Abstract
Ecosystem services provide the basis for all human activity. Maintaining their sustained function is of critical concern as the issues of sustainability addressed here in this encyclopedia are approached. At root, the ecosystem is a thermodynamic system receiving, collecting, transforming, and dissipating solar energy. The energy pathways are varied and complex and lead to the diversity of form and services available on the earth. Therefore, it is necessary to understand the ecosystem as a thermodynamic system and how the energy flows enter, interconnect, and disperse from the environmental system. Ecological network methodologies exist to investigate and analyze these flows. In particular, partitioning the flow into boundary input, noncycled internal flow, and cycled internal flow shows the extent to which reuse and recycling arise in ecosystems. The intricate, complex network structures are responsible for these processes all within the given thermodynamic constraints. Design of sustainable human systems could be informed by these organizational patterns, in order to use effectively the energy available. This article demonstrates the need for flow analysis, provides a brief example using a well-studied ecosystem, and discusses some of the ecosystem development tendencies which can be addressed using ecosystem flow analysis.
This chapter was originally published as part of the Encyclopedia of Sustainability Science and Technology edited by Robert A. Meyers. DOI:10.1007/978-1-4419-0851-3
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Definition
Ecosystem services provide the basis for all human activity. Maintaining their sustained function is of critical concern to the issues of sustainability addressed here in this encyclopedia. At root, the ecosystem is a thermodynamic system receiving, collecting, transforming, and dissipating solar energy. The energy pathways are varied and complex and lead to the diversity of form and services available on the earth. Therefore, it is necessary to understand the ecosystem as a thermodynamic system and how the energy flows enter, interconnect, and disperse from the environmental system. Ecological network methodologies exist to investigate and analyze these flows. In particular, partitioning the flow into boundary input, noncycled internal flow, and cycled internal flow shows the extent to which reuse and recycling arise in ecosystems. The intricate, complex network structures are responsible for these processes all within the given thermodynamic constraints. Design of sustainable human systems could be informed by these organizational patterns, in order to use effectively the energy available. This article demonstrates the need for flow analysis, provides a brief example using a well-studied ecosystem, and discusses some of the ecosystem development tendencies which can be addressed using ecosystem flow analysis.
Introduction
Ecosystems, like all environmental systems, are open, thermodynamic systems (Fig. 5.1). They take in energy from an external energy source – almost entirely from solar energy, although geothermal or geochemical energy drives some systems. Ecosystem structure is built with the energy, and then the degraded energy is passed back to the environment. For some time period the “stored solar energy” persists in the forms perceived on the earth’s surface as biomass stores of living and nonliving organic material – such as all of us. In Frank Herbert’s novel Dune, he envisioned that on an arid planet an important functional role of each individual was “carrying” water in oneself. In a world dominated by thermodynamic constraints, such as ours, everyone is an energy carrier. These stores are temporary and fluid. In this perspective, one could substitute the current object-mode paradigm with a flow-based, rheomode. In other words, all stocks are flows. The paper that one writes on now is only an ephemeral stage of the energy which started in the solar reactions, traveled to earth, was captured by a photosynthesizing organism, converted into storage in the xylem of that organism, and harvested and transformed into the useable product that one now holds. But that is not the end. Over time, the paper will slowly degrade or decompose, or perhaps the energy release will be sudden through combustion. In any case, the objects held are transitory states in a long-term dynamic from (energy) source to (energy) sink. Changing this view would help in appreciating better the difference between capital and income, because, for example, harvesting natural capital stock (i.e., a forest) into a flow (deforestation) is not equivalent yet treated as substitutable in the current accounts. The rheomode approach could help focus clearly on the difference and thus sustainability of stocks and flows.
Concerned about sustainability over a human time horizon, one must be aware of the constraints imposed by these sources and sinks. From the input side, clearly, it is necessary that one does not extract resources at a rate faster than they can regenerate. And concerning the output, the waste emissions should not exceed to the assimilative capacity of the local environment [1]. These are the most basic constraints imposed by open-system, thermodynamics. Humans have transformed the earth’s surface to maximize the capture of photosynthetic energy – think of the millennia over which the Chinese, Romans, Babylonians, etc., have manipulated and manicured the landscape for agricultural production. Still, these societies rose and fell within the solar energy domain. These societies collapsed if they overconsumed the base resources or if they polluted their local environments [2]. In addition to these persistent input–output constraints, there is a third sustainability consideration currently observed in the anthropocene, in that it is not only the input–output relations, but also the structure which is created. Modern infrastructure demands the continual input of high-quality (low entropy) energy of a form not naturally delivered by ecosystem services. Furthermore, the created structure locks us into the necessity of immense energy flows for maintenance. Perhaps an apt analogy can be offered through the Greek myth of Erysichthon, who was King of Thessaly. He angered the gods by cutting down a sacred tree, and as punishment was insatiably hungry. Importantly, the more he ate, the hungrier he became. Our infrastructure, like Erysichthon, does not sit idle but continually demands upkeep such that the more structure, the more resources are needed to support this structure. It is not just about the present flows, but also the life cycle debt commitment as a result of the structure. Today that energy debt is paid almost entirely in the usage of fossil fuels – a nonrenewable resource. The scale of human activity seen today is because the application of fossil fuels to substitute solar fuels has released humans from one of the long-standing constraints on growth. And as a result, humans have exploded across the landscape. This growth was easy when there was sufficient energy to add to the system. In fact, the first growth form is boundary growth, taking energy into the system, and storing it as biomass. As long as there is more energy available, the system growth can occur unbound. The second stage of growth, network and information growth, is squeezing more utility out of the available source by coupling processes and improving efficiencies [3]. This can occur in parallel with boundary growth, but becomes a necessity when boundary growth is limited. In human systems, those immediate constraints are looming at least given the current fuel-mix options.
Natural ecosystems, dependent on the solar energy flows developed extremely complex and beautiful structures within these thermodynamic constraints. And it is a useful guide to learn from these systems as a more eco-friendly design is incorporated. Below, some of the ecosystem flow analysis methodologies are explored and applied.
Investigation of Ecosystem Flows Using Network Analysis
“There are no trash cans in nature.” This is a useful phrase reminding that waste from one entity is food/input for another. Energy, of course, has a higher dissipative factor in the reuse than material cycles such as nitrogen, phosphorus, and calcium, but still there is a complex network of pathways designed to utilize the energy available in natural ecosystems. In 1973, Hannon [4] introduced Leontief’s input–output methodology into ecology, applying it to the energy flow structure of an ecosystem. The ecosystem is represented by n compartments, and the energy flowing into compartments, within compartments and exiting compartments.
A network flow model is essentially an ecological food web (energy–matter flow of who eats whom), which also includes energy input, and nonfeeding pathways such as dissipative export out of the system and pathways to detritus. The first step is to identify the system of interest and place a boundary (real or conceptual) around it. Energy–matter transfers within the system boundary comprise the network; transfers crossing the boundary are either input or output to the network, and all transactions starting and ending outside the boundary without crossing it are external to the system and are not considered. The energy inflows and intra-system flows can be considered the production energy flow and the flows with no consumers such as metabolic energy and exported biomass are the respiration energy flow[5].
The data required for ecological network analysis are as follows: for each compartment in the network, the biomass and physiological parameters, such as consumption (C), production (P), respiration (R), and egestion (E), must be quantified [6]. Furthermore, the diet of each compartment must be apportioned amongst the inputs from other compartments (consumption) in the network. This apportionment of “who eats whom and by how much” can be depicted in a dietary flow matrix, F, where energy flows from column elements j to row elements i. For all compartments, inputs should balance outputs (C = P + R + E) in accordance with the conservation of matter and the laws of thermodynamics.
The sum of the flow matrix elements, f ij , gives the total inflow to compartmental i such that:
where z i is the boundary flow into i. The outflow from i can be expressed as:
where y i is the boundary outflow from i. At steady state, a necessary condition for the network flow analysis, T i,in = T i,out and one compartmental throughflow vector can be written as T = (T i ). The total system throughflow (TST) is given by the sum of the compartmental throughflows:
The motivation for flow partitioning begins with nondimensional flow intensities (i.e., throughflow-specific flows) which result when flows are divided by throughflows of originating compartments: g ij = f ij /T j . The elements of matrix G = (g ij ) give the dimensionless transfer efficiencies corresponding to each direct flow, f ij . Powers G m of this matrix give the indirect flow intensities associated with paths of lengths m = 2, 3, …. Due to dissipation, flow along these indirect paths approaches zero as m → ∞ so that the power series \( \sum\nolimits_{{m = 0}}^{\infty } {{\mathbf{G}^{\rm m}}} \) representing the sum of the initial, direct, and indirect flows converges to an integral flow intensity matrix, N:
N maps the steady-state input vector z into the steady-state system throughflow vector:
Term by term, flow intensities G m of different orders m are propagated over paths of different lengths m. The first term, I, brings the input vector z across the system boundary as input z j to each initiating compartment, j. The second term, G, produces the first-order direct transfers from each j to each i in the system. The remaining terms where m > 1 define mth order indirect flows associated with length m paths. As stated before, these go to zero in the limit as m → ∞, which is necessary for series convergence. This demonstrates that each “direct” flow f ij at steady state is actually composed of flow elements of all orders, m = 1, 2, …. In fact, a major result of this flow analysis is that indirect flows can dominate direct flows: \( \sum\nolimits_{{m = 2}}^{\infty } {{\mathbf{G}^{\text m}} > \mathbf{G}} \). In the above developments F, T, z, and y represent matter or energy fluxes, and G and N are dimensionless intensive flows.
Finn [7] developed a cycling index using this basic approach and, Higashi et al. [8] described a three-mode partition of the flows, expanded by Fath et al. [9] into five modes (Table 5.1). Mode 0 is the boundary input into the system. Mode 1 accounts for all flow in which substance moves from node j to a terminal node i for the first time only without cycling. Mode 2 is flow cycled at terminal nodes i of each (i, j) pair. Mode 3 is component-wise dissipative flow in the sense that it exits from node i never to return again to i. Mode 4 is the boundary output from i constituting systemically dissipative flows exiting the system (Fig. 5.2). δ ij is the Kronecker delta defined by δ ij = 1 for i = j and δ ij = 0 for i ≠ j.
Schematic of flow partitioning for a central node i, in relation to other network compartments. Flow reaches the node directly across the boundary, f(0), by passing through other compartments before reaching i, f(1), and leaving i to cycle back again, f(2). Outflows symmetrically mirror these inputs
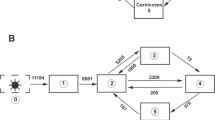
Network diagram of Cone Spring ecosystem energy flows [10]. All flows are in kcal/m2/year. Biomasses are in kcal/m2. Green arrows are exogenous boundary inflows. Black arrows are exports of useable energy. Red ground symbols represent metabolic energy loss
Portion of original energy that remains as path length increases (From Braner [11], reprinted with permission). There is still noticeable energy in the system after 10 steps
Note, the symmetry in that quantitatively Mode 0 = Mode 4, and Mode 1 = Mode 3. This is due to the conservation of mass/energy and at steady state what comes in must go out. Mode 2 represents the cycled flow which has additional impact on the system by staying in the system longer, increasing the residence time, and returning to its source of emanation. Therefore, total system throughflow can be written as:
And, on a nodal basis, throughflow is:
The mode partition designation clearly shows the contribution of flow within the entire system of interactions.
Example: Cone Spring Ecosystem Model
A classic example is the Cone Spring ecosystem model developed by Tilly [10]. In this model, there are five compartments representing: (1) plants, (2) bacteria, (3) detritivores, (4) carnivores, and (5) detritus (Fig. 5.3). There are 2 external inputs (to plants and detritus), 8 internal flows, and each compartment has boundary outflow representing metabolic or egestion losses. The internal flows from columns j to rows i are given by:
Compartmental throughflows are: T = [11 184, 11 484, 5 204, 2 384, 370] and TST = 30 627.
The nondimensional flow fractions are given by:
And the integral flow matrix is:
Looking at Table 5.2, it is seen that over 38% of the flow comes directly to a node from the first instant across a system boundary and 52% of the flow originates from one compartment and enters another compartment for the first time without cycling. Slightly over 9% of the total energy flow is material that has cycled by exiting and reentering the same compartmental node. In other words, about 3,000 kcal/m2/y of the total system throughflow is comprised of energy due to cyclic pathways which retain the energy in the system. This additional boost is important to the overall function of the Cone Spring ecosystem. A noticeable contribution of cycled flow is a common phenomenon in all ecosystems. Another way to demonstrate this importance of cycling, and the fundamental shift it has on how an ecosystem should be viewed, was given by Braner [11]. While investigating the same five-compartment Cone Spring model above, he showed that cyclic pathways identified by flow analysis reveal that the original boundary flow persists in the system much longer than obviously apparent. For contrast, in a five-compartment food chain model – a type often used, incorrectly, to represent an ecosystem – the longest path could only be four steps in length from X1 → X2 → X3 → X4 → X5. Real ecosystems have more complex structures with cycles. After those four steps, the original flow from compartment 1 would exit the system at compartment 5. According to Braner, more than 10% of the flow remains in the Cone Spring ecosystem after four steps. In fact, approximately 1% of the original flow remains after 9 steps and 0.001% is left after 15 steps (Fig. 5.4). A similar result is shown for two other ecosystems in the same figure. Therefore, the cycles, evident from flow analysis, play a very important role in the system having enough resource to function and provide ecosystem services.
Ecosystem Goal Functions
Flow analysis has another useful feature related to understanding ecosystem dynamics. Odum [12] proposed 24 different attributes which describe the ecosystem development, for aspects such as community energetics, nutrient dynamics, and overall homeostasis. The attributes dealing with energetics, which change during the ecosystem development, have also been formulated as ecological goal functions – which describe observable macroscopic patterns over time. They are not strict goal functions in the sense of mathematical optimization models (neither is economic utility although it is used as such), but indicate the tendency for ecosystems to follow during development, for example, during succession from early r-selected species and r-selecting environments to late K-selected species and K-selecting environments. Some of the more common goal functions employed include: maximum power [13], maximum dissipation [14], maximum cycling [15], maximum residence time [16], minimum specific dissipation [17, 18], maximum energy [19], and maximum ascendency [20] (see [9], for a detailed description of these). The idea is that the ecological network self-organizes itself in a way that leads to directional change in the property of these values. For example, maximum power, interpreted to mean the maximum throughflow in the network is given by: TST = f (0) + f (1) + f (2). Therefore, TST increases when there is more boundary flow (mode 0), more first passage flow (mode 1), or more cycled flow (mode 2). The mechanisms for this to increase practically relate to the system’s ability to capture more boundary flow by increasing the uptake. Both first passage flow and cycled flow also depend on the second stage of growth exemplified by the structure of the network and the efficiency of flows along each connection. Similar rationale can be made for the other goal functions listed above, and in fact it has been shown that the goal functions are complementary and mutually reinforcing in that the realization of one generally promotes the others. Together they provide a holistic view of ecosystem development through different thermodynamic perspectives. Again, the value of this ecosystem knowledge is obvious for application to design and to manage human systems sustainably. If ecosystem services are required, then the inherent dynamics of the systems used should be better understood. Human activities in line with these directions will be supported by natural processes, those that do not will experience additional resistance and therefore additional cost and difficulty. Humans are better off working with nature than against it if possible.
Conclusions and Future Directions
Ecosystem flow analysis clearly shows that the distribution of energy flow in a network is not simple. Some significant fraction of the energy remains in the system and cycles before exiting the system. This insight was evident in R. Lindeman’s [21] seminal work on Cedar Bog Lake in which he referred to his eight-compartment ecosystem as a “food-cycle.” Unfortunately, he did not have the quantitative tools at his disposal, like flow analysis, and to simplify the calculations, proceeded to analyze the system according to two distinct “food-chains,” although in reality they are linked and contain cycles. Further work in this area also neglected the presence and significance of food cycles until research in the mid-1970s (such as [22–25], and others) when network analysis techniques developed sufficiently to provide a holistic investigation of the ecosystem function. As stated above, this changes the way one must look at ecosystems, as processors and stores of energy flow. The energy does not pass quickly through but can remain and impact the system indirectly. The good news is that this flow which remains in the system is able to positively drive ecosystem processes and contribute to the overall sustainability of the system.
However, the lesson to take is that in the design of human systems, industrial processes are built sequentially, which have raw material → processing → product → disposal. There is little room for cycling and reuse. Remember, there are no trash cans in nature. Everything has a use and reuse. Efforts are now seen in industrial ecology promoting closed loop engineering and cradle-to-cradle considerations, but there is a long way to go, as evidenced by the massive amounts of raw material input and solid waste generated on a daily basis by human activity. Also, the flow analysis must include all parts and processes of the holistic integrated socio-ecological system. Future work is needed to continue to understand energy cycles in natural systems and furthermore, how to implement lessons from these into the design of socio-ecological systems. Ecosystem flow analysis clearly shows the input–output orientation flow resources have at their disposal for maintaining functional activity and can aid in sustainability science.
Abbreviations
- Consumer:
-
Heterotrophic organism that consumes other organisms for their energy requirements.
- Cycling:
-
The process by which energy or matter returns from its compartment of origin before exiting the system boundary.
- Ecological goal function:
-
Tendency observed in the orientation or directional development of ecological systems.
- Flow:
-
The transfer of energy or matter from one compartment in the system to another by active (feeding) or passive (death, egestion) means.
- Network analysis:
-
A mathematical tool to study objects as part of a connected system and to identify and quantify the direct and indirect effects in that system.
- Primary producer:
-
Photosynthesizing organism that captures external energy sources and brings it into the system as the basis for all subsequent thermodynamic activity.
- Thermodynamic system:
-
A bounded system defined by the quantities of energy and matter flowing through it.
Bibliography
Primary Literature
Daly HE, Townsend KN (1993) Valuing the earth: economics, ecology, ethics. MIT Press, Cambridge, MA, p 399
Diamond J (2005) Collapse: how societies choose to fail or succeed. Penguin Press, New York, p 575
Fath BD, Jørgensen SE, Patten BC, Straškraba M (2004) Ecosystem growth and development. Biosystems 77:213–228
Hannon B (1973) The structure of ecosystems. J Theor Biol 41:535–546
Levine SH (1977) Exploitation interactions and structure of ecosystems. J Theor Biol 69:345–355
Fath BD, Scharler U, Ulanowicz RE, Hannon B (2007) Ecological network analysis: network construction. Ecol Model 208:49–55
Finn JT (1976) Measures of ecosystem structure and function derived from analysis of flows. J Theor Biol 56:363–380
Higashi M, Patten BC, Burns TP (1993) Network trophic dynamics: the modes of energy utilization in ecosystems. Ecol Model 66:1–42
Fath BD, Patten BC, Choi JS (2001) Complementarity of ecological goal functions. J Theor Biol 208:493–506
Tilly LJ (1968) The structure and dynamics of Cone Spring. Ecol Monogr 38:169–197
Braner M (1985) A simple random path method for the analysis of flow networks. Ecol Model 28:165–180
Odum EP (1969) The strategy of ecosystem development. Science 164:262–270
Lotka AJ (1922) Contribution to the energetics of evolution. Proc Natl Acad Sci USA 8:147–151
Schneider ED, Kay JJ (1994) Life as a manifestation of the second law of thermodynamics. Math Comput Model 19:25–48
Morowitz HJ (1968) Energy flow in biology; biological organization as a problem in thermal physics. Academic, New York
Cheslak EF, Lamarra VA (1981) The residence time of energy as a measure of ecological organization. In: Mitsch WJ, Bossermann RW, Klopatek JM (eds) Energy and ecological modelling. Elsevier, Amsterdam, pp 591–600
Onsager L (1931) Reciprocal relations in irreversible processes. Phys Rev 37:405–426
Prigogine I, Wiame JM (1946) Biologie et Thermodynamique des Phenomenes Irreversible. Experientia II:451–453
Odum HT (1988) Self-organization, transformity, and information. Science 242:1132–1139
Ulanowicz RE (1997) Ecology, the ascendent perspective. Columbia University Press, New York
Lindeman RL (1942) The trophic dynamic aspect of ecology. Ecology 23:399–418
Pomeroy LR (1974) The ocean’s food web, a changing paradigm. Bioscience 24:499–504
Finn JT (1976) Measures of ecosystem structure and function derived from analysis of flows. J Theor Biol 56:363–380
Patten BC (1978) Systems approach to the concept of environment. Ohio J Sci 78:206–222
Patten BC (1985) Energy cycling in the ecosystem. Ecol Model 28:1–71
Books and Reviews
Burns TP (1989) Lindeman’s contradiction and the trophic structure of ecosystems. Ecology 70:1355–1362
Finn JT (1980) Flow analysis of models of the Hubbard Brook ecosystem. Ecology 61:562–571
Latham LG (2006) Network flow analysis algorithms. Ecol Model 192:586–600
Ulanowicz RE (1983) Identifying the structure of cycling in ecosystems. Math Biosci 65:219–237
Whipple SJ (1999) Analysis of ecosystem structure and function: extended path and flow analysis of a steady-state oyster reef model. Ecol Model 114:251–274
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Fath, B.D. (2013). Ecosystem Flow Analysis. In: Leemans, R. (eds) Ecological Systems. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-5755-8_5
Download citation
DOI: https://doi.org/10.1007/978-1-4614-5755-8_5
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-5754-1
Online ISBN: 978-1-4614-5755-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)