Abstract
Insulin-like growth factor-1 (IGF-1) and IGF-2 are circulating peptide growth factor hormones important in normal growth and development. Their biological effects are mediated by specific cell surface receptors, the type I IGF receptor (IGF1R) and the insulin receptor. In cancer, preclinical and epidemiological evidence support a role for these growth factors in regulating cancer risk and tumor biology. Thus, neutralization of these growth factors could play a role in cancer prevention and therapy.
Access provided by CONRICYT-eBooks. Download reference work entry PDF
Similar content being viewed by others
Keywords
Target: Insulin-Like Growth Factor I and II (IGF-I and IGF-II)
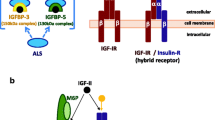
IGF-I and IGF-II are polypeptide hormones important in normal growth and development (Baker et al. 1993). As their name implies, they are homologous in structure to insulin (Bell et al. 1984; Jansen et al. 1983). Like insulin, both hormones interact with specific heterotetrameric transmembrane tyrosine kinase receptors. IGF-I has the highest affinity for the type I IGF receptor (IGF1R). IGF-II can activate this receptor but also has high affinity for the fetal form of the insulin receptor (insulin receptor A, IRA) (Frasca et al. 1999).
Biology of the Target
IGF-I expression is increased during puberty by action of growth hormone on the liver (Mauras 2001). Once produced, IGF-I circulates bound to a high-affinity binding protein (IGF binding protein-3) and is further stabilized by the “acid labile subunit” in a ternary complex (Martin and Baxter 2011; Domene et al. 2009). High levels of serum IGF-I are found in adult life, but most of it is bound in the ternary complex and not available for receptor binding. During periods of stress, IGF binding protein-specific protease can cleave the binding protein and release IGF-I to interact with receptors. Thus, adults have a substantial reservoir of IGF-I in the circulation. IGF-I can also be found in the bone matrix in complex with other IGF binding proteins (Govoni et al. 2005).
IGF-II has an important function in fetal growth. In mice, IGF-II is necessary for normal fetal development. In rodents, IGF-II levels drop shortly after birth. However, humans maintain high levels of IGF-II during adult life and its function is not understood.
IGF1R activation requires binding by either IGF-I or IGF-II to signal. Thus, ligand production and receptor binding are necessary for activation of this receptor system.
Target Assessment
IGF-I and IGF-II can be measured by commercially available ELISA assays (Frystyk et al. 2010). “Free” IGF-I (unbound to IGF binding proteins) has been reported to predict outcome in some clinical trials of anti-IGF1R drugs. This assay is not commercially available.
Role of the Target in Cancer
Rank: 5
High-Level Overview
Diagnostic, Prognostic, and Predictive
The level of IGF-I and insulin is associated with increased cancer risk (Vigneri et al. 2009; Renehan et al. 2004). In preliminary reports, levels of free IGF-I are associated with benefit from IGF1R inhibition by the monoclonal antibody figitumumab in non-small cell lung cancer trials (Gualberto et al. 2009).
Therapeutics
Since hepatic IGF-I is under the control of growth hormone, disruption of growth hormone action could be a cancer therapy. This possibility is supported by the clinical observation that humans with mutation in growth hormone receptor are unable to respond to growth hormone, have low levels of IGF-I and IGF-II, and rarely, if ever, get cancer. Strategies to disrupt growth hormone releasing hormone or growth hormone receptor have been described (Schally et al. 2008; Divisova et al. 2006). A polyethylene glycol-conjugated mutant of growth hormone is available for treatment of growth hormone access (acromegaly). This drug, pegvisomant, has been evaluated in normal subjects (Yin et al. 2007). However, its development in cancer was discontinued by the manufacturer.
Monoclonal antibodies that bind both IGF-I and IGF-II have been described (Dransfield et al. 2010; Goya et al. 2004). A ligand-binding antibody is currently in phase I clinical trial (NCT00816361).
Preclinical Summary
Substantial preclinical data in cell culture and mouse model systems have shown that IGF-I is a potent mitogen for cancer cells. Expression of IGF-I can accelerate tumorigenesis in mouse model systems (Kleinberg et al. 2008). IGF-II is an imprinted gene. Loss of imprinting allowing biallelic expression has been linked to cancer development and progression (Hu et al. 2011; Kaneda et al. 2007).
Anticipated High Impact Results
Publication of phase I and II results from IGF-I and IGF-II neutralizing antibody clinical trials.
References
Baker J, Liu JP, Robertson EJ, Efstratiadis A. Role of insulin-like growth factors in embryonic and postnatal growth. Cell. 1993;75:73–82.
Bell GI, Merryweather JP, Sanchez-Pescador R, Stempien MM, Priestley L, Scott J, Rall LB. Sequence of a cDNA clone encoding human preproinsulin-like growth factor II. Nature. 1984;310:775–7.
Divisova J, Kuiatse I, Lazard Z, Weiss H, Vreeland F, Hadsell DL, Schiff R, Osborne CK, Lee AV. The growth hormone receptor antagonist pegvisomant blocks both mammary gland development and MCF-7 breast cancer xenograft growth. Breast Cancer Res Treat. 2006;98:315–27.
Domene HM, Hwa V, Argente J, Wit JM, Camacho-Hubner C, Jasper HG, Pozo J, van Duyvenvoorde HA, Yakar S, Fofanova-Gambetti OV, Rosenfeld RG. Human acid-labile subunit deficiency: clinical, endocrine and metabolic consequences. Horm Res. 2009;72:129–41.
Dransfield DT, Cohen EH, Chang Q, Sparrow LG, Bentley JD, Dolezal O, Xiao X, Peat TS, Newman J, Pilling PA, Phan T, Priebe I, Brierley GV, Kastrapeli N, Kopacz K, Martik D, Wassaf D, Rank D, Conley G, Huang Y, Adams TE, Cosgrove L. A human monoclonal antibody against insulin-like growth factor-II blocks the growth of human hepatocellular carcinoma cell lines in vitro and in vivo. Mol Cancer Ther. 2010;9:1809–19.
Frasca F, Pandini G, Scalia P, Sciacca L, Mineo R, Costantino A, Goldfine ID, Belfiore A, Vigneri R. Insulin receptor isoform A, a newly recognized, high-affinity insulin- like growth factor II receptor in fetal and cancer cells. Mol Cell Biol. 1999;19:3278–88.
Frystyk J, Freda P, Clemmons DR. The current status of IGF-I assays – a 2009 update. Growth Horm IGF Res. 2010;20:8–18.
Govoni KE, Baylink DJ, Mohan S. The multi-functional role of insulin-like growth factor binding proteins in bone. Pediatr Nephrol. 2005;20:261–8.
Goya M, Miyamoto S, Nagai K, Ohki Y, Nakamura K, Shitara K, Maeda H, Sangai T, Kodama K, Endoh Y, Ishii G, Hasebe T, Yonou H, Hatano T, Ogawa Y, Ochiai A. Growth inhibition of human prostate cancer cells in human adult bone implanted into nonobese diabetic/severe combined immunodeficient mice by a ligand-specific antibody to human insulin-like growth factors. Cancer Res. 2004;64:6252–8.
Gualberto A, Dolled-Filhart M, Hixon M, Christensen J, Rimm D, Lee A, Wang Y, Pollak M, Paz-Ares L, Karp D. Molecular bases for sensitivity to figitumumab (CP-751,871) in NSCLC. J Clin Oncol. 2009;27:Abstr 8091.
Guevara-Aguirre J, Balasubramanian P, Guevara-Aguirre M, Wei M, Madia F, Cheng CW, Hwang D, Martin-Montalvo A, Saavedra J, Ingles S, de Cabo R, Cohen P, Longo VD. Growth hormone receptor deficiency is associated with a major reduction in pro-aging signaling, cancer, and diabetes in humans. Sci Transl Med. 2011;3:70ra13.
Hu Q, Gao F, Tian W, Ruteshouser EC, Wang Y, Lazar A, Stewart J, Strong LC, Behringer RR, Huff V. Wt1 ablation and Igf2 upregulation in mice result in Wilms tumors with elevated ERK1/2 phosphorylation. J Clin Invest. 2011;121:174–83.
Jansen M, van Schaik FM, Ricker AT, Bullock B, Woods DE, Gabbay KH, Nussbaum AL, Sussenbach JS, Van den Brande JL. Sequence of cDNA encoding human insulin-like growth factor I precursor. Nature. 1983;306:609–11.
Kaneda A, Wang CJ, Cheong R, Timp W, Onyango P, Wen B, Iacobuzio-Donahue CA, Ohlsson R, Andraos R, Pearson MA, Sharov AA, Longo DL, Ko MS, Levchenko A, Feinberg AP. Enhanced sensitivity to IGF-II signaling links loss of imprinting of IGF2 to increased cell proliferation and tumor risk. Proc Natl Acad Sci U S A. 2007;104:20926–31.
Kleinberg DL, Wood TL, Furth PA, Lee AV. Growth hormone and insulin-like growth factor-I in the transition from normal mammary development to preneoplastic mammary lesions. Endocr Rev. 2008;30:51–74.
Martin JL, Baxter RC. Signalling pathways of insulin-like growth factors (IGFs) and IGF binding protein-3. Growth Factors. 2011;29:235–44.
Mauras N. Growth hormone and sex steroids. Interactions in puberty. Endocrinol Metab Clin North Am. 2001;30:529–44.
Renehan AG, Zwahlen M, Minder C, O’Dwyer ST, Shalet SM, Egger M. Insulin-like growth factor (IGF)-I, IGF binding protein-3, and cancer risk: systematic review and meta-regression analysis. Lancet. 2004;363:1346–53.
Schally AV, Varga JL, Engel JB. Antagonists of growth-hormone-releasing hormone: an emerging new therapy for cancer. Nat Clin Pract Endocrinol Metab. 2008;4:33–43.
Steuerman R, Shevah O, Laron Z. Congenital IGF-I deficiency tends to confer protection against post-natal development of malignancies. Eur J Endocrinol. 2011;164:485–9.
Vigneri P, Frasca F, Sciacca L, Pandini G, Vigneri R. Diabetes and cancer. Endocr Relat Cancer. 2009;16:1103–23.
Yin D, Vreeland F, Schaaf LJ, Millham R, Duncan BA, Sharma A. Clinical pharmacodynamic effects of the growth hormone receptor antagonist pegvisomant: implications for cancer therapy. Clin Cancer Res. 2007;13:1000–9.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media New York
About this entry
Cite this entry
Yee, D. (2017). IGF 1 and IGF 2. In: Marshall, J. (eds) Cancer Therapeutic Targets. Springer, New York, NY. https://doi.org/10.1007/978-1-4419-0717-2_4
Download citation
DOI: https://doi.org/10.1007/978-1-4419-0717-2_4
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4419-0716-5
Online ISBN: 978-1-4419-0717-2
eBook Packages: Biomedical and Life SciencesReference Module Biomedical and Life Sciences