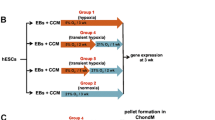
Abstract
Cartilage resides under a low oxygen tension within articulating joints. The oxygen tension within cartilage of the knee joint has been measured to be between 2% and 5% oxygen. Although the literature has historically termed this level of oxygen as hypoxia, particularly when doing experiments in vitro in this range, this is actually the physiological oxygen tension experienced in vivo and is more accurately termed physioxia. In general, culture of chondrogenic cells under physioxia has demonstrated a donor-dependent beneficial effect on chondrogenesis, with an upregulation in cartilage genes (SOX9, COL2A1, ACAN) and matrix deposition (sulfated glycosaminoglycans (sGAGs), collagen II). Physioxia also reduces the expression of hypertrophic markers (COL10A1, MMP13). This chapter will outline the methods for the expansion and differentiation of chondrogenic cells under physioxia using oxygen-controlled incubators and glove box environments, with the typical assays used for qualitative and quantitative assessment of chondrogenesis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Lafont JE (2010) Lack of oxygen in articular cartilage: consequences for chondrocyte biology. Int J Exp Pathol 91(2):99–106. https://doi.org/10.1111/j.1365-2613.2010.00707.x
Brighton CT, Heppenstall RB (1971) Oxygen tension in zones of the epiphyseal plate, the metaphysis and diaphysis. An in vitro and in vivo study in rats and rabbits. J Bone Joint Surg Am 53(4):719–728
Lund-Olesen K (1970) Oxygen tension in synovial fluids. Arthritis Rheum 13(6):769–776
Grant JL, Smith B (1963) Bone marrow gas tensions, bone marrow blood flow, and erythropoiesis in man. Ann Intern Med 58:801–809
Strobel S, Loparic M, Wendt D et al (2010) Anabolic and catabolic responses of human articular chondrocytes to varying oxygen percentages. Arthritis Res Ther 12(2):R34. https://doi.org/10.1186/ar2942
Markway BD, Cho H, Johnstone B (2013) Hypoxia promotes redifferentiation and suppresses markers of hypertrophy and degeneration in both healthy and osteoarthritic chondrocytes. Arthritis Res Ther 15(4):R92. https://doi.org/10.1186/ar4272
Egli RJ, Bastian JD, Ganz R et al (2008) Hypoxic expansion promotes the chondrogenic potential of articular chondrocytes. J Orthop Res 26(7):977–985. https://doi.org/10.1002/jor.20603
Acharya C, Adesida A, Zajac P et al (2012) Enhanced chondrocyte proliferation and mesenchymal stromal cells chondrogenesis in coculture pellets mediate improved cartilage formation. J Cell Physiol 227(1):88–97. https://doi.org/10.1002/jcp.22706
Bornes TD, Jomha NM, Mulet-Sierra A et al (2015) Hypoxic culture of bone marrow-derived mesenchymal stromal stem cells differentially enhances in vitro chondrogenesis within cell-seeded collagen and hyaluronic acid porous scaffolds. Stem Cell Res Ther 6:84. https://doi.org/10.1186/s13287-015-0075-4
Sheehy EJ, Buckley CT, Kelly DJ (2012) Oxygen tension regulates the osteogenic, chondrogenic and endochondral phenotype of bone marrow derived mesenchymal stem cells. Biochem Biophys Res Commun 417(1):305–310. https://doi.org/10.1016/j.bbrc.2011.11.105
Weijers EM, Van Den Broek LJ, Waaijman T et al (2011) The influence of hypoxia and fibrinogen variants on the expansion and differentiation of adipose tissue-derived mesenchymal stem cells. Tissue Eng Part A 17(21–22):2675–2685. https://doi.org/10.1089/ten.tea.2010.0661
Pattappa G, Krueckel J, Schewior R et al (2020) Physioxia expanded bone marrow derived mesenchymal stem cells have improved cartilage repair in an early osteoarthritic focal defect model. Biology (Basel) 9(8):230. https://doi.org/10.3390/biology9080230
Pattappa G, Johnstone B, Zellner J et al (2019) The importance of physioxia in mesenchymal stem cell chondrogenesis and the mechanisms controlling its response. Int J Mol Sci 20(3):484. https://doi.org/10.3390/ijms20030484
Anderson DE, Markway BD, Bond D et al (2016) Responses to altered oxygen tension are distinct between human stem cells of high and low chondrogenic capacity. Stem Cell Res Ther 7(1):154. https://doi.org/10.1186/s13287-016-0419-8
Bae HC, Park HJ, Wang SY et al (2018) Hypoxic condition enhances chondrogenesis in synovium-derived mesenchymal stem cells. Biomater Res 22:28. https://doi.org/10.1186/s40824-018-0134-x
Meyer EG, Buckley CT, Thorpe SD et al (2010) Low oxygen tension is a more potent promoter of chondrogenic differentiation than dynamic compression. J Biomech 43(13):2516–2523. https://doi.org/10.1016/j.jbiomech.2010.05.020
O’HEireamhoin S, Buckley CT, Jones E et al (2013) Recapitulating aspects of the oxygen and substrate environment of the damaged joint milieu for stem cell-based cartilage tissue engineering. Tissue Eng Part C Methods 19(2):117–127. https://doi.org/10.1089/ten.TEC.2012.0142
Bornes TD, Adesida AB, Jomha NM (2018) Articular cartilage repair with mesenchymal stem cells after chondrogenic priming: a pilot study. Tissue Eng Part A 24(9–10):761–774. https://doi.org/10.1089/ten.TEA.2017.0235
Muller J, Benz K, Ahlers M et al (2011) Hypoxic conditions during expansion culture prime human mesenchymal stromal precursor cells for chondrogenic differentiation in three-dimensional cultures. Cell Transplant 20(10):1589–1602. https://doi.org/10.3727/096368910X564094
Khan WS, Adesida AB, Hardingham TE (2007) Hypoxic conditions increase hypoxia-inducible transcription factor 2alpha and enhance chondrogenesis in stem cells from the infrapatellar fat pad of osteoarthritis patients. Arthritis Res Ther 9(3):R55. https://doi.org/10.1186/ar2211
Leijten J, Georgi N, Moreira Teixeira L et al (2014) Metabolic programming of mesenchymal stromal cells by oxygen tension directs chondrogenic cell fate. Proc Natl Acad Sci U S A 111(38):13954–13959. https://doi.org/10.1073/pnas.1410977111
Markway BD, Tan GK, Brooke G et al (2010) Enhanced chondrogenic differentiation of human bone marrow-derived mesenchymal stem cells in low oxygen environment micropellet cultures. Cell Transplant 19(1):29–42. https://doi.org/10.3727/096368909X478560
Lee HH, Chang CC, Shieh MJ et al (2013) Hypoxia enhances chondrogenesis and prevents terminal differentiation through PI3K/Akt/FoxO dependent anti-apoptotic effect. Sci Rep 3:2683. https://doi.org/10.1038/srep02683
Zhu M, Feng Q, Bian L (2014) Differential effect of hypoxia on human mesenchymal stem cell chondrogenesis and hypertrophy in hyaluronic acid hydrogels. Acta Biomater 10(3):1333–1340. https://doi.org/10.1016/j.actbio.2013.12.015
Adesida AB, Mulet-Sierra A, Jomha NM (2012) Hypoxia mediated isolation and expansion enhances the chondrogenic capacity of bone marrow mesenchymal stromal cells. Stem Cell Res Ther 3(2):9. https://doi.org/10.1186/scrt100
Pattappa G, Schewior R, Hofmeister I et al (2019) Physioxia has a beneficial effect on cartilage matrix production in interleukin-1 beta-inhibited mesenchymal stem cell chondrogenesis. Cell 8(8):936. https://doi.org/10.3390/cells8080936
Meretoja VV, Dahlin RL, Wright S et al (2013) The effect of hypoxia on the chondrogenic differentiation of co-cultured articular chondrocytes and mesenchymal stem cells in scaffolds. Biomaterials 34(17):4266–4273. https://doi.org/10.1016/j.biomaterials.2013.02.064
Portron S, Merceron C, Gauthier O et al (2013) Effects of in vitro low oxygen tension preconditioning of adipose stromal cells on their in vivo chondrogenic potential: application in cartilage tissue repair. PLoS One 8(4):e62368. https://doi.org/10.1371/journal.pone.0062368
Solchaga LA, Penick KJ, Welter JF (2011) Chondrogenic differentiation of bone marrow-derived mesenchymal stem cells: tips and tricks. Methods Mol Biol 698:253–278. https://doi.org/10.1007/978-1-60761-999-4_20
Johnstone B, Yoo J (2000) Bone marrow-derived mesenchymal progenitor cells. Methods Mol Biol 137:313–315. https://doi.org/10.1385/1-59259-066-7:313
Duval E, Leclercq S, Elissalde JM et al (2009) Hypoxia-inducible factor 1alpha inhibits the fibroblast-like markers type I and type III collagen during hypoxia-induced chondrocyte redifferentiation: hypoxia not only induces type II collagen and aggrecan, but it also inhibits type I and type III collagen in the hypoxia-inducible factor 1alpha-dependent redifferentiation of chondrocytes. Arthritis Rheum 60(10):3038–3048. https://doi.org/10.1002/art.24851
Johnstone B, Hering TM, Caplan AI et al (1998) In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res 238(1):265–272. https://doi.org/10.1006/excr.1997.3858
Yoo JU, Barthel TS, Nishimura K et al (1998) The chondrogenic potential of human bone-marrow-derived mesenchymal progenitor cells. J Bone Joint Surg Am 80(12):1745–1757
Erickson IE, Huang AH, Chung C et al (2009) Differential maturation and structure-function relationships in mesenchymal stem cell- and chondrocyte-seeded hydrogels. Tissue Eng Part A 15(5):1041–1052. https://doi.org/10.1089/ten.tea.2008.0099
Huang AH, Stein A, Tuan RS et al (2009) Transient exposure to transforming growth factor beta 3 improves the mechanical properties of mesenchymal stem cell-laden cartilage constructs in a density-dependent manner. Tissue Eng Part A 15(11):3461–3472. https://doi.org/10.1089/ten.TEA.2009.0198
Penick KJ, Solchaga LA, Welter JF (2005) High-throughput aggregate culture system to assess the chondrogenic potential of mesenchymal stem cells. BioTechniques 39(5):687–691. https://doi.org/10.2144/000112009
Markway BD, Cho H, Zilberman-Rudenko J et al (2015) Hypoxia-inducible factor 3-alpha expression is associated with the stable chondrocyte phenotype. J Orthop Res 33(11):1561–1570. https://doi.org/10.1002/jor.22930
Banfi A, Muraglia A, Dozin B et al (2000) Proliferation kinetics and differentiation potential of ex vivo expanded human bone marrow stromal cells: implications for their use in cell therapy. Exp Hematol 28(6):707–715. https://doi.org/10.1016/s0301-472x(00)00160-0
Holden P, Horton WA (2009) Crude subcellular fractionation of cultured mammalian cell lines. BMC Res Notes 2:243. https://doi.org/10.1186/1756-0500-2-243
Acknowledgements
This work was supported by the DFG Research network (FOR2407/1): ExCarBon, the European Union Horizon 2020 research and innovation program (EU No.: 814444 (MEFISTO), and a Shriners Hospitals for Children research grant.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Pattappa, G., Markway, B.D., Docheva, D., Johnstone, B. (2023). Physioxic Culture of Chondrogenic Cells. In: Stoddart, M.J., Della Bella, E., Armiento, A.R. (eds) Cartilage Tissue Engineering. Methods in Molecular Biology, vol 2598. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2839-3_5
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2839-3_5
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2838-6
Online ISBN: 978-1-0716-2839-3
eBook Packages: Springer Protocols