple genes.
7 Diagnosis
Diagnosis of NC cannot rely only on clinical grounds, since there are no specific clinical manifestations of NC.
7.1 Neuroimaging
Diagnosis of NC is mainly made by neuroimaging. MRI is more sensitive than CT for the diagnosis of viable and degenerating cysticerci, since it improves recognition of the perilesional edema and degenerative changes of the parasite, as well as of cysts located inside the ventricles or the subarachnoid space. However, CT is more sensitive than MRI for the detection of calcifications.
CT or MRI can identify the four developmental phases of cysticerci when located in the brain parenchyma. In the vesicular phase, the CT scan depicts circumscribed, round, hypodense areas, varying in size and number, without enhancement by contrast media (Zee et al. 2000). In the MRI, the vesicular larva appears with a CSF-like intensity signal on all sequences, with no surrounding high signal on T2-weighted images (Lucato et al. 2007; Mont’Alverne Filho et al. 2011). Both MRI and CT may show a high intensity or hyperdense, 2–3 mm mural nodule depicting the scolex, within some vesicular cysts (Fig. 4a). MRI sequences such as axial fluid attenuated inversion recovery (FLAIR) detect a significantly higher number of scolices than other sequences, which is helpful for improving the diagnosis of NC (Lucato et al. 2007). As the cyst degenerates, the contrast-enhanced CT scan shows an annular (colloidal phase) or nodular (nodular phase) enhancement surrounded by irregular perilesional edema (Fig. 4b). In this phase, the fluid content gives a slightly higher signal than CSF and is sometimes isodense with the parenchyma on MRI-T1 and/or proton density-weighted, and high signal on T2 images. The capsule shows a higher signal than the adjacent brain tissue, with thick ring enhancement on T1 images, while on T2 images there is a low ring signal surrounded by high signal lesion, due mostly to edema (Dumas et al. 1997; Zee et al. 2000). When the cyst dies it may disappear or become an inactive calcified nodule with homogeneous high density on CT or low intensity on proton-weighted MRI (Fig. 5b).
When the parasites are located in the subarachnoid space or within the ventricular system, recognition of parasites with MRI is more difficult, as parasites emit an intensity signal similar to that of the CSF, generally do not enhance after intravenous administration of contrast, and commonly lack a scolex. Thus, often only indirect signs of the presence of the parasite are available, such as the unilateral enlargement of the basal cistern (Lucato et al. 2007) (Fig. 6). Specific MRI sequences including diffusion-weighted MRI magnetization transfer ratio (MTR), 3D constructive interference in steady state (3DCISS), fast imaging employing steady state acquisition sequences (FIESTA; Fig. 6a), and FLAIR sequences have proven to be more sensitive tools to visualize the cyst wall (Braga et al. 2004; Fleury et al. 2011). In case of meningeal inflammatory process, gadolinium enhancement of MRI or contrast-enhanced CT may depict leptomeningeal thickening (Kioumehr et al. 1995) and hydrocephalus (Fig. 5a).
7.2 Immunodiagnosis
No optimal immunological test for NC diagnosis is yet available. The difficulties of developing a sensitive and specific immunological test for NC diagnosis are mainly the result of the proper characteristics of the disease. An immunological test useful for medical practice must be specific in terms of CNS localization and should differentiate between viable and non-viable forms of the parasite.
Different immunological tests have been developed. The most widely used tests aim at the detection of specific antibodies. In these cases, different types of antigens have been used: crude antigens or partially purified antigenic extracts of T. solium or of the related parasites T. crassiceps or T. saginata, and recombinant or synthetic proteins. Techniques have also evolved, from complement fixation test, indirect hemagglutination, to the enzyme-linked immunosorbent assay (ELISA) and enzyme-linked immunoelectrotransfer blot (EITB) assay (Tsang et al. 1989; Brandt et al. 1992), which are the two main techniques currently in use. Also, detection of antigens by monoclonal or polyclonal antibodies using the ELISA technique has been developed (Brandt et al. 1992).
Comparisons of these tests have given divergent results, in part due to differences in methodology between studies. Despite these sources of variations, it seems that EITB in sera has higher sensibility and specificity than ELISA to detect antibodies, at least when carried out in non-endemic areas (Ramos-Kuri et al. 1992). It is also clear that sensitivity of both tests falls in cases with a single parasite, when parasites are located in the brain parenchyma or are calcified (Wilson et al. 1991; Singh et al. 1999). In CSF, performance of Ab-ELISA and EITB seems to be quite similar (Proaño-Narvaez et al. 2002; Michelet et al. 2011). Antigen detection in sera or CSF is highly specific for detecting viable extra-parenchymal parasites (Fleury et al. 2007). Another important point is the feasibility of carrying out ELISA and EITB techniques in endemic countries. In fact, while ELISA can be accomplished in 20 h and costs around $2.00 per sample, EITB assay requires almost 2 weeks, sophisticated equipment, highly skilled personnel, and its cost is up to ten times greater than ELISA (Proaño-Narvaez et al. 2002).
Despite the current immunological and imaging advances, the diagnosis of NC is still a challenge in many patients. Del Brutto et al. (2001) proposed diagnostic criteria for NC based on clinical, imaging, immunological and epidemiological features. This proposal, though not validated so far, may be useful to identify patients with parenchymal, but not extraparenchymal, forms of NC (Machado 2010). Such diagnostic criteria should be revised to incorporate current scientific knowledge, in order to achieve a new consensus on the diagnosis of NC.
8 Treatment
The treatment of NC should be individualized, based on the pathogenesis and natural history of the disease in each patient. Therapy in most cases is limited to symptomatic treatment with anti-seizure medication (ASM) for patients with seizures. Regarding duration of the ASM following an acute NC episode, some clinicians routinely continue ASM for 1 year, but shorter or longer intervals have also been recommended (Carpio et al. 2013; Takayanagui et al. 2011). It is assumed that the risk of seizures is substantial as long as there is an active ongoing process as characterized by persistence of edema around the degenerating lesion. Because of this, CT scan is a useful tool for these treatment decisions. It is appropriate to monitor cyst activity with CT scanning or MRI and to continue ASM until resolution of the acute lesion (Carpio et al. 2013). Seizures occurring in individuals after resolution of edema and reabsorption or calcification of the degenerating cyst should be considered unprovoked; in this situation, long-term ASM is warranted (Fig. 7).
Mannitol or oral glycerol is used if high intracranial pressure is a feature; analgesics should be given for headache. Corticosteroids are often administered for NC, on the premise that they reduce inflammation and edema around dying parenchymal cysts, and are also recommended for treatment of large subarachnoid cysts and arachnoiditis. However, the doses, duration, form and, importantly, timing of administration of corticosteroids are not clear. Alternative non-hormonal anti-inflammatory agents are dextrochlorpheniramine, ketoprofen, and immunosuppressants such as azathioprine and methotrexate. The efficacy of these drugs in NC has not, however, been widely confirmed.
Surgery is currently restricted mainly to placement of ventricular shunts for hydrocephalus and, in occasional cases of accessible racemose subarachnoid cysts and intraventricular cysts, mainly by endoscopic approach (Goel et al. 2008; Proano et al. 2009; Torres-Corzo et al. 2010). Transitional or degenerative cysts, regardless of their size or location should not be biopsied or removed if differential diagnosis has been discarded, since the parasite is dead and will disappear or become calcified spontaneously (Singh et al. 2010).
Treatment for NC with antihelminthic drugs (AHD) has been available for at least 25 years, but their use has always been controversial. Praziquantel (PZQ) was used for the first time in México (Robles and Chavarrıa 1979) and albendazole (ALB) was used for the first time in China (Xiao et al. 1986) to cure NC. PZQ is an acylated isoquinole-pyrazine with broad antihelminthic activity. Its mechanism of action is not fully understood, but it is assumed that PZQ changes metabolism and intracellular calcium, with the main side effect of inhibition of muscle movements (Garcia-Dominguez et al. 1991). ALB is a benzimidazole with a broad antihelminthic spectrum, whose anticysticercal effect is by inhibition of glucose uptake by parasitic membranes, causing energy depletion (Lacey 1990). To date there are no controlled clinical trials to establish definitive doses and duration of treatment. The most frequent treatment scheme for PZQ is 50 mg/kg/day for 15 days and for ALB, 15 mg/kg/day for 8 days (Carpio et al. 2013).
A meta-analysis of treatment of NC reported disappearance of viable parenchymal cysts in 44 % of patients who were treated with ALB vs. 19 % of the placebo group. In contrast, disappearance of degenerative cysts in 72 % of patients of the ALB group and in 63 % of the placebo group was not statistically significant (Del Brutto et al. 2006). An Editorial comment of this meta-analysis (Del Brutto et al. 2006) stated that selected studies were small and heterogeneous, and only 5 of 11 were of good quality. The Editorial comment concluded that studies provided limited evidence of a modest efficacy of NC treatment. A recent Cochrane review (Abba et al. 2010) concluded that in adults with viable cysts the use of ALB is associated with a decrease in the number of cysts, but with no difference in seizure recurrence between ALB and no treatment.
Regarding extraparenchymal cysts, the management is even more unclear. Although AHD have demonstrated efficacy in some cases, it is also clear that not all the cases respond to current treatment (Das et al. 2007; Carpio et al. 2008; Cárdenas et al. 2010a, b). This is why it is a priority to search for new treatment alternatives (Jung-Cook 2012; Diazgranados-Sánchez et al. 2008).
9 Eradication of the Disease
Cysticercosis has been considered a neglected “tools-ready disease” (WHO 2007) and a potentially eradicable disease (Task Force 1993). Simple approaches, such as interrupting the parasite´s life cycle by placing fences to avoid the contact of pigs with human feces eventually contaminated with T. solium eggs could eradicate the disease. Some other strategies have been proposed and tested: massive AHD treatment of humans to reduce the number of tapeworm carriers (Allan et al. 1997; Sarti et al. 2000), health education programs, improvement of pig-management and sanitary conditions (Ngowi et al. 2008; Sarti et al. 2000), treatment of infected pigs (Sikasunge et al. 2008) and vaccination of rural pigs (Huerta et al. 2001; Sciutto et al. 2007; Gonzalez et al. 2001). Although almost all these strategies have shown relative efficacy, they have been evaluated only in studies on small cohorts and during limited periods of time. It is relevant to mention that a pilot control program in the poorest states of Mexico is ongoing, based on health and sanitary education associated with vaccination of pigs, and the preliminary results are encouraging (De Aluja et al. 2012).
The main and urgent strategy to be carried out for NC is the implementation of specific national and international health policies. Also, since pathogen transmission does not respect borders, the implementation of multinational and regional networks is indispensable to fight against this parasitic disease. Currently, networks of specialists have been organized in Africa, Asia, Europe and Latin America. These efforts must be encouraged, but as eradication depends mainly on social, economic and political factors, their real impact will depend on convincing national governments to make of this objective a public health priority.
10 Conclusions and Future Perspectives
NC is still endemic in countries with poor sanitation, and is being increasingly reported in high income countries due to migration and tourism. The morbidity and mortality caused by this preventable disease are unacceptable in the twenty-first century. As shown in this chapter, many questions are now solved, although problems related to immunology, diagnosis and treatment are still open. One of the reasons of the slow progress is that this disease occurs mainly in low income countries, where diagnosis tools are not available to all the population and where only few researchers are interested in the problem. Also, the epidemiologic transition, present in most of these countries, boosts the authorities to disregard it and to focus efforts to diseases of the “first world”. Multidisciplinary research is needed, mainly to identify sensitive and specific diagnostic tools of low cost, to investigate the factors involved in the lack of response to treatment of some patients and to propose new therapeutic compounds. Knowledge of the T. solium genome will certainly represent an important step forward for many of these issues, inspiring optimism. In addition, close communication with local authorities is indispensable regarding prevention, because control programs will be successful only if they are involved in their application.
References
(1993) Recommendations of the International Task Force for Disease Eradication. MMWR Recomm Rep 42:1–38
Abba K, Ramaratnam S, Ranganathan LN (2010) Antihelmintics for people with neurocysticercosis. Cochrane Database Syst Rev 3, CD000215
Adalid-Peralta L, Fleury A, García-Ibarra TM, Hernández M, Parkhouse M, Crispín JC, Voltaire-Proaño J, Cárdenas G, Fragoso G, Sciutto E (2012) Human neurocysticercosis: in vivo expansion of peripheral regulatory T cells and their recruitment in the central nervous system. J Parasitol 98:142–148
Agapejev S, Pouza AF, Bazan R, Faleiros AT (2007) Clinical and evolutive aspects of hydrocephalus in neurocysticercosis. Arq Neuropsiquiatr 65:674–680
Aguilar-Díaz H, Bobes RJ, Carrero JC et al (2006) The genome project of Taenia solium. Parasitol Int 55(Suppl):S127–S130
Allan JC, Velasquez-Tohom M, Fletes C, Torres-Alvarez R, Lopez-Virula G, Yurrita P, Soto de Alfaro H, Rivera A, Garcia-Noval J (1997) Mass chemotherapy for intestinal Taenia solium infection: effect on prevalence in humans and pigs. Trans R Soc Trop Med Hyg 91:595–598
Alsina GA, Johnson JP, McBride DQ, Rhoten PRL, Mehringer CM, Stokes JK (2002) Spinal neurocysticercosis. Neurosurg Focus 12:1–7
Aluja A, Escobar A, Escobedo F, Flisser A et al (1987) Cisticercosis: una recopilación actualizada de los conocimientos básicos para el manejo y control de la cisticercosis causada por Taenia solium. Fondo de Cultura Económica, Biblioteca de la salud, México
Alvarez JI, Colegial CH, Castaño CA et al (2002) The human nervous tissue in proximity to granulomatous lesions induced by Taenia solium metacestodes displays an active response. J Neuroimmunol 127:139–144
Antoniuk SA, Bruck I, Dos Santos LH et al (2001) Seizures associated with calcifications and edema in neurocysticercosis. Pediatr Neurol 25:309–311
Bhattarai R, Budke CM, Carabin H et al (2012) Estimating the non-monetary burden of neurocysticercosis in Mexico. PLoS Negl Trop Dis 6:e1521
Blocher J, Schmutzhard E, Wilkins PP, Gupton PN, Schaffert M, Auer H, Gotwald T, Matuja W, Winkler AS (2011) A cross-sectional study of people with epilepsy and neurocysticercosis in Tanzania: clinical characteristics and diagnostic approaches. PLoS Negl Trop Dis 5:e1185
Bobes RJ, Fragoso G, Reyes-Montes M del R, Duarte-Escalante E, Vega R, de Aluja AS, Zúñiga G, Morales J, Larralde C, Sciutto E (2010) Genetic diversity of Taenia solium cysticerci from naturally infected pigs of central Mexico. Vet Parasitol 168:130–135
Braga F, Rocha AJ, Gomes HR, Filho GH, Silva CJ, Fonseca RB (2004) Noninvasive MR cisternography with fluid-attenuated inversion recovery and 100% Supplemental O2 in the evaluation of neurocysticercosis. AJNR Am J Neuroradiol 25:295–297
Brandt JR, Geerts S, De Deken R et al (1992) A monoclonal antibody-based ELISA for the detection of circulating excretory-secretory antigens in Taenia saginata cysticercosis. Int J Parasitol 22:471–477
Bueno EC, Vaz AJ, Machado LD, Livramento JA (2000) Neurocysticercosis: detection of IgG, IgA and IgE antibodies in cerebrospinal fluid, serum and saliva samples by ELISA with Taenia solium and Taenia crassiceps antigens. Arq Neuropsiquiatr 58:18–24
Carabin H, Ndimubanzi PC, Budke CM, Nguyen H, Qian Y, Cowan LD, Stoner JA, Rainwater E, Dickey M (2011) Clinical manifestations associated with neurocysticercosis: a systematic review. PLoS Negl Trop Dis 5:e1152
Cárdenas G, Carrillo-Meza R, Jung H et al (2010a) Subarachnoidal neurocysticercosis non-responsive to cysticidal drugs: a case series. BMC Neurol 10:16
Cárdenas G, Jung H, Ríos C, Fleury A, Soto-Hernández JL (2010b) Severe cysticercal meningitis: clinical and imaging characteristics. Am J Trop Med Hyg 82:121–125
Carpio A, Fleury A. Hauser WA (2013) Neurocysticercosis: five new things. Neurol Clin Pract 3:118–125
Carpio A, Hauser W (2002) Prognosis for seizure recurrence in patients with newly diagnosed neurocysticercosis. Neurology 59:1730–1734
Carpio A, Hauser WA (2009) Epilepsy in the developing world. Curr Neurol Neurosci Rep 9:319–326
Carpio A, Placencia M, Santillan F, Escobar A (1994) A proposal for a classification of neurocysticercosis. Can J Neurosci 21:43–47
Carpio A, Escobar A, Hauser WA (1998) Epilepsy and cysticercosis: a critical review. Epilepsia 39:1025–1040
Carpio A, Kelvin E, Bagiella E, Leslie D, Leon P, Andrews H, Hauser WA, Ecuadorian Neurocysticercosis Group (2008) Effects of albendazole treatment on neurocysticercosis: a randomized controlled trial. J Neurol Neurosurg Psychiatry 79:1050–1055
Chavarría A, Roger B, Fragoso G, Tapia G, Fleury A, Dumas M, Dessein A, Larralde C, Sciutto E (2003) TH2 profile in asymptomatic Taenia solium human neurocysticercosis. Microbes Infect 5:1109–1115
Chavarría A, Fleury A, García E, Márquez C, Fragoso G, Sciutto E (2005) Relationship between the clinical heterogeneity of neurocysticercosis and the immune-inflammatory profiles. Clin Immunol 116:271–278
Chavarría A, Fleury A, Bobes RJ, Morales J, Fragoso G, Sciutto E (2006) A depressed peripheral cellular immune response is related to symptomatic neurocysticercosis. Microbes Infect 8:1082–1089
Croker C, Redelings M, Reporter R, Sorvillo F, Mascola L, Wilkins P (2012) The impact of neurocysticercosis in California: a review of hospitalized cases. PLoS Negl Trop Dis 6:e1480
Cukiert A, Puglia P, Scapolan HB, Vilela MM, Marino Júnior R (1994) Congruence of the topography of intracranial calcifications and epileptic foci. Arq Neuropsiquiatr 52:289–294
Das K, Mondal GP, Banerjee M, Mukherjee BB, Singh OP (2007) Role of antiparasitic therapy for seizures and resolution of lesions in neurocysticercosis patients: an 8 year randomised study. J Clin Neurosci 14:1172–1177
De Aluja A, Morales Soto J, Sciutto E (2012) A programme to control taeniosis-cysticercosis (Taenia solium) in Mexico. In: Rodriguez-Morales A (ed) Current topics in tropical medicine. InTech, New York, available from: http://www.intechopen.com/books/current-topics-in-tropical-medicine/a-program-to-control-taeniosis-cysticercosis-taenia-solium-in-mexico.
De Souza A, Thennarasu K, Yeshraj G, Kovoor JM, Nalini A (2009) Randomized controlled trial of albendazole in new onset epilepsy and MRI confirmed solitary cerebral cysticercal lesion: effect on long-term seizure outcome. J Neurol Sci 276:108–114
Del Brutto OH, Rajshekhar V, White AC Jr et al (2001) Proposed diagnostic criteria for neurocysticercosis. Neurology 57:177–183
Del Brutto OH, Roos KL, Coffey CS, Garcıa HH (2006) Meta-analysis: cysticidal drugs for neurocysticercosis: albendazole and praziquantel. Ann Intern Med 45:43–51
Diazgranados-Sánchez JA, Barrios-Arrázola G, Costa JL, Burbano-Pabon J, Pinzón-Bedoya J (2008) Ivermectin as a therapeutic alternative in neurocysticercosis that is resistant to conventional pharmacological treatment. Rev Neurol 46:671–674
Dumas JL, Visy JM, Belin C, Gaston A, Goldlust D, Dumas M (1997) Parenchymal neurocysticercosis: follow-up and staging by MRI. Neuroradiology 39:12–18
Escobar A (1983) The pathology of neurocysticercosis. In: Palacios E, Rodriguez-Carbajal J, Taveras JM (eds) Cysticercosis of central nervous system. Charles C Thomas, Springfield
Estañol B, Corona T, Abad P (1986) A prognostic classification of cerebral cysticercosis: therapeutic implications. J Neurol Neurosurg Psychiatry 49:1131–1134
Ferreira LS, Zanardi VA, Scotoni AE, Li LM, Guerreiro MM (2001) Childhood epilepsy due to neurocysticercosis: a comparative study. Epilepsia 42:1438–1444
Ferreira LS, Li LM, Zanardi VA, Guerreiro MM (2002) Number and viability of parasite influence seizure frequency in children with neurocysticercosis. Arq Neuropsiquiatr 60:909–911
Fleury A, Morales J, Bobes RJ, Dumas M, Yánez O, Piña J, Carrillo-Mezo R, Martínez JJ, Fragoso G, Dessein A, Larralde C, Sciutto E (2006) An epidemiological study of familial neurocysticercosis in an endemic Mexican community. Trans R Soc Trop Med Hyg 100:551–558
Fleury A, Hernández M, Avila M, Cárdenas G, Bobes RJ, Huerta M, Fragoso G, Uribe-Campero L, Harrison LJ, Parkhouse RM, Sciutto E (2007) Detection of HP10 antigen in serum for diagnosis and follow-up of subarachnoidal and intraventricular human neurocysticercosis. J Neurol Neurosurg Psychiatry 78:970–974
Fleury A, Escobar A, Fragoso G, Sciutto E, Larralde C (2010) Clinical heterogeneity of human neurocysticercosis results from complex interactions among parasite, host and environmental factors. Trans R Soc Trop Med Hyg 104:243–250
Fleury A, Carrillo-Mezo R, Flisser A, Sciutto E, Corona T (2011) Subarachnoid basal neurocysticercosis: a focus on the most severe form of the disease. Expert Rev Anti Infect Ther 9: 123–133
Flisser A, Espinoza B, Tovar A, Plancarte A, Correa D (1986) Host–parasite relationship in cysticercosis: immunologic study in different compartments of the host. Vet Parasitol 20:95–102
Forlenza OV, Filho AH, Nobrega JP, dos Ramos Machado L, de Barros NG, de Camargo CH, da Silva MF (1997) Psychiatric manifestations of neurocysticercosis: a study of 38 patients from a neurology clinic in Brazil. J Neurol Neurosurg Psychiatry 62: 612–616
Garcia HH, Pretell EJ, Gilman RH, Martinez SM, Moulton LH, Del Brutto OH, Herrera G, Evans CA, Gonzalez AE; Cysticercosis Working Group in Peru (2004) A trial of antiparasitic treatment to reduce the rate of seizures due to cerebral cysticercosis. N Engl J Med 350:249–258
Garcia-Dominguez C, Correa D, Rabiela MT, Flisser A (1991) Praziquantel treatment of muscle Taenia solium cysticercosis. 4. Reversible in vitro effect. Parasitol Res 77:691–696
Goel RK, Ahmad FU, Vellimana AK, Suri A, Chandra PS, Kumar R, Sharma BS, Mahapatra AK (2008) Endoscopic management of intraventricular neurocysticercosis. J Clin Neurosci 15:1096–1101
Goel D, Mittal M, Bansal KK, Singhal A (2010) Natural history of solitary cerebral cysticercosis cases after albendazole therapy: a longitudinal follow-up study from India. Acta Neurol Scand 121:204–208
Gonzalez AE, Gavidia C, Falcon N, Bernal T, Verastegui M, Garcia HH, Gilman RH, Tsang VC; Cysticercosis Working Group in Peru (2001) Protection of pigs with cysticercosis from further infections after treatment with oxfendazole. Am J Trop Med Hyg 65:15–18
Goodman K, Ballagh SA, Carpio A (1999) Case control study of seropositivity for cysticercosis in Cuenca, Ecuador. Am J Trop Med Hyg 60:70–74
Huerta M, de Aluja AS, Fragoso G, Toledo A, Villalobos N, Hernández M, Gevorkian G, Acero G, Díaz A, Alvarez I, Avila R, Beltrán C, Garcia G, Martinez JJ, Larralde C, Sciutto E (2001) Synthetic peptide vaccine against Taenia solium pig cysticercosis: successful vaccination in a controlled field trial in rural Mexico. Vaccine 20:262–266
Jain S, Padma MV, Kanga U, Mehra NK, Puri A, Maheshwari MC (1999) Family studies and human leukocyte antigen class II typing in Indian probands with seizures in association with single small enhancing computed tomography lesions. Epilepsia 40:232–238
Jung-Cook H (2012) Pharmacokinetic variability of antihelmintics: implications for the treatment of neurocysticercosis. Expert Rev Clin Pharmacol 5:21–30
Kelvin EA, Carpio A, Bagiella E, Leslie D, Leon P, Andrews H, Hauser WA; Ecuadorian Neurocysticercosis Group (2009a) The association of host age and gender with inflammation around neurocysticercosis cysts. Ann Trop Med Parasitol 103:487–499
Kelvin EA, Carpio A, Hesdorffer DC, Bagiella E, Leslie D, Leon P, Andrews H, Hauser WA; Ecuadorian Neurocysticercosis Group (2009b) Investigation of familial aggregation of seizures in neurocysticercosis patients. Epilepsy Res 84:67–71
Kelvin EA, Carpio A, Bagiella E, Leslie D, Leon P, Andrews H, Hauser WA; Ecuadorian Neurocysticercosis Group (2011) Seizure in people with newly diagnosed active or transitional neurocysticercosis. Seizure 20:119–125
Kioumehr F, Dadsetan MR, Feldman N, Mathison G, Moosavi H, Rooholamini SA, Verma RC (1995) Postcontrast MRI of cranial meninges: leptomeningitis versus pachymeningitis. J Comput Assist Tomogr 19:713–720
Kowacs PA, Rogacheski E, Muzzio J, Werneck LC (2006) The role of the irritative zone and of the number and distribution of calcifications in the severity of epilepsy associated with intracranial calcifications. Arq Neuropsiquiatr 64:905–911
Lacey E (1990) Mode of action of benzimidazoles. Parasitol Today 6:112–115
Laclette JP, Ornelas Y, Merchant MT, Willms K (1982) Ultrastructure of the surrounding envelopes of Taenia solium eggs. In: Flisser A, Willms K, Laclette JP et al (eds) Cysticercosis: present state of knowledge and perspectives. Academic, New York
Laclette JP, Shoemaker CB, Richter D, Arcos L, Pante N, Cohen C, Bing D, Nicholson-Weller A (1992) Paramyosin inhibits complement C1. J Immunol 148:124–128
Leite JP, Terra-Bustamante VC, Fernandes RM, Santos AC, Chimelli L, Sakamoto AC, Assirati JA, Takayanagui OM (2000) Calcified neurocysticercotic lesions and postsurgery seizure control in temporal lobe epilepsy. Neurology 55:1485–1491
Loureiro das Chagas MG, D’Oliveira A Jr, Tavares-Neto J (2003) Clinical aspects of neurocysticercosis at semi-desert region of the Brazilian northeast. Arq Neuropsiquiatr 61:398–402
Lucato LT, Guedes MS, Sato JR, Bacheschi LA, Machado LR, Leite CC (2007) The role of conventional MR imaging sequences in the evaluation of neurocysticercosis: impact on characterization of the scolex and lesion burden. AJNR Am J Neuroradiol 28:1501–1504
Machado LR (2010) The diagnosis of neurocysticercosis: a closed question? Arq Neuropsiquiatr 68:1–2
Manreza ML (2000) Epilepsia e neurocisticerose. In: Guerreiro CAM, Guerreiro MM, Cendes F, Lopes-Cendes I (eds) Epilepsia. Lemos Editorial, Sao Paulo
Maravilla P, Souza V, Valera A et al (2003) Detection of genetic variation in Taenia solium. J Parasitol 89:1250–1254
Maravilla P, Gonzalez-Guzman R, Zuñiga G, Peniche A, Dominguez-Alpizar JL, Reyes-Montes R, Flisser A (2008) Genetic polymorphism in Taenia solium cysticerci recovered from experimental infections in pigs. Infect Genet Evol 8:213–216
Michelet L, Fleury A, Sciutto E, Kendjo E, Fragoso G, Paris L, Bouteille B (2011) Human neurocysticercosis: comparison of different diagnostic tests in cerebrospinal fluid. J Clin Microbiol 49:195–200
Mont’Alverne Filho FE, Machado Ldos R, Lucato LT, Leite CC (2011) The role of 3D volumetric MR sequences in diagnosing intraventricular neurocysticercosis: preliminary results. Arq Neuropsiquiatr 69:74–78
Murthy JM, Yangala R (1999) Acute symptomatic seizures – incidence and etiological spectrum: a hospital-based study from South India. Seizure 8:162–165
Nash TE, Pretell EJ, Lescano AG, Bustos JA, Gilman RH, Gonzalez AE, Garcia HH; Cysticercosis Working Group in Peru (2008) Perilesional brain oedema and seizure activity in patients with calcified neurocysticercosis: a prospective cohort and nested case-control study. Lancet Neurol 7:1099–1105
Ndimubanzi PC, Carabin H, Budke CM, Nguyen H, Qian YJ, Rainwater E, Dickey M, Reynolds S, Stoner JA (2010) A systematic review of the frequency of neurocyticercosis with a focus on people with epilepsy. PLoS Negl Trop Dis 4:e870
Ngowi HA, Carabin H, Kassuku AA, Mlozi MR, Mlangwa JE, Willingham AL 3rd (2008) A health-education intervention trial to reduce porcine cysticercosis in Mbulu District, Tanzania. Prev Vet Med 85:52–67
Pal DK, Carpio A, Sander JWAS (2000) Neurocysticercosis and epilepsy. J Neurol Neurosurg Psychiatry 68:137–143
Patel R, Jha S, Yadav RK (2006) Pleomorphism of the clinical manifestations of neurocysticercosis. Trans R Soc Trop Med Hyg 100:134–141
Proaño JV, Torres-Corzo J, Rodríguez-Della Vecchia R, Guizar-Sahagun G, Rangel-Castilla L (2009) Intraventricular and subarachnoid basal cisterns neurocysticercosis: a comparative study between traditional treatment versus neuroendoscopic surgery. Childs Nerv Syst 25:1467–1475
Proaño-Narvaez JV, Meza-Lucas A, Mata-Ruiz O, García-Jerónimo RC, Correa D (2002) Laboratory diagnosis of human neurocysticercosis: double-blind comparison of enzyme-linked immunosorbent assay and electroimmunotransfer blot assay. J Clin Microbiol 40:2115–2118
Rabiela MT, Rivas A, Flisser A (1989) Morphological types of Taenia solium cysticerci. Parasitol Today 5:357–359
Ramos-Kuri M, Montoya RM, Padilla A, Govezensky T, Díaz ML, Sciutto E, Sotelo J, Larralde C (2002) Immunodiagnosis of neurocysticercosis. Disappointing performance of serology (enzyme-linked immunosorbent assay) in an unbiased sample of neurological patients. Arch Neurol 49:633–636
Rathore C, Thomas B, Kesavadas C, Radhakrishnan K (2012) Calcified neurocysticercosis lesions and hippocampal sclerosis: potential dual pathology? Epilepsia 53:e60-62. doi: 10.1111/j.1528-1167.2011.03386.x
Robles C, Chavarrıa M (1979) Presentacion de un caso clınico de cisticercosis cerebral tratado medicamente con un nuevo farmaco: Praziquantel. Salud Pub Mex XXI:603–618
Rodrigues CL, de Andrade DC, Livramento JA, Machado LR, Abraham R, Massaroppe L, Lucato LT, Caramelli P (2012) Spectrum of cognitive impairment in neurocysticercosis: differences according to disease phase. Neurology 78:861–866
Rosenfeld EA, Byrd SE, Shulman ST (1996) Neurocysticercosis among children in Chicago. Clin Infect Dis 23:262–268
Ruiz-Garcia M, Gonzalez-Astiazarán A, Rueda-Franco F (1997) Neurocysticercosis in children. Clinical experience in 122 patients. Childs Nerv Syst 13:608–612
Sáenz B, Ruíz-Garcia M, Jiménez E, Hernández-Aguilar J, Suastegui R, Larralde C, Sciutto E, Fleury A (2006) Neurocysticercosis: clinical. radiologic, and inflammatory differences between children and adults. Pediatr Infect Dis J 25:801–803
Sakamoto AC et al (1999) Cysticercosis and epilepsy. In: Kotagal P, Luders HO (eds) The epilepsies: etiologies and prevention. Academic, San Diego
Sarti E, Schantz PM, Avila G, Ambrosio J, Medina-Santillán R, Flisser A (2000) Mass treatment against human taeniasis for the control of cysticercosis: a population-based intervention study. Trans R Soc Trop Med Hyg 94:85–89
Sciutto E, Rosas G, Hernández M, Morales J, Cruz-Revilla C, Toledo A, Manoutcharian K, Gevorkian G, Blancas A, Acero G, Hernández B, Cervantes J, Bobes RJ, Goldbaum FA, Huerta M, Diaz-Orea A, Fleury A, de Aluja AS, Cabrera-Ponce JL, Herrera-Estrella L, Fragoso G, Larralde C (2007) Improvement of the synthetic tri-peptide vaccine (S3Pvac) against porcine Taenia solium cysticercosis in search of a more effective, inexpensive and manageable vaccine. Vaccine 25:1368–1378
Sharma P, Garg RK, Verma R, Singh MK, Shukla R (2011) Risk of seizure recurrence in patients of new-onset partial seizure having a solitary cysticercus granuloma of brain or normal neuroimaging. J Neurol Sci 301:21–26
Sikasunge CS, Johansen MV, Willingham AL 3rd, Leifsson PS, Phiri IK (2008) Taenia solium porcine cysticercosis: viability of cysticerci and persistency of antibodies and cysticercal antigens after treatment with oxfendazole. Vet Parasitol 158:57–66
Singh G (1997) Neurocysticercosis in South-Central America and the Indian subcontinent. A comparative evaluation. Arq Neuropsiquiatr 55:349–356
Singh G, Kaushal V, Ram S, Kaushal RK, Dhanuka AK, Khurana S (1999) Cysticercus immunoblot assay in patients with single, small enhancing lesions and multilesional neurocysticercosis. J Assoc Physicians India 47:476–479
Singh G, Rajshekhar V, Murthy JM, Prabhakar S, Modi M, Khandelwal N, Garcia HH (2010) A diagnostic and therapeutic scheme for a solitary cysticercus granuloma. Neurology 75:2236–2245
Singhi P, Ray M, Singhi S, Khandelwal N (2000) Clinical spectrum of 500 children with neurocysticercosis and response to albendazole therapy. J Child Neurol 15:207–213
Sotelo J, Marin C (1987) Hydrocephalus secondary to cysticercotic arachnoiditis. A long-term follow-up review of 92 cases. J Neurosurg 66:686–689
Sotelo J, Guerrero J, Rubio F (1988) A new classification based on active and inactive forms. Arch Intern Med 148:442–445
Takayanagui OM, Odashima NS, Bonato PS, Lima JE, Lanchote VL (2011) Medical management of neurocysticercosis. Expert Opin Pharmacother 12:2845–2856
Terra-Bustamante VC, Coimbra ER, Rezek KO, Escorsi-Rosset SR, Guarnieri R, Dalmagro CL, Inuzuka LM, Bianchin MM, Wichert-Ana L, Alexandre V, Takayanagui OM, Araújo D, dos Santos AC, Carlotti CG, Walz R, Markowitsch HJ, Sakamoto AC (2005) Cognitive performance of patients with mesial temporal lobe epilepsy and incidental calcified neurocysticercosis. J Neurol Neurosurg Psychiatry 76:1080–1083
Thussu A, Chattopadhyay A, Sawhney IM, Khandelwal N (2008) Albendazole therapy for single small enhancing CT lesions (SSECTL) in the brain in epilepsy. J Neurol Neurosurg Psychiatry 79:272–275
Torres-Corzo JG, Tapia-Pérez JH, Vecchia RR, Chalita-Williams JC, Sánchez-Aguilar M, Sánchez-Rodríguez JJ (2010) Endoscopic management of hydrocephalus due to neurocysticercosis. Clin Neurol Neurosurg 112:11–16
Tsang VC, Brand JA, Boyer AE (1989) An enzyme-linked immunoelectrotransfer blot assay and glycoproteins antigens for diagnosing human cysticercosis (Taenia solium). J Infect Dis 174: 1007–1009
Vega R, Piñero D, Ramanankandrasana B, Dumas M, Bouteille B, Fleury A, Sciutto E, Larralde C, Fragoso G (2003) Population genetic structure of Taenia solium from Madagascar and Mexico: implications for clinical profile diversity and immunological technology. Int J Parasitol 33:1479–1485
Velasco TR, Zanello PA, Dalmagro CL, Araújo D Jr, Santos AC, Bianchin MM, Alexandre V Jr, Walz R, Assirati JA, Carlotti CG Jr, Takayanagui OM, Sakamoto AC, Leite JP (2006) Calcified cysticercotic lesions and intractable epilepsy: a cross sectional study of 512 patients. J Neurol Neurosurg Psychiatry 77:485–488
WHO (2007) Global plan to combat neglected tropical diseases 2008-2015. World Health Organization, Geneva
Wilson M, Gryan R, Fried J, Ware D, Schantz P, Pilcher JB, Tsang VCW (1991) Clinical evaluation of the cysticercosis enzyme-linked immunoelectrotransfer blot in patients with neurocysticercosis. J Infect Dis 164:1007–1009
Winkler AS, Blocher J, Auer H, Gotwald T, Matuja W, Schmutzhard E (2008) Anticysticercal and antitoxocaral antibodies in people with epilepsy in rural Tanzania. Trans R Soc Trop Med Hyg 102:1032–1038
Xiao ZX, Zhao CY, Liu LP (1986) Albendazole treatment in cerebral cysticercosis. Chin J Int Med 25:100–102
Zee CS, Go JL, Kim PE, DiGiorgio CM (2000) Imaging of neurocysticercosis. Neuroimaging Clin N Am 10:391–407
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Carpio, A., Fleury, A. (2014). Neurocysticercosis: Neurology and Neurobiology. In: Bentivoglio, M., Cavalheiro, E., Kristensson, K., Patel, N. (eds) Neglected Tropical Diseases and Conditions of the Nervous System. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-8100-3_7
Download citation
DOI: https://doi.org/10.1007/978-1-4614-8100-3_7
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-8099-0
Online ISBN: 978-1-4614-8100-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)