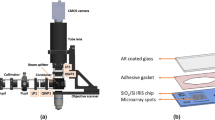
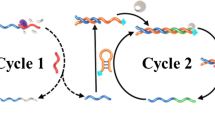
Abstract
Quantification of circulating microRNAs (miRNAs) or viral RNAs is of great significance because of their broad relevance to human health. Currently, quantitative reverse transcription polymerase chain reaction (qRT-PCR), as well as microarray and gene sequencing, are considered mainstream techniques for miRNA identification and quantitation and the gold standard for SARS-CoV2 detection in the COVID-19 pandemic. However, these laboratory techniques are challenged by the low levels and wide dynamic range (from aM to nM) of miRNAs in a physiological sample, as well as the difficulty in the implementation in point-of-care settings. Here, we describe a one-step label-free electrochemical sensing technique by assembling self-folded multi-stem DNA-redox probe structure on gold microelectrodes and introducing a reductant, tris(2-carboxyethyl) phosphine hydrochloride (TCEP), in the detection buffer solution to achieve ultrasensitive detection with a detection limit of 0.1 fM that can be further improved if needed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Dong H, Lei J, Ding L, Wen Y, Ju H, Zhang X (2013) MicroRNA: function, detection, and bioanalysis. Chem Rev 113:6207–6233
Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120:15–20
Cullen BR (2009) Viral and cellular messenger RNA targets of viral microRNAs. Nature 457:421–425
Laterza OF, Lim L, Garrett-Engele PW, Vlasakova K, Muniappa N, Tanaka WK, Johnson JM, Sina JF, Fare TL, Sistare FD, Glaab WE (2009) Plasma MicroRNAs as sensitive and specific biomarkers of tissue injury. Clin Chem 55:1977–1983
Hayes J, Peruzzi PP, Lawler S (2014) MicroRNAs in cancer: biomarkers, functions and therapy. Trends Mol Med 20:460–469
Wang K, Zhang S, Marzolf B, Troisch P, Brightman A, Hu Z (2009) Circulating microRNAs, potential biomarkers for drug-induced liver injury. Proc Natl Acad Sci U S A 106:4402–4407
Michel V, Yuan Z, Ramsubir S, Bakovic M (2006) Choline transport for phospholipid synthesis. Exp Bio Med 231:490–504
Klein D (2002) Quantification using real-time PCR technology: applications and limitations. Trends Mol Med 8:257–260
Lim LP (2003) The microRNAs of Caenorhabditis elegans. Genes Dev 17:991–1008
Liang Y, Ridzon D, Wong L, Chen C (2007) Characterization of microRNA expression profiles in normal human tissues. BMC Genomics 8:166
Liu CG, Calin GA, Meloon B, Gamllel N, Sevignani C, Ferracin M, Dumitru CD, Shimizu M, Zupo S, Dono M, Alder H, Bullrich F, Negrini M, Croce CM (2004) An oligonucleotide microchip for genome-wide microRNA profiling in human and mouse tissues. Proc Natl Acad Sci U S A 101:9740–9744
Lodes MJ, Caraballo M, Suciu D, Munro S, Kumar A, Anderson B (2009) Detection of cancer with serum miRNAs on an oligonucleotide microarray. PLoS One 4:e6229
Asaga S, Kuo C, Nguyen T, Terpenning M, Giuliano AE, Hoon DSB (2011) Direct serum assay for MicroRNA-21 concentrations in early and advanced breast cancer. Clin Chem 57:84–91
Zhi F, Cao X, Xie X, Wang B, Dong W, Gu W, Ling Y, Wang R, Yang Y, Liu Y (2013) Identification of circulating MicroRNAs as potential biomarkers for detecting acute myeloid leukemia. PLoS One 8:e56718
Gaur A, Jewell DA, Liang Y, Ridzon D, Moore JH, Chen C, Ambros VR, Israel MA (2007) Characterization of microRNA expression levels and their biological correlates in human cancer cell lines. Cancer Res 67:2456–2468
Zhang J, Li Z, Wang H, Wang Y, Jia H, Yan J (2011) Ultrasensitive quantification of mature microRNAs by real-time PCR based on ligation of a ribonucleotide-modified DNA probe. Chem Commun 47:9465–9467
Gao ZQ, Yang ZC (2006) Detection of microRNAs using electrocatalytic nanoparticle tags. Anal Chem 78:1470–1477
Labib M, Khan N, Ghobadloo SM, Cheng J, Pezacki JP, Berezovski MV (2013) Three-mode electrochemical sensing of ultralow MicroRNA levels. J Am Chem Soc 135:3027–3038
Labib M, Ghobadloo SM, Khan N, Kolpashchikov DM, Berezovski MV (2013) Four-way junction formation promoting ultrasensitive electrochemical detection of MicroRNA. Anal Chem 85:9422–9427
Labib M, Khan N, Berezovski MV (2015) Protein Electrocatalysis for direct sensing of circulating MicroRNAs. Anal Chem 87:1395–1403
Idili A, Parolo C, Alvarez-Diduk R, Merkoçi A (2021) Rapid and efficient detection of the SARS-CoV-2 spike protein using an electrochemical aptamer-based sensor. ACS Sensors 6(8):3093–3101
Alafeef M, Dighe K, Moitra P, Pan D (2020) Rapid, ultrasensitive, and quantitative detection of SARS-CoV-2 using antisense oligonucleotides directed electrochemical biosensor chip. ACS Nano 14(12):17028–17045
Kumar N, Shetti NP, Jagannath S, Aminabhavi TM (2022) Electrochemical sensors for the detection of SARS-CoV-2 virus. Chem Eng J 430:132966
Wang T, Viennois E, Merlin D, Wang G (2015) Microelectrode miRNA sensors enabled by enzyme less electrochemical signal amplification. Anal Chem 87:8173–8180
White RJ, Plaxco KW (2010) Exploiting binding-induced changes in probe flexibility for the optimization of electrochemical biosensors. Anal Chem 82:73–76
Acknowledgements
The microRNA work was supported by grants from the Department of Veterans Affairs (Merit Award: BX002526 to D. M.) and the National Institute of Health of Diabetes and Digestive and Kidney by the Grants RO1-DK-071594 and RO1-DK-064711 (to D. M.). Updated procedures and viral RNA analysis were developed under the financial support of NSF RAPID CHE-2034498.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Ake, S., Kamila, S., Wang, G. (2023). Quantification of MicroRNAs or Viral RNAs with Microelectrode Sensors Enabled by Electrochemical Signal Amplification. In: Dalmay, T. (eds) MicroRNA Detection and Target Identification. Methods in Molecular Biology, vol 2630. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2982-6_9
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2982-6_9
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2981-9
Online ISBN: 978-1-0716-2982-6
eBook Packages: Springer Protocols