Abstract
Nutritional influences have sustained consequences for growth and wellbeing in later life. To date, evidence has emerged that various chronic diseases in adulthood, such as cardiovascular disease or diabetes mellitus, have origins in the period of growth. Overweight during childhood has both immediate and long-term health consequences. Over the last few decades, the obesity prevalence in children has increased dramatically in USA and European countries [1]. Since obesity is taking place in stable populations over a very short period of time, the epidemic of obesity cannot be attributed to a changed genetic background [2]. Although the contribution of hereditary factors has to be acknowledged [3], it is accepted that current obesity trends are driven by changes in exposure to environmental factors. Nutritional factors are thought to play a major role in the rising prevalence of obesity worldwide.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
FormalPara Key Points-
Emerging evidence suggests that modifying diet quality in childhood offers preventive potential for health in later life with regard to later body composition or puberty onset.
-
To date, evidence from prospective analyses focusing on single nutrients and foods is inconclusive regarding the relevance of few dietary factors in childhood for body composition.
-
With regard to puberty timing, current evidence suggests a beneficial influence of higher intakes of vegetable protein and isoflavone, and lower animal protein intakes.
-
These beneficial nutrients may be indicative of a specific dietary pattern.
-
Dietary pattern analyses, which consider diet as a whole and account for interactions of nutrients and foods, may provide much needed insights into dietary factors implicated in growth during childhood.
Introduction
Nutritional influences have sustained consequences for growth and wellbeing in later life. To date, evidence has emerged that various chronic diseases in adulthood, such as cardiovascular disease or diabetes mellitus, have origins in the period of growth. Overweight during childhood has both immediate and long-term health consequences. Over the last few decades, the obesity prevalence in children has increased dramatically in USA and European countries [1]. Since obesity is taking place in stable populations over a very short period of time, the epidemic of obesity cannot be attributed to a changed genetic background [2]. Although the contribution of hereditary factors has to be acknowledged [3], it is accepted that current obesity trends are driven by changes in exposure to environmental factors. Nutritional factors are thought to play a major role in the rising prevalence of obesity worldwide.
Puberty onset is a milestone of growth. Age of puberty timing is considered to be of general public health relevance, since an early age at puberty onset may be an intermediary factor for a number of diseases in adulthood, including hormone-related cancers [4, 5], earlier all-cause mortality [6], and a higher risk of metabolic syndrome or cardiovascular disease [7]. The influence of modifiable risk factors such as nutrition on puberty onset has thus been addressed by a number of studies.
The purpose of this chapter is to summarize the available observational evidence regarding the role of nutritional factors for growth. The review begins with an introduction of the use of dietary pattern analyses (in particular among children) and an introduction to critical periods during growth, and is then organized into two sections as follows: relevance of diet quality and dietary factors for (1) body composition and (2) puberty timing. The final section discusses public health relevance.
Diet Quality: Nutrients, Foods/Food Groups and Dietary Patterns
As far as nutritional influences on health are concerned, energy intake/energy density and nutrient compositions of diets, i.e., intakes of carbohydrate, fat, protein as well as micronutrients, have been investigated systematically over the last years. Besides nutrients, the unfavorable contribution of several food items or food groups has been discussed, e.g., snacks, French fries, sugar-sweetened beverages (SSBs), fast foods, and sweets, while other frequently consumed food item or food groups have been suggested to have a protective effect, such as fruits, vegetables, legumes, and high-fiber breakfast cereals.
However, analyses which focus on a single or a few nutrients or foods have several conceptual limitations. Meals consist of a variety of foods with complex combinations of nutrients, which are likely to be interactive. A dietary pattern may thus be more relevant to identify associations between nutrition and health outcomes. Since dietary patterns may be easier for the public to interpret or translate into daily diets, studying dietary patterns could also have important public health implications. It is impossible to measure dietary patterns directly. Statistical methods are thus used to analyze the collected dietary information and to characterize dietary patterns. To date, three approaches have been used in the literature: dietary indices, factor analysis and cluster analysis, and reduced rank regression (RRR).
Dietary Indices
Dietary indices build on a priori defined parameters. To date, a variety of dietary indices, which are typically constructed on the basis of current dietary recommendations, have been proposed to assess overall diet quality. Evaluating the health effects of adherence to dietary recommendations by individuals can provide a practical way for the public to translate the recommendations into diets, e.g., the healthy eating index (HEI) [8] is a summary measure of the degree to which an individual’s diet conforms to the serving recommendations of the US Department of Agriculture Food Guide Pyramid for five major food groups and to specific recommendations in the US Dietary Guidelines for Americans.
To date, several dietary indices have been developed for children: the Nutritional Quality Index, the Youth Healthy Eating Index (YHEI), the Revised Children’s Diet Quality Index (RC-DQI), the Dietary Guideline Index for Children and Adolescents (DGI-CA), the diet quality index (DQI), the healthy diet indicator (HDI), and the Mediterranean diet score (Table 12.1). These measure adhere to different dietary recommendations: nutrients- or food groups-specific recommendations, or a combination of both.
Factor Analysis and Cluster Analysis
Factor analysis includes both common factor analysis and principal component analysis (PCA). PCA is commonly used to define dietary patterns. Factor analysis is a multivariate statistical technique to identify common underlying dimensions (factors or patterns) of food consumption. Factor analysis aggregates specific food items or food groups on the basis of the degree to which food items in the dataset are correlated with one another. Then, a summary score for each pattern is derived and can be used in either correlation or regression analysis to examine associations between various eating patterns and the outcome of interest. Compared with factor analysis, cluster analysis aggregates individuals into relatively homogeneous subgroups (clusters) with similar food consumption. Individuals are classified into distinct clusters or groups. After the identification of cluster procedure, further analyses are used to interpret association of the dietary patterns with outcomes.
Both factor analysis and cluster analysis, which are purely explorative, have been commonly used in adults, while to date few studies among children used these approaches to address the relevance of nutrition for body composition (Table 12.2).
Reduced Rank Regression
PCA is purely explorative, i.e., it explains as much variation in food intake as possible, however, that does not mean that much variation in important nutrients will be explained. Therefore, it might be wiser to focus on the variation in those nutrients that presumably affect the incidence of disease. The statistical method known as reduced rank regression (RRR), which provides the opportunity to determine linear functions of predictors (foods) by maximizing the explained variation in responses (disease-related nutrients), has thus been applied in epidemiology [9]. RRR is neither an a priori nor a purely exploratory statistical method. Since RRR uses both information sources, data from the study and prior information to define responses, it represents an a posteriori method. To date, few studies in children and adolescents have used the RRR method to address diet quality (Table 12.2).
Critical Periods During Growth
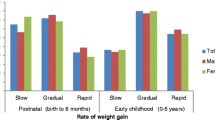
Critical periods of development have been well recognized for many behavioral and physiologic developmental processes. The possibility that nutritional alterations during critical periods of development entrain nutritional states, such as obesity, has also been mentioned [10, 11]. In 1994, Dietz defined critical periods for the overweight/obesity development as “a developmental stage, in which physiologic alterations increase the later prevalence of obesity” [12]. According to observational evidence, there appear to be at least three critical periods in childhood for the development of obesity and its complications in late life [11, 13, 14]: gestation and early infancy, the period of preschool and preadolescence, and finally, the period of adolescence (Fig. 12.1). Regarding diet quality and eating behavior, both early life and mid-childhood may represent particularly sensitive phases for their long-term effects in later life. Furthermore, the hormonal changes regulating appetite, satiety, and fat distribution that occur during puberty, especially physiological insulin resistance [15], may put adolescents at increased risk for weight gain [16]. Hence, diet quality in these critical periods may have sustained consequences for growth and wellbeing in later life.
Relevance of Diet Quality for Body Composition
In Early Life
Maternal nutritional status at conception has been associated with obesity or adverse health outcomes in general [17]. Evidence for the role of diet quality during pregnancy is only emerging. Low birth weight is commonly interpreted as a marker of poor fetal nutrition. However, the influences from prenatal period may be modified by the early postnatal period, one of the critical “windows” for growth. The importance of early postnatal life for the body composition development is highlighted by the fact that breastfeeding seems to reduce obesity risk in later childhood [18, 19]. Also, concern has been expressed that higher protein intakes during the period of complementary feeding and early childhood could be associated with both body mass index (BMI) and percentage body fat in later childhood [20, 21]. Recently, added sugar intake at low intake levels during early childhood has been suggested to not be critical for body composition in childhood, however, detrimental effects on body composition development may emerge when added sugar intakes are increased to higher levels [22]. Of interest, the factors discussed as potentially relevant to body composition in early life may be markers for overall diet quality, e.g., breastfeeding is discussed to be confounded by associations with other behavioral factors, and increased intake of added sugar may resemble a marker for other behavioral changes contributing to increases in BMI.
In Mid-Childhood and Puberty
The period of preschool and preadolescence is another important time window for body composition development. Furthermore, adolescents change their lifestyle as they test their autonomy and assert their independence [23], which may affect both their eating behaviors and their physical activity. When addressing the relevance of dietary factors for body composition during these periods, it should be borne in mind that cross-sectional studies do not allow statements on the direction of the observed association, an issue of particular relevance for the outcome overweight/obesity. Therefore, only prospective evidence is reviewed in the following section.
Nutrients and Foods
Energy Intake
Overweight is generally believed to be a consequence of an energy imbalance. Current longitudinal studies, which have addressed the relevance of energy intake in childhood on the body composition, have however not shown significant associations of energy intake with childhood weight gain [24, 25], which may reflect the methodological challenge to accurately measure energy intake. Recently, interest in dietary energy density, i.e., the ratio of the amount of food to total energy intake, has emerged. Higher energy density is commonly interpreted as a marker of diet quality. However, the few prospective studies relating energy density in children to their body composition have yielded mixed results [26–30] (Table 12.3).
Macronutrients
Macronutrients, which are thought to play a role in regulation of body weight, have been investigated systematically (Table 12.3). The evidence from studies analyzing the association between dietary fat and obesity among children is inconsistent [31–38]. In addition, type of dietary fat has received little attention in relation to childhood obesity. The existent prospective studies on protein intake in children and body composition development have yielded mixed results: three studies have shown an association with later overweight/obesity [32, 39, 40], while two other analyses found no relation [34, 37]. In most [24, 39, 41], but not all studies [32], intakes of total carbohydrate among children were not related to the development of body mass.
Carbohydrate Quality
More recently, increased attention to quality of dietary carbohydrate rather than quantity of carbohydrate intake has begun to clarify the role of dietary carbohydrate in the obesity epidemic. High fiber intake has been reported to yield beneficial effects on objective and subjective measures of satiety in experimental studies conducted in adults [42]. However, among children, only one [43] of the few [44–46] prospective observational studies has demonstrated that dietary fiber intakes were independently associated with measures of obesity (Table 12.3). Additionally, dietary whole grain is supposed to be beneficial for body weight regulation, since whole grains are rich in a myriad of nutrients, including vitamins, minerals, phytochemicals, and other substances that have been related to body weight regulation [47]. To date, the only existent prospective analysis regarding adolescents suggested that whole-grain intake is not relevant for the body composition development during puberty [46] (Table 12.3).
In addition, high dietary glycemic index (GI: calculated as the blood glucose response to a 50 g (or 25 g) carbohydrate portion of food, expressed as a percentage of the same amount of carbohydrate from a reference food, either glucose or white bread [48]) has been proposed as a risk factor for weight gain. However, regarding children and adolescents, two prospective analyses [45, 46] reported no association between dietary GI and BMI or body fatness (Table 12.3).
In particular, the increased consumption of SSB brought up discussions on the relevance of added sugar and SSB for weight gain especially in childhood. However, the results from prospective studies focusing on sucrose intake and added sugar intake in children are inconsistent [45, 49, 50] (Table 12.3). To date, four meta-analyses were conducted [51–54] addressing the relevance of SSB for body composition. Two [52, 53] of them concluded that increased consumption of SSB in children and adolescents is associated with obesity risk, while another meta-analysis [51] judged the effect as almost zero. Mattes et al. did not find any significant effect of lowering the consumption of SSB on BMI, however, in the meta-analysis of a subgroup, the authors found a consistent effect of interventions on the BMI of subjects with higher BMI or obesity at baseline [54]. Since the publication of the meta-analyses, further nine cohort studies in children [55–60] and adolescents [61–63] regarding the relation of SSB with body composition have yielded mixed results. Taken together, according to a number of studies including meta-analysis, an increased consumption of SSB among children and adolescents may be related to later overweight/obesity risk. However, in view of inconsistence, the level of evidence should be judged to indicate a possible association only (Table 12.3). In addition, an increased risk of obesity is especially observed in children and adolescents with initially already increased BMI or already existing overweight.
Dietary Pattern
Overall, the current evidence for dietary factors implicated in body composition development suggests little relevance of diet among childhood except for a potential role of SSB. In this context, it should be noted that consumption of SSB may be strongly related to overall lifestyle and thus be indicative of overall diet quality. It is thus intriguing to consider that analysis of diet quality and/or dietary pattern may provide more insights into the role of diet in the development of body composition during childhood. To date, five cross-sectional studies [64–68] in children and adolescents have directly examined the association between diet quality using dietary quality indices and body composition. Using YHEI scores to characterize the diet quality of 16,452 children aged 9–14 years, Feskanich et al. reported that children with higher YHEI scores (higher diet quality) had a lower BMI [64]. Similarly, Hurley et al. analyzed the diet quality among 317 US children using YHEI scores and they found that lower percent body/abdominal fat was associated with higher diet quality [65]. In addition, using data from the National Health and Examination Survey 1999–2002 from 1,521 children aged 2–5 years, the authors have shown that childhood obesity prevalence decreased significantly with increasing diet quality characterized by RC-DQI score [66]. In line with this analysis, Jennings et al. suggested that children with higher diet quality indices based on the US (DQI) and the WHO (HDI) healthy eating guidelines have lower body weight [67]. However, only a weak positive association was reported in a recent study conducted in 4–10-year-old Australian children [68]: using the age-specific DGI-CA, weak positive associations were observed between DGI-CA score and BMI or waist circumference Z-scores. Since cross-sectional data in relation of body composition suffer from potential reverse causality, prospective analyses, which are able to consider the baseline weight status, may be more insightful. To date, only two prospective studies [69, 70] were conducted in children using dietary pattern methods (Table 12.2). The analysis from the DONALD study used two dietary quality indices to identify the diet quality among 222 German children. In this study, diet quality in preadolescents was not independently associated with later body composition. A dietary quality index created on the basis of previous knowledge is an “a priori” dietary pattern [55]. As outlined above, dietary pattern analyses with PCA, which describes the variation in intake of food items, is an alternative approach. Cutler et al. [70] examined associations between dietary patterns identified with PCA based on dietary data in an adolescent cohort and weight status both cross-sectionally and over a 5-year period. In cross-sectional analyses, dietary patterns characterized by higher vegetable intakes were associated with lower risk of overweight in girls, whereas higher adherence to a “sweet and salty snack food” pattern was associated with lower overweight risk in boys. However, these associations were no longer significant in prospective analyses after adjusting for baseline weight status [70].
The fact that in these studies no long-term associations were seen between dietary pattern in children and later overweight may on the one hand relates to the overarching relevance of baseline body composition for later body mass in comparing to current diet quality. Dietary pattern predicting the change of body composition could be of interest in future analyses. On the other hand, the absence of clear relations may be due to the method used for the identification of dietary pattern. Compared with dietary quality indices, PCA is an a posteriori statistical method and solely describes the variation in dietary data. Instead, relating the correlation structure of foods and nutrient intakes to the health outcomes, by methods such as RRR, may reveal more meaningful dietary pattern. This approach has been used in a recent multiple cross-sectional analysis [71] identifying dietary pattern among children predictive of bone and body fat (Table 12.2). This analysis suggested that a dietary pattern characterized by a high intake of dark-green and deep-yellow vegetables was related to low fat mass and high bone mass, while high fried-food intake was related to high fat mass and high processed-meat intake was related to high bone mass. Adjustment for body composition and energy intake at baseline did not influence these associations. Since the patterns identified using RRR in this cohort may change during the time and may not be reproduced in other cohorts, further studies are needed before firm conclusions can be drawn for children. In summary, although an examination of dietary patterns has certain advantages over traditional methods of examining single foods or nutrients, more evidence using advanced approaches is needed to fully elucidate benefits of nutrition for body composition development.
Relevance of Diet Quality for Puberty Timing
Nutrition during early life, which is largely reflected by breastfeeding, has been discussed as a potential determinant of pubertal timing. However, prospective analyses have not found an independent association of breastfeeding with age at menarche [72–75], age at take-off, or age at peak height velocity [74]. Furthermore, protein intake in the first and second year of life may not to be critical for the timing of puberty onset [76].
The influence of various nutrient/food intakes in childhood on puberty timing is increasingly acknowledged in a number of studies. We recently reviewed these data (for details on this summary of observational data please see [77]). To date, energy intake, intakes of fat and fatty acids, fiber, protein (animal protein and vegetable protein), micronutrients, and dietary isoflavone intake have been investigated in various observational studies for their potential relevance to puberty timing. Of them, six analyses suggest notable associations between dietary protein intakes and puberty timing: children in the highest groups of vegetable or animal protein experience their puberty onset up to 7 months later or 7 months earlier, respectively (Table 12.4). One study has shown a strong association between dietary isoflavones and puberty timing among girls, i.e., girls with high isoflavone intakes may experience the onset of breast development and peak height velocity approximately 7–8 months later (Table 12.4). Delays in pubertal timing in response to beneficial dietary habits (higher intakes of vegetable protein and isoflavone, and lower animal protein intakes) may be of substantial public health relevance: a later age at both peak height velocity and menarche is related to a reduced risk of breast cancer, and a later menarcheal age is also associated with a lower total mortality. These nutrients identified as beneficial for later puberty onset may indicate a specific relevance of diet quality. More analyses focusing on the overall diet quality are thus warranted.
Public Health Consideration
Body composition and body fat distribution change rapidly during growth; in addition, puberty onset is to date considered to be of general public health relevance for both sexes. Emerging evidence suggests a relevance of diet quality to both body composition development and puberty timing. Future studies in diverse population are needed to identify which aspects of diet quality or which pattern is of particular relevance. This information could be of major public health relevance, since long-term eating habits are shaped during childhood and adolescence. While it is generally accepted that poor diet quality could conduce overweight/obesity, the importance of diet quality for puberty onset, which is relevant to health in later life, is currently not recognized. Educational interventions focusing on diet quality should not be limited to children and adolescent individuals. Parents with good dietary awareness (or nutritional knowledge) are thought to be more likely to make healthy food choices for their children [78]. Moreover, the intake preference of children is influenced not only by the types of foods present in the household but also by the amount of those foods available to them. The parental influence of food choice should thus be taken into account for the promotion of dietary habits of children. In addition to family dynamics, school factors and the “built environment”, which refers to the neighborhoods and communities in which children live, also play a role in influencing the diet quality among youth. Finally, the food industries should be urged to increase their efforts to provide healthier food choices to beneficially influence the diet quality of children and adolescents.
Conclusions
In conclusion, emerging evidence suggests that modifying diet quality in early life could offer preventive potential for health in later life with regard to later body composition or puberty onset. Current approaches focusing on single nutrients and foods have identified only few dietary factors in childhood as potentially relevant for body composition. With regard to puberty timing, current evidence suggests a beneficial influence of dietary protein and isoflavone intake, which may indicate a particular relevance of overall diet quality. The approach of dietary pattern analyses, which considers the interaction of nutrients and foods, may provide new insights into the effect of diet as a whole and should be adapted for this issue.
Abbreviations
- BMI:
-
Body mass index
- DGI-CA:
-
Dietary Guideline Index for Children and Adolescents
- DQI:
-
Diet quality index
- GI:
-
Glycemic index
- HDI:
-
Healthy diet indicator
- HEI:
-
Healthy eating index
- PCA:
-
Principal component analysis
- RC-DQI:
-
Revised Children’s Diet Quality Index
- RRR:
-
Reduced rank regression
- SSB:
-
Sugar-sweetened beverage
- YHEI:
-
Youth Healthy Eating Index
References
Ogden CL, Carroll MD, Curtin LR, Lamb MM, Flegal KM. Prevalence of high body mass index in US children and adolescents, 2007–2008. JAMA. 2010;303(3):242–9.
Ebbeling CB, Pawlak DB, Ludwig DS. Childhood obesity: public-health crisis, common sense cure. Lancet. 2002;360(9331):473–82.
Farooqi IS, O’Rahilly S. Genetic factors in human obesity. Obes Rev. 2007;8 Suppl 1:37–40.
Velie EM, Nechuta S, Osuch JR. Lifetime reproductive and anthropometric risk factors for breast cancer in postmenopausal women. Breast Dis. 2005;24:17–35.
Garner MJ, Turner MC, Ghadirian P, Krewski D. Epidemiology of testicular cancer: an overview. Int J Cancer. 2005;116(3):331–9.
Jacobsen BK, Oda K, Knutsen SF, Fraser GE. Age at menarche, total mortality and mortality from ischaemic heart disease and stroke: the Adventist Health Study, 1976–88. Int J Epidemiol. 2009;38(1):245–52.
Remsberg KE, Demerath EW, Schubert CM, Chumlea WC, Sun SS, Siervogel RM. Early menarche and the development of cardiovascular disease risk factors in adolescent girls: the Fels Longitudinal Study. J Clin Endocrinol Metab. 2005;90(5):2718–24.
Kennedy ET, Ohls J, Carlson S, Fleming K. The Healthy Eating Index: design and applications. J Am Diet Assoc. 1995;95(10):1103–8.
Hoffmann K, Schulze MB, Schienkiewitz A, Nothlings U, Boeing H. Application of a new statistical method to derive dietary patterns in nutritional epidemiology. Am J Epidemiol. 2004;159(10):935–44.
Godfrey KM, Barker DJ. Fetal nutrition and adult disease. Am J Clin Nutr. 2000;71(5 Suppl):1344S–52.
Rolland-Cachera MF, Deheeger M, Guilloud-Bataille M, Avons P, Patois E, Sempe M. Tracking the development of adiposity from one month of age to adulthood. Ann Hum Biol. 1987;14(3):219–29.
Dietz WH. Critical periods in childhood for the development of obesity. Am J Clin Nutr. 1994;59(5):955–9.
Ravelli GP, Stein ZA, Susser MW. Obesity in young men after famine exposure in utero and early infancy. N Engl J Med. 1976;295(7):349–53.
Braddon FE, Rodgers B, Wadsworth ME, Davies JM. Onset of obesity in a 36 year birth cohort study. Br Med J (Clin Res Ed). 1986;293(6542):299–303.
Hannon TS, Janosky J, Arslanian SA. Longitudinal study of physiologic insulin resistance and metabolic changes of puberty. Pediatr Res. 2006;60(6):759–63.
de Ridder CM, Thijssen JH, Bruning PF, Van den Brande JL, Zonderland ML, Erich WB. Body fat mass, body fat distribution, and pubertal development: a longitudinal study of physical and hormonal sexual maturation of girls. J Clin Endocrinol Metab. 1992;75(2):442–6.
Robinson SM, Godfrey KM. Feeding practices in pregnancy and infancy: relationship with the development of overweight and obesity in childhood. Int J Obes (Lond). 2008;32 Suppl 6:S4–10.
Arenz S, Ruckerl R, Koletzko B, von Kries R. Breast-feeding and childhood obesity—a systematic review. Int J Obes Relat Metab Disord. 2004;28(10):1247–56.
Owen CG, Martin RM, Whincup PH, Smith GD, Cook DG. Effect of infant feeding on the risk of obesity across the life course: a quantitative review of published evidence. Pediatrics. 2005;115(5):1367–77.
Günther AL, Buyken AE, Kroke A. Protein intake during the period of complementary feeding and early childhood and the association with body mass index and percentage body fat at 7 y of age. Am J Clin Nutr. 2007;85(6):1626–33.
Koletzko B. Long-term consequences of early feeding on later obesity risk. In: Rigo J, Ziegler EE, editors. Protein and energy requirements in infancy and childhood. Basel: Karger AG; 2006. p. 1–18.
Herbst A, Diethelm K, Cheng G, Alexy U, Icks A, Buyken AE. Direction of associations between added sugar intake in early childhood and body mass index at age 7 years may depend on intake levels. J Nutr. 2011;141(7):1348–54.
Irwin Jr CE, Igra V, Eyre S, Millstein S. Risk-taking behavior in adolescents: the paradigm. Ann N Y Acad Sci. 1997;817:1–35.
Bogaert N, Steinbeck KS, Baur LA, Brock K, Bermingham MA. Food, activity and family—environmental vs biochemical predictors of weight gain in children. Eur J Clin Nutr. 2003;57(10):1242–9.
Maffeis C, Talamini G, Tato L. Influence of diet, physical activity and parents’ obesity on children’s adiposity: a four-year longitudinal study. Int J Obes Relat Metab Disord. 1998;22(8):758–64.
Johnson L, Mander AP, Jones LR, Emmett PM, Jebb SA. A prospective analysis of dietary energy density at age 5 and 7 years and fatness at 9 years among UK children. Int J Obes (Lond). 2008;32(4):586–93.
Kral TV, Berkowitz RI, Stunkard AJ, Stallings VA, Brown DD, Faith MS. Dietary energy density increases during early childhood irrespective of familial predisposition to obesity: results from a prospective cohort study. Int J Obes (Lond). 2007;31(7):1061–7.
Gunther AL, Stahl LJ, Buyken AE, Kroke A. Association of dietary energy density in childhood with age and body fatness at the onset of the pubertal growth spurt. Br J Nutr. 2011;106(3):345–9.
Butte NF, Cai G, Cole SA, Wilson TA, Fisher JO, Zakeri IF, et al. Metabolic and behavioral predictors of weight gain in Hispanic children: the Viva la Familia Study. Am J Clin Nutr. 2007;85(6):1478–85.
McCaffrey TA, Rennie KL, Kerr MA, Wallace JM, Hannon-Fletcher MP, Coward WA, et al. Energy density of the diet and change in body fatness from childhood to adolescence; is there a relation? Am J Clin Nutr. 2008;87(5):1230–7.
Klesges RC, Klesges LM, Eck LH, Shelton ML. A longitudinal analysis of accelerated weight gain in preschool children. Pediatrics. 1995;95(1):126–30.
Skinner JD, Bounds W, Carruth BR, Morris M, Ziegler P. Predictors of children’s body mass index: a longitudinal study of diet and growth in children aged 2–8 y. Int J Obes Relat Metab Disord. 2004;28(4):476–82.
Lee Y, Mitchell DC, Smiciklas-Wright H, Birch LL. Diet quality, nutrient intake, weight status, and feeding environments of girls meeting or exceeding recommendations for total dietary fat of the American Academy of Pediatrics. Pediatrics. 2001;107(6):E95.
Carruth BR, Skinner JD. The role of dietary calcium and other nutrients in moderating body fat in preschool children. Int J Obes Relat Metab Disord. 2001;25(4):559–66.
Robertson SM, Cullen KW, Baranowski J, Baranowski T, Hu S, de Moor C. Factors related to adiposity among children aged 3 to 7 years. J Am Diet Assoc. 1999;99(8):938–43.
Newby PK, Peterson KE, Berkey CS, Leppert J, Willett WC, Colditz GA. Dietary composition and weight change among low-income preschool children. Arch Pediatr Adolesc Med. 2003;157(8):759–64.
Magarey AM, Daniels LA, Boulton TJ, Cockington RA. Does fat intake predict adiposity in healthy children and adolescents aged 2–15 y? A longitudinal analysis. Eur J Clin Nutr. 2001;55(6):471–81.
Jago R, Baranowski T, Baranowski JC, Thompson D, Greaves KA. BMI from 3–6 y of age is predicted by TV viewing and physical activity, not diet. Int J Obes (Lond). 2005;29(6):557–64.
Rolland-Cachera MF, Deheeger M, Akrout M, Bellisle F. Influence of macronutrients on adiposity development: a follow up study of nutrition and growth from 10 months to 8 years of age. Int J Obes Relat Metab Disord. 1995;19(8):573–8.
Gunther AL, Remer T, Kroke A, Buyken AE. Early protein intake and later obesity risk: which protein sources at which time points throughout infancy and childhood are important for body mass index and body fat percentage at 7 y of age? Am J Clin Nutr. 2007;86(6):1765–72.
Klesges RC, Isbell TR, Klesges LM. Relationship between dietary restraint, energy intake, physical activity, and body weight: a prospective analysis. J Abnorm Psychol. 1992;101(4):668–74.
Pereira MA, Ludwig DS. Dietary fiber and body-weight regulation. Observations and mechanisms. Pediatr Clin North Am. 2001;48(4):969–80.
Davis JN, Alexander KE, Ventura EE, Toledo-Corral CM, Goran MI. Inverse relation between dietary fiber intake and visceral adiposity in overweight Latino youth. Am J Clin Nutr. 2009;90(5):1160–6.
Iqbal Kring SI, Heitmann BL. Fiber intake, not dietary energy density, is associated with subsequent change in BMI z-score among sub-groups of children. Obes Facts. 2008;1(6):331–8.
Buyken AE, Cheng G, Gunther AL, Liese AD, Remer T, Karaolis-Danckert N. Relation of dietary glycemic index, glycemic load, added sugar intake, or fiber intake to the development of body composition between ages 2 and 7 y. Am J Clin Nutr. 2008;88(3):755–62.
Cheng G, Karaolis-Danckert N, Libuda L, Bolzenius K, Remer T, Buyken AE. Relation of dietary glycemic index, glycemic load, and fiber and whole-grain intakes during puberty to the concurrent development of percent body fat and body mass index. Am J Epidemiol. 2009;169(6):667–77.
Slavin JL, Martini MC, Jacobs Jr DR, Marquart L. Plausible mechanisms for the protectiveness of whole grains. Am J Clin Nutr. 1999;70(3 Suppl):459S–63.
Jenkins DJ, Wolever TM, Taylor RH, Barker H, Fielden H, Baldwin JM, et al. Glycemic index of foods: a physiological basis for carbohydrate exchange. Am J Clin Nutr. 1981;34(3):362–6.
Williams CL, Strobino BA. Childhood diet, overweight, and CVD risk factors: the Healthy Start project. Prev Cardiol. 2008;11(1):11–20.
Ruottinen S, Niinikoski H, Lagstrom H, Ronnemaa T, Hakanen M, Viikari J, et al. High sucrose intake is associated with poor quality of diet and growth between 13 months and 9 years of age: the special Turku Coronary Risk Factor Intervention Project. Pediatrics. 2008;121(6):e1676–85.
Forshee RA, Anderson PA, Storey ML. Sugar-sweetened beverages and body mass index in children and adolescents: a meta-analysis. Am J Clin Nutr. 2008;87(6):1662–71.
Malik VS, Willett WC, Hu FB. Sugar-sweetened beverages and BMI in children and adolescents: reanalyses of a meta-analysis. Am J Clin Nutr. 2009;89(1):438–9; author reply 439–440.
Vartanian LR, Schwartz MB, Brownell KD. Effects of soft drink consumption on nutrition and health: a systematic review and meta-analysis. Am J Public Health. 2007;97(4):667–75.
Mattes RD, Shikany JM, Kaiser KA, Allison DB. Nutritively sweetened beverage consumption and body weight: a systematic review and meta-analysis of randomized experiments. Obes Rev. 2011;12(5):346–65.
Johnson L, Mander AP, Jones LR, Emmett PM, Jebb SA. Is sugar-sweetened beverage consumption associated with increased fatness in children? Nutrition. 2007;23(7–8):557–63.
Dubois L, Farmer A, Girard M, Peterson K. Regular sugar-sweetened beverage consumption between meals increases risk of overweight among preschool-aged children. J Am Diet Assoc. 2007;107(6):924–34; discussion 934–925.
Fiorito LM, Marini M, Francis LA, Smiciklas-Wright H, Birch LL. Beverage intake of girls at age 5 y predicts adiposity and weight status in childhood and adolescence. Am J Clin Nutr. 2009;90(4):935–42.
Kral TV, Stunkard AJ, Berkowitz RI, Stallings VA, Moore RH, Faith MS. Beverage consumption patterns of children born at different risk of obesity. Obesity (Silver Spring). 2008;16(8):1802–8.
Lim S, Zoellner JM, Lee JM, Burt BA, Sandretto AM, Sohn W, et al. Obesity and sugar-sweetened beverages in African-American preschool children: a longitudinal study. Obesity (Silver Spring). 2009;17(6):1262–8.
Tam CS, Garnett SP, Cowell CT, Campbell K, Cabrera G, Baur LA. Soft drink consumption and excess weight gain in Australian school students: results from the Nepean study. Int J Obes (Lond). 2006;30(7):1091–3.
Libuda L, Alexy U, Sichert-Hellert W, Stehle P, Karaolis-Danckert N, Buyken AE, et al. Pattern of beverage consumption and long-term association with body-weight status in German adolescents–results from the DONALD study. Br J Nutr. 2008;99(6):1370–9.
Laurson K, Eisenmann JC, Moore S. Lack of association between television viewing, soft drinks, physical activity and body mass index in children. Acta Paediatr. 2008;97(6):795–800.
Vanselow MS, Pereira MA, Neumark-Sztainer D, Raatz SK. Adolescent beverage habits and changes in weight over time: findings from Project EAT. Am J Clin Nutr. 2009;90(6):1489–95.
Feskanich D, Rockett HR, Colditz GA. Modifying the Healthy Eating Index to assess diet quality in children and adolescents. J Am Diet Assoc. 2004;104(9):1375–83.
Hurley KM, Oberlander SE, Merry BC, Wrobleski MM, Klassen AC, Black MM. The healthy eating index and youth healthy eating index are unique, nonredundant measures of diet quality among low-income. African American adolescents. J Nutr. 2009;139(2):359–64.
Kranz S, Findeis JL, Shrestha SS. Use of the Revised Children’s Diet Quality Index to assess preschooler’s diet quality, its sociodemographic predictors, and its association with body weight status. J Pediatr (Rio J). 2008;84(1):26–34.
Jennings A, Welch A, van Sluijs EM, Griffin SJ, Cassidy A. Diet quality is independently associated with weight status in children aged 9–10 years. J Nutr. 2011;141(3):453–9.
Golley RK, Hendrie GA, McNaughton SA. Scores on the dietary guideline index for children and adolescents are associated with nutrient intake and socio-economic position but not adiposity. J Nutr. 2011;141(7):1340–7.
Cheng G, Gerlach S, Libuda L, Kranz S, Gunther AL, Karaolis-Danckert N, et al. Diet quality in childhood is prospectively associated with the timing of puberty but not with body composition at puberty onset. J Nutr. 2010;140(1):95–102.
Cutler GJ, Flood A, Hannan PJ, Slavin JL, Neumark-Sztainer D. Association between major patterns of dietary intake and weight status in adolescents. Br J Nutr. 2012;108(2):349–56.
Wosje KS, Khoury PR, Claytor RP, Copeland KA, Hornung RW, Daniels SR, et al. Dietary patterns associated with fat and bone mass in young children. Am J Clin Nutr. 2010;92(2):294–303.
Moisan J, Meyer F, Gingras S. A nested case–control study of the correlates of early menarche. Am J Epidemiol. 1990;132(5):953–61.
Merzenich H, Boeing H, Wahrendorf J. Dietary fat and sports activity as determinants for age at menarche. Am J Epidemiol. 1993;138(4):217–24.
Karaolis-Danckert N, Buyken AE, Sonntag A, Kroke A. Birth and early life influences on the timing of puberty onset: results from the DONALD (DOrtmund Nutritional and Anthropometric Longitudinally Designed) Study. Am J Clin Nutr. 2009;90(6):1559–65.
Blell M, Pollard TM, Pearce MS. Predictors of age at menarche in the newcastle thousand families study. J Biosoc Sci. 2008;40(4):563–75.
Gunther AL, Karaolis-Danckert N, Kroke A, Remer T, Buyken AE. Dietary protein intake throughout childhood is associated with the timing of puberty. J Nutr. 2010;140(3):565–71.
Cheng G, Buyken AE, Shi L, Karaolis-Danckert N, Kroke A, Wudy S, et al. Beyond overweight: nutrition as an important lifestyle factor influencing the puberty timing. Nutr Rev. 2012;70(3):133–52.
Variyam JN, Blaylock J, Lin B, Ralston K, Smallwood D. Mother’s nutrition knowledge and children’s dietary intakes. Am J Agric Econ. 1999;81:373–84.
Berkey CS, Gardner JD, Frazier AL, Colditz GA. Relation of childhood diet and body size to menarche and adolescent growth in girls. Am J Epidemiol. 2000;152(5):446–52.
Kissinger DG, Sanchez A. The association of dietary factors with the age of menarche. Nutr Res. 1987;7:471–9.
Remer T, Shi L, Buyken AE, Maser-Gluth C, Hartmann MF, Wudy SA. Prepubertal adrenarchal androgens and animal protein intake independently and differentially influence pubertal timing. J Clin Endocrinol Metab. 2010;95(6):3002–9.
Rogers IS, Northstone K, Dunger DB, Cooper AR, Ness AR, Emmett PM. Diet throughout childhood and age at menarche in a contemporary cohort of British girls. Public Health Nutr. 2010;13(12):2052–63.
de Ridder CM, Thijssen JH. Van ‘t Veer P, van Duuren R, Bruning PF, Zonderland ML, Erich WB. Dietary habits, sexual maturation, and plasma hormones in pubertal girls: a longitudinal study. Am J Clin Nutr. 1991;54(5):805–13.
Cheng G, Remer T, Prinz-Langenohl R, Blaszkewicz M, Degen GH, Buyken AE. Relation of isoflavones and fiber intake in childhood to the timing of puberty. Am J Clin Nutr. 2010;92(3):556–64.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Cheng, G., Buyken, A.E. (2013). Diet Quality in Childhood: Impact on Growth. In: Preedy, V., Hunter, LA., Patel, V. (eds) Diet Quality. Nutrition and Health. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4614-7339-8_12
Download citation
DOI: https://doi.org/10.1007/978-1-4614-7339-8_12
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4614-7338-1
Online ISBN: 978-1-4614-7339-8
eBook Packages: MedicineMedicine (R0)