Abstract
The 2013 ESC-ESH Guidelines for the management of arterial hypertension stated that in elderly (>80 years) individuals, lowering SBP to < 150mmHg is strongly evidence-based. These guidelines are mainly based on the results of the Hypertension in the Very Elderly (HYVET) study which showed the beneficial effect of decreasing SBP with antihypertensive treatment in patients older than 80 years. This trial was conducted in a highly selected population of robust elderly patients with very low comorbidities. Thus, despite the interest of these results, we believe that the question of the benefits of antihypertensive treatment in the very old is not unequivocally settled and may depend on the magnitude of co-morbidities, frailty and functional decline.
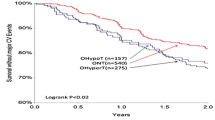
The PARTAGE (Predictive values of blood pressure and arterial stiffness in institutionalized very aged population) multicenter study performed in very old, frail subjects living in nursing homes reported a 30% increase in all-cause mortality in patients ranked in the lowest tertile of SBP (<130mmHg) compared to the two upper tertiles. These results remained identical after adjusting for several confounders such as age, gender, history of previous CV disease, index of co-morbidity, cognitive function and autonomy status. These paradoxical results can be explained by the fact that in these very old frail subjects, a low SBP may not simply be a sign of so-called good arterial health, but often of malnutrition and of co-morbidities such as heart failure, neurological disorders etc., as well as other concomitant conditions associated with poor prognosis. Irrespective of the underlying explanation, the present results indicate that the BP levels in very old “frail” individuals are evidently not reliable. These findings raise the question as to relevance of BP levels as an indicator of the cardiovascular risk in these subjects. This is of major interest since iatrogenic-induced problems are also a major issue in geriatric subjects. In fact, at the present time, no study has provided evidence that higher morbidity-mortality rates in those with very low BP is due to low BP in itself or is just a sign of general bad health.
Interventional clinical trials will provide critical information to guide physicians in order to fixe specific goals for treating hypertension and decreasing the risk of cardiovascular complications in these very old and frail individuals.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
The continuously increasing number of elderly people, especially those 80+ years of age, leads into a growing population prone to frailty, multiple comorbidities, and partial loss of autonomy. This is now one of the target populations for geriatric medicine necessitating the development of specific diagnostic and therapeutic approaches [1]. These approaches cannot be derived however from a simple extrapolation of the strategies applied in younger populations or even in elderly robust populations. Thus, assessment of cardiovascular (CV) risk in these subjects represents a major issue. High blood pressure (BP), especially systolic hypertension, is a common condition in the elderly and is considered a major determinant not only of CV morbidity and mortality but also of several other age-related diseases, frailty, and loss of autonomy [2]. Thus, high blood pressure has been related to several age-related diseases such as osteoporosis [3] and Alzheimer’s disease [4]. It has also been shown that life expectancy is reduced in middle-aged hypertensives [5, 6].
Therefore, there is no doubt that high blood pressure and high risk of morbidity and mortality go hand in hand. This concept has dominated hypertension epidemiology as well as clinical trials which have shown that in hypertensive subjects, the more important the decrease in systolic or diastolic BP (DBP), the more important the benefits in terms of cardiovascular morbidity and mortality in young, middle-aged, and older populations of both genders [7–10]. Numerous large clinical trials in individuals 65 years or older showed that antihypertensive treatment in older adults is as beneficial as that in younger adults [11]. Thus, large studies such as the Systolic Hypertension in the Elderly Program [12], Swedish Trial in Old Patients [13], Medical Research Council [14], Systolic Hypertension in Europe [15], and Systolic Hypertension in China [16] have shown the benefits of reducing BP in elderly subjects. The results of the Hypertension in the Very Elderly (HYVET) study [17] also showed the beneficial effect of antihypertensive treatment in patients 80 years or older.
Although the concept “the lower the blood pressure, the better the prognosis” may be a very appealing message in terms of public health policy, several results especially in the elderly show that this dogma may not always represent the truth. One of the most typical examples is the inverse relationship between DBP and CV risk observed in elderly individuals [18–21]. Furthermore, such an inverse relationship with risk has even been observed with systolic blood pressure (SBP) in very elderly subjects presenting several comorbidities [22, 23]. Actually, although in relatively fit elderly people [7, 17] high SBP is associated with higher morbidity and mortality, no association between high BP and morbidity and mortality was found in very old persons with several comorbidities [22, 24, 25]. Notably, a J-shape or an inverse relationship between BP (both systolic and diastolic) and morbidity/mortality was observed in a subset of studies [26, 27]. Clinical studies in very old persons reported a poor prognosis in those with systolic BP (SBP) <140 mmHg [28, 29]. The J-shape phenomenon associated with excessive diastolic BP reduction in elderly patients has been reported in the Practitioner’s Trial on the Efficacy of Antihypertensive Treatment in the Elderly [30] and in some post hoc analyses of large clinical studies, such as the Systolic Hypertension in the Elderly Program [31] and Systolic Hypertension in Europe [32]. In another post hoc analysis of the Systolic Hypertension in the Elderly Program, the greatest benefits in terms of lowering stroke risk were observed in patients with SBP <150 mmHg but not in those with SBP <140 mmHg [33]. Accordingly, excessive reduction of BP in the elderly, particularly in elderly patients with cardiovascular disease, might be harmful because of decreased perfusion of target organs [30].
Hence, the issue that we have attempted to address herein is what does measured BP really means and what are its determinants during the aging process. Actually, as we have developed in a recent review, the demographic, technological, and therapeutic changes over the past 20–30 years make possible now to reconsider the concept of the “role of blood pressure in the cardiovascular risk determination” [34].
The Transition from Diastolic to Systolic and Pulse Pressure (PP) During Aging
The Reasons of Increase in PP with Age
Until the ages of 50–60 years, both SBP and DBP increase with age. Thereafter, in the majority of cases, SBP increases with age disproportionally to DBP. The most common cause for the disruption of the correlation between SBP and DBP (leading to an excessive increase in SBP and PP) is the progressive stiffening of the arterial wall [35, 36]. Indeed, arterial stiffness develops as a consequence of several structural and functional changes of large arteries. Wall hypertrophy, calcium deposits, and changes in the extracellular matrix, such as an increase in collagen and in fibronectin, fragmentation and disorganization of the elastin network, non-enzymatic cross-links, and cell-matrix interactions, are the predominant structural determinants of the decrease in elastic properties and the development of large-artery stiffness [37].
It is important at this juncture to point out that SBP is dependent on left ventricular performance and on the stiffness of the aorta and other large arteries [35]. Thus, peak systolic pressure will be greater if the arterial wall is more rigid. On the other hand, after closure of the aortic valves, arterial pressure gradually falls as blood is drained to peripheral vascular networks. Minimum DBP is determined by the duration of the diastolic interval and the rate at which pressure falls. The rate of fall in pressure is influenced by the rate of outflow, i.e., peripheral resistance, and by viscoelastic arterial properties. At a given vascular resistance, the drop in diastolic pressure will be greater if the rigidity of large arteries is increased. The viscoelastic properties of arterial walls are also a determinant of the speed of propagation of the arterial pressure wave (pulse wave velocity (PWV)) and of the timing of wave reflections. Thus, stiffening of the arteries increases PWV and may be responsible for an earlier return of the reflected waves, which overlaps the incident pressure wave, thus further contributing to the increase in SBP and PP [35, 36]. Moreover, several clinical cross-sectional and longitudinal studies have shown that increases in arterial stiffness with age are not linear, being more pronounced after the age of 55–60 [38, 39], which may in turn explain the more pronounced increase in PP after this age, as reported in the Framingham study [19]. In addition to age, any disease and/or situation that induces an accelerated increase in arterial stiffness will be clinically expressed by an increase in SBP and PP. Diabetes is a typical example of accelerated arterial aging leading to a more noticeable increase in PP with age as compared to nondiabetic patients, due to a more pronounced increase in arterial stiffness [40–42]. In accordance with this concept, increased PP with age is more pronounced in diabetics with initial micro- or macroalbuminuria and retinopathy, suggesting that the progression in arterial aging is more prominent in the presence of target organ damage [42].
The Increasing Impact of Systolic/Pulse Pressure in the Elderly
Taking into account these considerations can better explain why SBP and PP better reflect CVD risk in older subjects, whereas DBP better reflects the risk in younger subjects [20, 43]. Indeed, DBP in young patients is predominantly dependent on peripheral resistance, and, therefore, low DBP reflects low peripheral resistance. In addition, in younger subjects with hyperkinetic circulation, DBP is less variable than SBP, thus better reflecting cardiovascular risk. In older subjects, a low DBP may reflect high arterial stiffness, which is a major manifestation of arterial aging, rather than low peripheral resistance [35, 36]. In this case, low DBP is associated with high SBP and high PP and increased cardiovascular risk. The clinical application of these considerations is that, as clearly stated in the latest guidelines of the JNC, “in persons older than 50 years, SBP is a much more important cardiovascular risk factor than DBP” [8].
Also, in 2003, for the first time, the European recommendations on the management of hypertension [44] have suggested that PP may represent an independent risk factor and that therapeutic studies should henceforth be conducted to assess the benefits of reducing PP in terms of cardiovascular morbidity and mortality, especially among those over 60 years of age [43]. Indeed, since the first study, conduced in 1989, which demonstrated a positive association between PP and target organ damage [45], a large number of clinical studies notably over the past 10 years have shown that increased PP is a strong predictor of coronary disease, incidence of heart failure, and cardiovascular morbidity and mortality, independently of mean BP levels [19, 46–51]. Such observations have been made in a variety of different populations but are apparently more pronounced in diabetics and elderly subjects. Threshold PP risk values have been proposed, notably a value of approximately 65 mmHg [52, 53]. The association between PP and CV mortality has also been observed in elderly patients enrolled in large clinical trials, as shown in a meta-analysis published in 2002 [54], during which seven clinical trials in the elderly were analyzed (EWPHE, HEP, MRC1, MRC2, SHEP, STOP, Syst-Eur). The subjects enrolled in these trials were elderly patients with systolic-diastolic hypertension or isolated systolic hypertension.
The SBP/PP-Related Increase in Cardiovascular Risk: Is It Only a Barometric Phenomenon?
To date, at least three hypotheses can be put forward to explain the association between SBP/PP and CV risk. Actually, these three hypotheses are complementary rather than contradictory:
-
(a)
PP increases arterial cyclic stress: Experimental studies indicate that fatigue and fracture of elastic fibers within the arterial wall are related to both steady-state and pulsatile stress [55, 56]. In vivo, the former is primarily dependent on mean arterial pressure, whereas the latter is related to amplitude of PP and also to heart rate. Therefore, increased PP by itself could be responsible for cardiac and arterial fatigue and subsequent complications such as left ventricular hypertrophy, arterial hypertrophy and dilatation, endothelial damage, and extracellular matrix changes.
-
(b)
Altered ventricular-aortic coupling influences myocardial perfusion by elevating the proportion of coronary flow during the systolic time period [57]. Thus, increased PP and low DBP lead to decreased coronary perfusion, which mainly occurs during the diastolic phase of the cardiac cycle.
-
(c)
PP as an indicator of arterial stiffness: PP is associated with CV risk since it is an indicator of arterial stiffness; therefore, PP is merely an epiphenomenon and not responsible for cardiovascular alterations. This third hypothesis seems to be the main explanation of the relationship between SBP/PP and CV morbidity and mortality. This assumption is based on the fact that the risk related to the PP is mainly observed in the elderly and is due to both high SBP and low DBP [19, 43], reflecting the typical clinical manifestations of arterial aging on the macro- and microcirculation.
The results of epidemiological studies conducted in subjects over 80 years of age also support the view that the association between PP and CV complications is not observed when other than arterial stiffness are the main determinants of PP. In a study involving very elderly institutionalized patients (mean age 87 years.), we found that PP failed to predict cardiovascular mortality. In fact, in this very frail population, low SBP and low PP mainly reflect comorbidities than low arterial stiffness; this explains why in this population aortic PWV, a direct indicator of arterial stiffness, was an independent predictor of cardiovascular mortality [25].
Similar results were also observed in other observational studies in very old, frail patients with absence or even reverse relationships between BP levels and cardiovascular risk [23, 27].
We recently studied this question in the PARTAGE (Predictive Values of Blood Pressure and Arterial Stiffness in Institutionalized Very Aged Population) multicenter study performed in 1,130 frail subjects 80 years or older living in nursing homes [39]. Almost 80 % of the participants were treated for hypertension with a mean 2.2 ± 1.0 drugs, and 63 % of men and 53 % of women treated for hypertension had an SBP <140 mmHg [58]. This contrasts with the much lower frequency (38 % of men, 23 % of women) with SBP <140 mmHg reported for subjects 80 years or older treated for hypertension in a community-living setting [59]. In the PARTAGE study [25], after 2 years, there was a 30 % increase in all-cause mortality in patients ranked in the lowest tertile of SBP (<130 mmHg) compared to the two upper tertiles. These results held after adjusting for several confounders such as age, sex, history of previous cardiovascular disease, index of comorbidity (Charlson), cognitive function (MMSE), and autonomy status (ADL). Thus, a low SBP in very old frail subjects may not simply be a sign of good arterial health. Rather, it might be the expression of malnutrition, heart failure, neurological disorders, and other comorbidities associated with poor prognosis. In fact, at present no study has provided evidence that higher morbidity-mortality rates in elderly patients with very low BP are due to low BP itself or are just a sign of general bad health. It is therefore of particular relevance that participants in the PARTAGE study had several comorbidities and were polymedicated (receiving on average 7.1 different drugs).
Hence, the absence or even the inverse associations between BP levels and CVD risk in the very elderly appears to be linked to several age-related changes as summarized below:
-
(a)
The presence of frequent comorbidities in the very elderly, in particular denutrition, heart failure, and several neurological disorders, reduces BP levels, thereby masking the association between high BP and CVD risk [60].
-
(b)
Exaggeration of BP variability, mainly SBP and PP variability, due to alterations in homeostatic mechanisms. Arterial stiffness, baroreceptor failure, and neurological diseases are responsible for this variability and for the presence of orthostatic or postprandial hypotension [61]. Therefore, SBP and PP recorded during casual measurements may not reflect more permanent SBP and PP levels. Actually, several studies have shown that in the elderly, 24 h ambulatory [62–64] or self-measured [65] BP is a better predictor of cardiovascular risk than clinical BP. Bjorklund et al. [66] showed that PP measured with ABPM has the most powerful prognostic value for cardiovascular morbidity. The Ohasama study [67] also showed that the prediction of stroke was much more precise with self-measurements than with casual clinical measurements, whereas Bobrie et al. [65] have clearly demonstrated in 5,000 treated hypertensive elderly subjects that home measurements but not clinical measurements of BP were able to predict cardiovascular events in these subjects. In the PARTAGE study, we investigated this specific question in the very old institutionalized individuals by comparing the BP levels obtained with standard casual measurements with those recorded following multiple self-measurements. These analyses showed no difference in both BP levels [39] and the predictive value of these BP levels on morbidity and mortality [59]. This result is in contrast with the results observed in community-living more robust elderly persons and shows that the “paradoxical” relationship between BP and events observed in the very old frail subjects cannot be the sole result of BP variability.
-
(c)
Finally, we should mention the relatively frequent overestimation of BP levels in the presence of severe mediacalcosis (pseudohypertension) [68] due to the lack of compressibility of peripheral arteries. However, a recent study has shown that the role of “pseudohypertension” in the elderly has been exaggerated and that what has been perceived as false elevation in brachial BP, as compared with intra-arterial pressure, is the result of discrepancies in office/clinic BP versus home/ambulatory measurements [69]. The authors concluded this interesting analysis on this topic, pointing out that “there are no legitimate elevated BP phenotypes that should be labeled as spurious, artifactual, or as pseudohypertension.”
Therefore, we think that the “paradoxical” results observed in old frail individuals are mainly related to the presence of comorbidities and/or conditions that profoundly modify the regulation of blood pressure in these subjects. These data point out the fact that BP measurements are not adequate or even misleading for the evaluation of CVD risk in the very old patients.
Influence of Arterial Aging on the Response to the Antihypertensive Treatment
Beyond this epidemiological evidence, the response to the antihypertensive treatments clearly shows that arterial aging should be taken into account in order to answer a number of questions: Why is SBP not controlled in the majority of the treated hypertensives? Is there an optimal BP decrease? What is the J-shape curve threshold for DBP, SBP, and PP? Should we be apprehensive of an excessive decrease of the BP in frail patients?
Failure to Control SBP
It has been suggested that SBP should be under 140 mmHg and that DBP be under 90 mmHg for all treated hypertensive subjects, unless diabetes or target organ damage is present, in which case lower BP levels (<130/85 mmHg) should be targeted [8, 70, 71]. However, this latter statement has been recently questioned by the latest guidelines of the ESC-ESH [2, 72]. Observational studies from several countries have demonstrated that among treated hypertensive individuals, the proportion of those who are well controlled is less than 30 % [73, 74], and a recent survey in the United Kingdom indicated that only 6 % of hypertensive subjects presented BP levels below the limit of 140/90 mmHg [75].
In France, the situation is similar: In a study conducted in a general elderly population (over 60 years of age) in Nancy (Northeast part of France), we found that only 50 % of treated patients were well controlled, i.e., SBP <140 and DBP <90 mmHg [76]. This study showed 2 key results: First, better control rates were observed in women than in men, probably due to a better compliance to the treatment by women. Second, most patients had a well-controlled DBP but still had high SBP levels. Thus, among uncontrolled subjects, 84 % were uncontrolled only for SBP (>140 mmHg) and 14 % for both SBP and DBP (>90 mmHg), while only less than 2 % were controlled for SBP but uncontrolled for DBP. These results are of importance in the prognosis of treated patients, since lack of control of SBP (but not DBP) has been shown to be a major determinant of mortality in treated hypertensives [77].
Several factors can contribute to a poor control of high SBP. Among these, the increasing prevalence of obesity, sedentary life, and high-salt diet can contribute to the resistance to antihypertensive treatment [78]. In addition to these important factors, arterial aging is the main determinant, which could explain current failure in controlling systolic blood pressure. Hence, despite the use of combination therapies, SBP in most patients remains well above the goals determined by international guidelines. However, as we mentioned above, the opposite is observed in very old frail individuals who show much lower BP levels due to the presence of several comorbidities and polymedication [39].
BP Drop with Treatment: The “J-Shape Curve”
Classically, a clinical relevant decrease in BP following antihypertensive treatment signifies a decrease by at least 6–7 mmHg, since this threshold is considered to be associated with a significant decrease in cardiovascular complications [8]. We believe that in order to correctly answer this question, we must follow a different approach: The clinical relevant decrease in BP is the one that results from an improvement in arterial function. In other words, a permanent progressive decrease in DBP and SBP of 10 mmHg in a 50-year-old hypertensive subject with a pretreatment level of 160/100 mmHg can be considered as clinically relevant since it is certainly due to a significant improvement in microcirculation and a decrease in peripheral vascular resistance. On the other hand, the same decrease in DBP but without a decrease in SBP in a 75-year-old diabetic with an initial BP of 175/100 is clearly a bad sign since it reflects the incapacity of the drug to reduce arterial stiffness which is the main determinant of systolic hypertension in this subject. Finally, an abrupt decrease in SBP and DBP from 180/85 to 160/70 in an 80-year-old subject following a combination therapy may be dangerous since it may be an indicator of dehydration and/or a decrease in cardiac output and systolic function due to administered drugs. By contrast, a progressive decrease in SBP by 20 mmHg and DBP by 10 mmHg could be again an indicator of improvement of vascular stiffness and peripheral resistance.
This approach is clearly more complicated than “tell me a number and I’ll tell you the risk,” but it nonetheless remains the only single possibility to truly answer questions regarding the “J”-shape curve and the clinical relevance and benefits from systolic and/or diastolic BP reduction, especially in the various subgroups of elderly and frail patients.
The “J-curve” describes the relationship between BP and the risk of CV morbidity and mortality. CV risk is high for an elevated BP level and is reduced in parallel with BP reduction until a nadir is reached, below which further BP reduction increases risk [79–81]. Several studies have shown that a J-shape curve exists mainly between DBP and coronary disease especially patients with several CV diseases [79, 80].
Thus, the “J-curve” legitimately brings the motto “the lower, the better” into question and confirms the need for using further diagnostic methods to evaluate arterial hypertension and personalizing the treatment. As mentioned earlier, high PP and isolated systolic hypertension in the elderly are signs of exaggerated vascular aging; thus, these subjects may be considered at high CV risk. Thus, in the presence of large-artery stiffening, antihypertensive treatment can excessively reduce DBP levels and notably protodiastolic pressure, hence contributing to a reduction in coronary flow. Thus, the association between the lowering of BP and the increase in cardiovascular risk recorded in clinical trials most likely results from marked arteriosclerosis and/or a previous unknown coronary artery disease [81].
The question of the J-curve is also of particular interest in the very old subjects. As we mentioned above, the results of the HYVET study [17] showed the beneficial effect of antihypertensive treatment in patients 80 years or older. However, one should always have in mind the design of this study: Actually, this trial was conducted in a highly selected population of fit elderly patients with baseline SBP >160 mmHg and a target SBP <150 mmHg.
Based on these considerations, the 2013 ESC-ESH Guidelines [2] for the management of arterial hypertension stated that “in the elderly, evidence is limited to individuals with initial SBP of >160 mmHg, whose SBP was reduced to values <150 mmHg but not <140 mmHg.” Therefore, the recommendation of lowering SBP to <150 mmHg in elderly individuals with SBP >160 mmHg is strongly evidence based. However, at least in elderly individuals 80 years or older, antihypertensive treatment may be considered at SBP >140 mmHg and aimed at values <140 mmHg, if the individuals are fit and treatment is well tolerated [2].
Future Directions
The development in the elderly of several age-related diseases and conditions makes that DBP and even SBP in the very old, may be misleading in the understanding of the underlying arterial state and therefore the evaluation of the cardiovascular risk. As a consequence, no clear recommendation exists regarding target BP in the management of hypertension in very old frail subjects. Moreover, little is known about whether low BP levels (e.g., SBP <130 mmHg) in these individuals in response to therapy increase rather than decrease morbidity and mortality. And then a key question is whether an aggressive lowering of SBP through the use of multiple drugs might be deleterious in very old frail subjects, tipping their delicate balance of survival. Further clinical trials, comparing different therapeutic strategies, will provide critical information to guide physicians how to treat hypertension in these individuals. The knowledge generated by these trials will have major consequences in terms of both individual and public health.
References
Tolson D, Morley JE, Rolland Y, Vellas B. Advancing nursing home practice: the International Association of Geriatrics and Gerontology Recommendations. Geriatr Nurs. 2011;32:195–7.
Authors/Task Force Members, Mancia G, Fagard R, et al. 2013 ESH/ESC Guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J. 2013;34:2159–219.
Alagiakrishnan K, Juby A, Hanley D, Tymchak W, Sclater A. Role of vascular factors in osteoporosis. J Gerontol. 2003;58:362–6.
Launer L, Webster Ross G, Petrovitch H, Masaki K, Foley D, White LR, Havlik RJ. Midlife blood pressure and dementia: the Honolulu-Asia aging study. Neurobiol Aging. 2000;21:49–55.
Franco OH, Peeters A, Bonneux L, De Laet C. Blood pressure in adulthood and life expectancy with cardiovascular disease in men and women: life course analysis. Hypertension. 2005;46:280–6.
Benetos A, Thomas F, Bean KE, Pannier B, Guize L. Role of modifiable risk factors in life expectancy in the elderly. J Hypertens. 2005;23:1803–8.
Lewington S, Clarke R, Qizilbash N, Peto R, Collins R, Prospective Studies Collaboration. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 2002;360:1903–13.
Chobanian AV, Bakris GL, Black HR, et al. Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. National Heart, Lung, and Blood Institute. National High Blood Pressure Education Program Coordinating Committee. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension. 2003;42:1206–52.
Law MR, Morris JK, Wald NJ. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ. 2009;338:b1665. doi:10.1136/bmj.b1665.
Turnbull F, Neal B, Ninomiya T, Blood Pressure Lowering Treatment Trialists’ Collaboration, et al. Effects of different regimens to lower blood pressure on major cardiovascular events in older and younger adults: meta-analysis of randomised trials. BMJ. 2008;336:1121–3.
Blood Pressure Lowering Treatment Trialists C, Turnbull F, Neal B, et al. Effects of different regimens to lower blood pressure on major cardiovascular events in older and younger adults: meta-analysis of randomised trials. BMJ. 2008;336:1121–3.
Prevention of stroke by antihypertensive drug treatment in older persons with isolated systolic hypertension. Final results of the Systolic Hypertension in the Elderly Program (SHEP). SHEP Cooperative Research Group. JAMA. 1991;265:3255–64.
Dahlof B, Lindholm LH, Hansson L, et al. Morbidity and mortality in the Swedish Trial in Old Patients with Hypertension (STOP-Hypertension). Lancet. 1991;338:1281–5.
Medical Research Council trial of treatment of hypertension in older adults: principal results. MRC Working Party. BMJ. 1992;304:405–12.
Staessen JA, Fagard R, Thijs L, et al. Randomised double-blind comparison of placebo and active treatment for older patients with isolated systolic hypertension. The Systolic Hypertension in Europe (Syst-Eur) Trial Investigators. Lancet. 1997;350:757–64.
Liu L, Wang JG, Gong L, et al. Comparison of active treatment and placebo in older Chinese patients with isolated systolic hypertension. Systolic Hypertension in China (Syst-China) Collaborative Group. J Hypertens. 1998;16:1823–9.
Beckett NS, Peters R, Fletcher AE, et al. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358:1887–98.
Benetos A, Zureik M, Morcet J, et al. A decrease in diastolic blood pressure combined with an increase in systolic blood pressure is associated with a higher cardiovascular mortality in men. J Am Coll Cardiol. 2000;35:673–80.
Franklin SS, Khan SA, Wong ND, Larson MG, Levy D. Is pulse pressure useful in predicting risk for coronary heart disease ? Circulation. 1999;100:354–60.
Khattar RS, Swales JD, Dore C, Senior R, Lahiri A. Effects of aging on the prognostic significance of ambulatory systolic, diastolic and pulse pressure in essential hypertension. Circulation. 2001;104:783–9.
Blacher J, Staessen JA, Girerd X, et al. Pulse pressure not mean pressure determines cardiovascular risk in older hypertensive patients. Arch Intern Med. 2000;160:1085–9.
Boshuizen HC, Izaks GJ, van Buuren S, Ligthart GJ. Blood pressure and mortality in elderly people aged 85 and older: community based study. BMJ. 1998;316:1780–4.
Satish S, Zhang DD, Goodwin JS. Clinical significance of falling blood pressure among older adults. J Clin Epidemiol. 2001;54:961–7.
Meaume S, Benetos A, Henry OF, et al. Aortic pulse wave velocity predicts cardiovascular mortality in subjects >70 years of age. Arterioscler Thromb Vasc Biol. 2001;21:2046–50.
Askari M, Kiely DK, Lipsitz LA. Is pulse pressure a predictor of cardiovascular complications in a frail elderly nursing home population? Aging Clin Exp Res. 2004;16:206–11.
Oates DJ, Berlowitz DR, Glickman ME, et al. Blood pressure and survival in the oldest old. J Am Geriatr Soc. 2007;55:383–8.
Molander L, Lovheim H, Norman T, et al. Lower systolic blood pressure is associated with greater mortality in people aged 85 and older. J Am Geriatr Soc. 2008;56:1853–9.
Lagaay AM, van Asperen IA, Hijmans W. The prevalence of morbidity in the oldest old, aged 85 and over: a population-based survey in Leiden, The Netherlands. Arch Gerontol Geriatr. 1992;15:115–31.
Rastas S, Pirttila T, Viramo P, et al. Association between blood pressure and survival over 9 years in a general population aged 85 and older. J Am Geriatr Soc. 2006;54:912–8.
Ogihara T. Practitioner’s trial on the efficacy of antihypertensive treatment in the elderly hypertension (the PATE-hypertension study) in Japan. Am J Hypertens. 2000;13:461–7.
Fagard RH, Staessen JA, Thijs L, et al. On-treatment diastolic blood pressure and prognosis in systolic hypertension. Arch Intern Med. 2007;167:1884–91.
Somes GW, Pahor M, Shorr RI, et al. The role of diastolic blood pressure when treating isolated systolic hypertension. Arch Intern Med. 1999;159:2004–9.
Perry Jr HM, Davis BR, Price TR, et al. Effect of treating isolated systolic hypertension on the risk of developing various types and subtypes of stroke: the Systolic Hypertension in the Elderly Program (SHEP). JAMA. 2000;284:465–71.
Benetos A, Salvi P, Lacolley P. Blood pressure regulation during the aging process: the end of the ‘hypertension era’? J Hypertens. 2011;29:646–52.
Safar ME, Levy BI, Struijker-Boudier H. Current perspectives on arterial stiffness and pulse pressure in hypertension and cardiovascular disease. Circulation. 2003;107:2864–9.
O’Rourke MF, Frolich ED. Pulse pressure: is this a clinically useful risk factor ? Hypertension. 1999;34:372–4.
Lakatta E. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: part III: cellular and molecular clues to heart and arterial aging. Circulation. 2003;107:490–7.
Benetos A, Adamopoulos C, Bureau JM, et al. Determinants of accelerated progression of arterial stiffness in normotensive subjects and in treated hypertensive subjects over a 6-year period. Circulation. 2002;105:1202–7.
Benetos A, Buatois S, Salvi P, et al. Blood pressure and pulse wave velocity values in the institutionalized elderly aged 80 and over: baseline of the PARTAGE Study. J Hypertens. 2010;28:41–50.
Ronnback M, Fagerudd J, Forsblom C, Pettersson-Fernholm K, Reunanen A, Groop PH, on behalf of Finnish Diabetic Nephropathy Study Group. Altered age-related blood pressure pattern in type 1 diabetes. Circulation. 2004;110:1076–82.
Salomaa V, Riley W, Kark JD, Nardo C, Folsom AR. Non-insulin dependent diabetes mellitus and fasting glucose and insulin concentrations are associated with arterial stiffness indexes. ARIC Study Circulation. 1995;91:1432–43.
Schram MT, Kostense PJ, Van Dijk RA, et al. Diabetes, pulse pressure and cardiovascular mortality: the Hoorn Study. J Hypertens. 2002;20:1743–51.
Franklin SS, Larson MG, Khan SA, et al. Does the relation of blood pressure to coronary heart disease risk change with aging? Framingham Heart Study Circulation. 2001;103:1245–9.
European Society of Hypertension-European Society of Cardiology Guidelines Committee. 2003 European Society of Hypertension–European Society of Cardiology Guidelines Committee. Guidelines for the management of arterial hypertension. J Hypertens. 2003;21:1011–53.
Darne B, Girerd X, Safar M, Cambien F, Guize L. Pulsatile versus steady component of blood pressure: a cross-sectional analysis and a prospective analysis on cardiovascular mortality. Hypertension. 1989;13:392–400.
Mitchell GF, Moye LA, Braunwald E, et al. for the SAVE Investigators. Sphygmomanometric determined pulse pressure is a powerful independent predictor of recurrent events after myocardial infarction in patients with impaired left ventricular function. Circulation. 1997;96:4254–60.
Chae CU, Pfeffer MA, Glynn RJ, Mitchell GF, Taylor JO, Hennekens CH. Increased pulse pressure and risk of heart failure in the elderly. JAMA. 1999;281:634–9.
Benetos A, Safar M, Rudnichi A, et al. Pulse pressure, a predictor of long-term cardiovascular mortality. Hypertension. 1997;30:1410–5.
Kengne AP, Czernichow S, Huxley R, et al. Blood pressure variables and cardiovascular risk new findings from ADVANCE. Hypertension. 2009;54:399–404.
Bangalore S, Messerli FH, Franklin SS, Mancia G, Champion A, Pepine CJ. Pulse pressure and risk of cardiovascular outcomes in patients with hypertension and coronary artery disease: an INternational VErapamil SR-trandolapril Study (INVEST) analysis. Eur Heart J. 2009;30:1395–401.
Miura K, Nakagawa H, Ohashi Y, et al. Four blood pressure indexes and the risk of stroke and myocardial infarction in Japanese men and women. A meta-analysis of 16 cohort studies. Circulation. 2009;119:1892–8.
Benetos A, Rudnichi A, Safar M, Guize L. Pulse pressure and cardiovascular mortality in normotensive and hypertensive subjects. Hypertension. 1998;32:560–4.
de Simone G, Roman MJ, Alderman MH, Galderisi M, de Divitiis O, Devereux RB. Is high pulse pressure a marker of preclinical cardiovascular disease? Hypertension. 2005;45:575–9.
Gasowski J, Fagard RH, Staessen JA, et al. INDANA Project Collaborators. Pulsatile blood pressure component as predictor of mortality in hypertension: a meta-analysis of clinical trial control groups. J Hypertens. 2002;20:145–51.
Nichols WW, O’Rourke MF. McDonald’s blood flow in arteries. Theoretical, experimental and clinical principles. 5th ed. London: Edward Arnold; 2005.
Safar ME, Smulyan H. Coronary ischemic disease, arterial stiffness, and pulse pressure. Am J Hypertens. 2004;17:724–6.
Kass DA. Ventricular arterial stiffening: integrating the pathophysiology. Hypertension. 2005;46:185–93.
Lloyd-Jones DM, Evans JC, Levy D. Hypertension in adults across the age spectrum: current outcomes and control in the community. JAMA. 2005;294:466–72.
Benetos A, Gautier S, Labat C, et al. Mortality and cardiovascular events are best predicted by low central/peripheral pulse pressure amplification but not by high blood pressure levels in elderly nursing home subjects: the PARTAGE (Predictive Values of Blood Pressure and Arterial Stiffness in Institutionalized Very Aged Population) study. J Am Coll Cardiol. 2012;60:1503–11.
Skoog I, Lernfelt B, Landahl S, et al. 15-year longitudinal study of blood pressure and dementia. Lancet. 1996;347:1141–5.
Vanhanen H, Thijs L, Birkenhager W, Bulpitt C, Tilvis R, Sarti C, Tuomilehto J, Staessen JA. Prevalence and persistency of orthostatic blood pressure fall in older patients with isolated systolic hypertension. Syst-Eur Investigators. Hum Hypertens. 1996;10:607–12.
Staessen JA, Thijs L, O’Brien ET, et al. Ambulatory pulse pressure as predictor of outcome in older patients with systolic hypertension. Am J Hypertens. 2002;15:835–43.
Staessen JA, Thijs L, Fagard R, et al. Predicting cardiovascular risk using conventional vs ambulatory blood pressure in older patients with systolic hypertension. JAMA. 1999;282:539–46.
Fagard RH, Staessen JA, Thijs L, et al. Response to antihypertensive therapy in older patients with sustained and nonsustained systolic hypertension. Circulation. 2000;102:1139–44.
Bobrie G, Chatellier G, Genes N, Clerson P, Vaur L, Vaisse B, Menard J, Mallion JM. Cardiovascular prognosis of “masked hypertension” detected by blood pressure self-measurement in elderly treated hypertensive patients. JAMA. 2004;291:1342–9.
Björklund K, Lind L, Zethelius B, Berglund L, Lithell H. Prognostic significance of 24-h ambulatory blood pressure characteristics for cardiovascular morbidity in a population of elderly men. J Hypertens. 2004;22:1691–7.
Asayama K, Ohkubo T, Kikuya M, Metoki H, Hoshi H, Hashimoto J, Totsune K, Satoh H, Imai Y. Prediction of stroke by self-measurement of blood pressure at home versus casual screening blood pressure measurement in relation to the Joint National Committee 7 classification: the Ohasama study. Stroke. 2004;35:2356–61.
Mac Mahon M, Sheahan NF, Colgan MP, Walsh B, Malone J, Coakley D. Arterial closing pressure correlates with diastolic pseudohypertension in the elderly. J Gerontol A Biol Sci Med Sci. 1995;50A:56–8.
Franklin SS, Wilkinson IB, McEniery CM. Unusual hypertensive phenotypes: what is their significance? Hypertension. 2012;59:173–8.
Clinical practice guidelines: management of adults with essential hypertension- 2005 update – Société française d’hypertension artérielle. Haute Autorité de Santé HAS July 2005. Management of adults with essential hypertension-2005 update-guidelines. J Mal Vasc. 2006;31:16–33.
The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC) 2007. Guidelines for the management of arterial hypertension. J Hypertens. 2007;25:1105–87.
Mancia G, Laurent S, Agabiti-Rosei E. Reappraisal of European guidelines on hypertension management: a European Society of Hypertension Task Force document. J Hypertens. 2009;27:2121–58.
Burt VL, Cutler JA, Higgins M, et al. Trends in the prevalence, awareness, treatment, and control of hypertension in the adult US population: data from the Health Examination Surveys, 1960 to 1991. Hypertension. 1995;26:60–9.
Marques-Vidal P, Tuomilehto J. Hypertension awareness, treatment and control in the community: is the “rule of halves” still valid? J Hum Hypertens. 1997;11:213–20.
Colhoun HM, Dong W, Poulter NR. Blood pressure screening, management and control in England: results from the health survey for England 1994. J Hypertens. 1998;16:747–52.
Perret-Guillaume C, Miget P, Aubry C, Gueguen R, Steyer E, Benetos A. Contrôle de la pression artérielle par le traitement antihypertenseur chez le sujet de plus de 60 ans. Rev Med Interne. 2006;27:285–90.
Benetos A, Thomas F, Bean K, Gautier S, Smulyan H, Guize L. Prognostic value of systolic and diastolic pressure in treated hypertensive men. Arch Intern Med. 2002;162:577–81.
Chobanian AV. The hypertension paradox – More uncontrolled disease despite improved therapy. N Engl J Med. 2009;361:878–87.
Kannel WB, Wilson PWF, Nam BH, D’Agostino RB, Li J. A likely explanation for the J-curve of blood pressure cardiovascular risk. Am J Cardiol. 2004;94:380–4.
Messerli FH, Mancia G, Conti CR, et al. Dogma disputed: can aggressively lowering blood pressure in hypertensive patients with coronary artery disease be dangerous? Ann Intern Med. 2006;44:884–93.
Messerli FH, Panjrath GS. The J-curve between blood pressure and coronary artery disease or essential hypertension: exactly how essential? J Am Coll Cardiol. 2009;54:1827–34.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag London
About this chapter
Cite this chapter
Benetos, A., Gautier, S., Safar, M.E. (2014). Modifications of Blood Pressure Profiles in the Very Old: Role of Frailty and Comorbidities. In: Safar, M., O'Rourke, M., Frohlich, E. (eds) Blood Pressure and Arterial Wall Mechanics in Cardiovascular Diseases. Springer, London. https://doi.org/10.1007/978-1-4471-5198-2_30
Download citation
DOI: https://doi.org/10.1007/978-1-4471-5198-2_30
Published:
Publisher Name: Springer, London
Print ISBN: 978-1-4471-5197-5
Online ISBN: 978-1-4471-5198-2
eBook Packages: MedicineMedicine (R0)