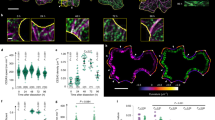
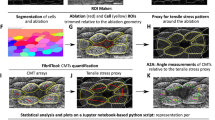
Abstract
Leaf epidermis pavement cells form highly complex shapes with interlocking lobes and necks at their anticlinal walls. The microtubule cytoskeleton plays essential roles in pavement cell morphogenesis, in particular at necks. Vice versa, shape generates stress patterns that regulate microtubule organization. Genetic or pharmacological perturbations that affect pavement cell shape often affect microtubule organization. Pavement cell shape and microtubule organization are therefore closely interconnected. Here, we present commonly used approaches for the quantitative analysis of pavement cell shape characteristics and of microtubule organization. In combination with ablation experiments, these methods can be applied to investigate how different genotypes (or treatments) affect the organization and stress responsiveness of the microtubule cytoskeleton.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jacques E, Verbelen JP, Vissenberg K (2014) Review on shape formation in epidermal pavement cells of the Arabidopsis leaf. Funct Plant Biol 41:914–921
Vofely RV, Gallagher J, Pisano GD, Bartlett M, Braybrook SA (2019) Of puzzles and pavements: a quantitative exploration of leaf epidermal cell shape. New Phytol 221:540–552
Zhang CH, Halsey LE, Szymanski DB (2011) The development and geometry of shape change in Arabidopsis thaliana cotyledon pavement cells. BMC Plant Biol 11
Sampathkumar A, Krupinski P, Wightman R, Milani P, Berquand A, Boudaoud A, Hamant O, Jonsson H, Meyerowitz EM (2014) Subcellular and supracellular mechanical stress prescribes cytoskeleton behavior in Arabidopsis cotyledon pavement cells. elife 3
Majda M, Grones P, Sintorn IM, Vain T, Milani P, Krupinski P, Zagorska-Marek B, Viotti C, Jonsson H, Mellerowicz EJ et al (2017) Mechanochemical polarization of contiguous cell walls shapes plant pavement cells. Dev Cell 43:290–304
Belteton SA, Li WL, Yanagisawa M, Hatam FA, Quinn MI, Szymanski MK, Marley MW, Turner JA, Szymanski DB (2021) Real-time conversion of tissue-scale mechanical forces into an interdigitated growth pattern. Nat Plants 7:826–841
Eng RC, Schneider R, Matz TW, Carter R, Ehrhardt DW, Jonsson H, Nikoloski Z, Sampathkumar A (2021) KATANIN and CLASP function at different spatial scales to mediate microtubule response to mechanical stress in Arabidopsis cotyledons. Curr Biol 31:3262–3274
Wong JH, Kato T, Belteton SA, Shimizu R, Kinoshita N, Higaki T, Sakumura Y, Szymanski DB, Hashimoto T (2019) Basic Proline-rich protein-mediated microtubules are essential for lobe growth and flattened cell geometry. Plant Physiol 181:1535–1551
Mitra D, Kumari P, Quegwer J, Klemm S, Möller B, Poeschl Y, Pflug P, Stamm G, Abel S, Bürstenbinder K (2019) Microtubule-associated protein IQ67 DOMAIN5 regulates morphogenesis of leaf pavement cells in Arabidopsis thaliana. J Exp Bot 70:529–543
Lin W, Tang W, Pan X, Huang A, Gao X, Anderson CT, Yang Z (2022) Arabidopsis pavement cell morphogenesis requires FERONIA binding to pectin for activation of ROP GTPase signaling. Curr Biol 32(497–507):e494
Tang W, Lin W, Zhou X, Guo J, Dang X, Li B, Lin D, Yang Z (2022) Mechano-transduction via the pectin-FERONIA complex activates ROP6 GTPase signaling in Arabidopsis pavement cell morphogenesis. Curr Biol 32(508–517):e503
Lauster T, Stöckle D, Gabor K, Haller T, Krieger N, Lotz P, Mayakrishnan R, Späth E, Zimmermann S, Livanos P et al (2022) Arabidopsis pavement cell shape formation involves spatially confined ROPGAP regulators. Curr Biol 32:532–544
Zhang C, Lauster T, Tang W, Houbaert A, Zhu SF, Eeckhout D, De Smet I, De Jaeger G, Jacobs TB, Xu T et al (2022) ROPGAP-dependent interaction between brassinosteroid and ROP2-GTPase signaling controls pavement cell shape in Arabidopsis. Curr Biol 32:518–531
Landrein B, Hamant O (2013) How mechanical stress controls microtubule behavior and morphogenesis in plants: history, experiments and revisited theories. Plant J 75:324–338
Belteton SA, Li WL, Yanagisawa M, Hatam FA, Quinn MI, Szymanski MK, Marley MW, Turner JA, Szymanski DB (2021) Real-time conversion of tissue-scale mechanical forces into an interdigitated growth pattern. Nat Plants 7:989–989
Boudaoud A, Burian A, Borowska-Wykret D, Uyttewaal M, Wrzalik R, Kwiatkowska D, Hamant O (2014) FibrilTool, an ImageJ plug-in to quantify fibrillar structures in raw microscopy images. Nat Protoc 9:457–463
Püspöki Z, Storath M, Sage D, Unser M (2016) Transforms and operators for directional bioimage analysis: a survey. Adv Anat Embryol Cell Biol 219:69–93
Celler K, Fujita M, Kawamura E, Ambrose C, Herburger K, Holzinger A, Wasteneys GO (2016) Microtubules in plant cells: strategies and methods for immunofluorescence, transmission electron microscopy, and live cell imaging. Methods Mol Biol 1365:155–184
Lincoln C, Britton JH, Estelle M (1990) Growth and development of the axr1 mutants of Arabidopsis. Plant Cell 2:1071–1080
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B et al (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682
Möller B, Glaß M, Misiak D, Posch S (2016) MiToBo - a toolbox for image processing and analysis. J Open Res Software 4:e17
Wu TC, Belteton S, Pack J, Szymanski DB, Umulis D (2016) LobeFinder: a convex hull-based method for quantitative boundary analyses of lobed plant cells. Plant Physiol 171:2331–2342
Nowak J, Eng RC, Matz T, Waack M, Persson S, Sampathkumar A, Nikoloski Z (2021) A network-based framework for shape analysis enables accurate characterization of leaf epidermal cells. Nat Commun 12
Möller B, Poeschl Y, Plötner R, Bürstenbinder K (2017) PaCeQuant: a tool for high-throughput quantification of pavement cell shape characteristics. Plant Physiol 175:998–1017
Möller B, Poeschl Y, Klemm S, Bürstenbinder K (2019) Morphological analysis of leaf epidermis pavement cells with PaCeQuant. Methods Mol Biol 1992:329–349
Poeschl Y, Möller B, Müller L, Bürstenbinder K (2020) User-friendly assessment of pavement cell shape features with PaCeQuant: novel functions and tools. Methods Cell Biol 160:349–363
Möller B, Zergiebel L, Bürstenbinder K (2019) Quantitative and comparative analysis of global patterns of (microtubule) cytoskeleton organization with CytoskeletonAnalyzer2D. Methods Mol Biol 1992:151–171
Bürstenbinder K, Möller B, Plötner R, Stamm G, Hause G, Mitra D, Abel S (2017) The IQD family of calmodulin-binding proteins links calcium signaling to microtubules, membrane subdomains, and the nucleus. Plant Physiol 173:1692–1708
Freeman H, Davis LS (1977) A corner-finding algorithm for chain-coded curves. Ieee T Comput 26:297–303
Belteton SA, Sawchuk MG, Donohoe BS, Scarpella E, Szymanski DB (2018) Reassessing the roles of PIN proteins and anticlinal microtubules during pavement cell morphogenesis. Plant Physiol 176:432–449
Acknowledgments
Katharina Bürstenbinder would like to thank the Deutsche Forschungsgemeinschaft (DFG) (grant numbers BU2955/2-1 and BU2955/1-2), the German-Israeli Foundation for Scientific Research and Development (GIF) (grant number G-1482-423.13/2018), and the Leibniz Association for funding.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Klemm, S., Buhl, J., Möller, B., Bürstenbinder, K. (2023). Quantitative Analysis of Microtubule Organization in Leaf Epidermis Pavement Cells. In: Hussey, P.J., Wang, P. (eds) The Plant Cytoskeleton. Methods in Molecular Biology, vol 2604. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2867-6_4
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2867-6_4
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2866-9
Online ISBN: 978-1-0716-2867-6
eBook Packages: Springer Protocols