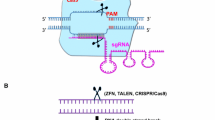
Abstract
Genetic editing of hematopoietic stem and progenitor cells can be employed to understand gene-function relationships underlying hematopoietic cell biology, leading to new therapeutic approaches to treat disease. The ability to collect, purify, and manipulate primary cells outside the body permits testing of many different gene editing approaches. RNA-guided nucleases, such as CRISPR, have revolutionized gene editing based simply on Watson-Crick base-pairing, employed to direct activity to specific genomic loci. Given the ease and affordability of synthetic, custom RNA guides, testing of precision edits or large random pools in high-throughput screening studies is now widely available. With the ever-growing number of CRISPR nucleases being discovered or engineered, researchers now have a plethora of options for directed genomic change, including single base edits, nicks or double-stranded DNA cuts with blunt or staggered ends, as well as the ability to target CRISPR to other cellular oligonucleotides such as RNA or mitochondrial DNA. Except for single base editing strategies, precise rewriting of larger segments of the genetic code requires delivery of an additional component, templated DNA oligonucleotide(s) encoding the desired changes flanked by homologous sequences that permit recombination at or near the site of CRISPR activity. Altogether, the ever-growing CRISPR gene editing toolkit is an invaluable resource. This chapter outlines available technologies and the strategies for applying CRISPR-based editing in hematopoietic stem and progenitor cells.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Broeders M et al (2020) Sharpening the molecular scissors: advances in gene-editing technology. iScience 23:100789
Ishino Y et al (1987) Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J Bacteriol 169:5429–5433
Mojica FJ et al (2005) Intervening sequences of regularly spaced prokaryotic repeats derive from foreign genetic elements. J Mol Evol 60:174–182
Anderson WF, Blaese RM, Culver K (1990) The ADA human gene therapy clinical protocol: points to consider response with clinical protocol. Hum Gene Ther 1:331–362
Bortesi L et al (2016) Patterns of CRISPR/Cas9 activity in plants, animals and microbes. Plant Biotechnol J 14:2203–2216
Avecilla ST et al (2016) How do I perform hematopoietic progenitor cell selection? Transfusion 56:1008–1012
Pickar-Oliver A, Gersbach CA (2019) The next generation of CRISPR-Cas technologies and applications. Nat Rev Mol Cell Biol 20:490–507
Xu S, Luk K, Yao Q et al (2019) Editing aberrant splice sites efficiently restores beta-globin expression in beta-thalassemia. Blood 133:2255–2262
Agudelo D, Duringer A, Bozoyan L et al (2017) Marker-free coselection for CRISPR-driven genome editing in human cells. Nat Methods 14:615–620
Shahbazi R, Sghia-Highes G, Reid JL et al (2019) Targeted homology-directed repair in blood stem and progenitor cells with CRISPR nanoformulations. Nat Mater 18:1124–1132
Thuronyi BW, Koblan LW, Levy JM et al (2019) Continuous evolution of base editors with expanded target compatibility and improved activity. Nat Biotechnol 37:1070–1079
Anzalone AV, Randolph PB, Davis JR et al (2019) Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576:149–157
Zeng J, Wu Y, Ren C et al (2020) Therapeutic base editing of human hematopoietic stem cells. Nat Med 26:535–541
Newby GA, Yen JS, Woodard KJ et al (2021) Base editing of haematopoietic stem cells rescues sickle cell disease in mice. Nature 595:295–302
Wu Y, Zeng J, Roscoe BP et al (2019) Highly efficient therapeutic gene editing of human hematopoietic stem cells. Nat Med 25:776–783
Klompe SE, Vo PLC, Halpin-Healy TS et al (2019) Transposon-encoded CRISPR-Cas systems direct RNA-guided DNA integration. Nature 571:219–225
Hendel A, Bak RO, Clark JT et al (2015) Chemically modified guide RNAs enhance CRISPR-Cas genome editing in human primary cells. Nat Biotechnol 33:985–989
Osborn MJ, Webber BR, Knipping F et al (2016) Evaluation of TCR gene editing achieved by TALENs, CRISPR/Cas9, and megaTAL nucleases. Mol Ther 24:570–581
Schumann K, Lin S, Boyer E et al (2015) Generation of knock-in primary human T cells using Cas9 ribonucleoproteins. Proc Natl Acad Sci U S A 112:10437–10442
Gundry MC, Brunetti L, Lin A et al (2016) Highly efficient genome editing of murine and human hematopoietic progenitor cells by CRISPR/Cas9. Cell Rep 17:1453–1461
DeWitt MA, Magis W, Bray NL et al (2016) Selection-free genome editing of the sickle mutation in human adult hematopoietic stem/progenitor cells. Sci Transl Med 8:360ra134
Lau CH, Tin C (2019) The synergy between CRISPR and chemical engineering. Curr Gene Ther 19:147–171
Ortinski PI, O’Donovan BO, Dong X et al (2017) Integrase-deficient lentiviral vector as an all-in-one platform for highly efficient CRISPR/Cas9-mediated gene editing. Mol Ther Methods Clin Dev 5:153–164
Maggio I, Zittersteijn HA, Wang Q et al (2020) Integrating gene delivery and gene-editing technologies by adenoviral vector transfer of optimized CRISPR-Cas9 components. Gene Ther 27:209–225
Ran FA, Hsu PD, Wright J et al (2013) Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8:2281–2308
Tycko J, Myer VE, Hsu PD (2016) Methods for optimizing CRISPR-Cas9 genome editing specificity. Mol Cell 6:355–370
Nishimasu H, Shi X, Ishiguro S et al (2018) Engineered CRISPR-Cas9 nuclease with expanded targeting space. Science 361:1259–1262
Kleinstiver BP, Sousa AA, Walton RT et al (2019) Engineered CRISPR-Cas12a variants with increased activities and improved targeting ranges for gene, epigenetic and base editing. Nat Biotechnol 37:276–282
Cortizas EM, Zahn A, Hajjar MR et al (2013) Alternative end-joining and classical nonhomologous end-joining pathways repair different types of double-strand breaks during class-switch recombination. J Immunol 191:5751–5763
Yu KR, Natanson H, Dunbar CE (2016) Gene editing of human hematopoietic stem and progenitor cells: promise and potential hurdles. Hum Gene Ther 27:729–740
Yeh CD, Richardson CD, Corn JE (2019) Advances in genome editing through control of DNA repair pathways. Nat Cell Biol 21:1468–1478
Roth TL, Puig-Saus C, Yu R et al (2018) Reprogramming human T cell function and specificity with non-viral genome targeting. Nature 559:405–409
Hornung V, Latz E (2010) Intracellular DNA recognition. Nat Rev Immunol 10:123–130
Yoshimi K, Kunihiro Y, Kaneko T et al (2016) ssODN-mediated knock-in with CRISPR-Cas for large genomic regions in zygotes. Nat Commun 7:10431–10431
Miura H, Quadros RM, Gurumurthy CB et al (2018) Easi-CRISPR for creating knock-in and conditional knockout mouse models using long ssDNA donors. Nat Protoc 13:195–215
Bak RO, Dever DP, Porteus MH (2018) CRISPR/Cas9 genome editing in human hematopoietic stem cells. Nat Protoc 13:358–376
Khan IF, Hirata RK, Russell DW (2011) AAV-mediated gene targeting methods for human cells. Nat Protoc 6:482–501
Grieger JC, Choi VW, Samulski RJ (2006) Production and characterization of adeno-associated viral vectors. Nat Protoc 1:1412–1428
Fu Y, Folden JA, Khayter C et al (2013) High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nat Biotechnol 31:822–826
Sentmanat MF, Peters ST, Florian CP et al (2018) A survey of validation strategies for CRISPR-Cas9 editing. Sci Rep 8:888
Brinkman EK, Chen T, Amendola M et al (2014) Easy quantitative assessment of genome editing by sequence trace decomposition. Nucleic Acids Res 42:e168
Martin F, Sanchez-Hernandez S, Gutierrez-Guerrero A et al (2016) Biased and unbiased methods for the detection of off-target cleavage by CRISPR/Cas9: an overview. Int J Mol Sci 17(9):1507
Charlesworth CT, Camarena J, Cromer MK et al (2018) Priming human repopulating hematopoietic stem and progenitor cells for Cas9/sgRNA gene targeting. Mol Ther Nucleic Acids 12:89–104
Humbert O, Radtke S, Samuelson C et al (2019) Therapeutically relevant engraftment of a CRISPR-Cas9-edited HSC-enriched population with HbF reactivation in nonhuman primates. Sci Transl Med 11:eaaw3768
Brunetti L, Gundry MC, Kitano A et al (2018) Highly efficient gene disruption of murine and human hematopoietic progenitor cells by CRISPR/Cas9. J Vis Exp 10:57278
Tran NT, Sommermann T, Graf R et al (2019) Efficient CRISPR/Cas9-mdiated gene knockin in mouse hematopoietic stem and progenitor cells. Cell Rep 28:3510–3522 e5
Kaltenbach JP, Kaltenbach MH, Lyons WB (1958) Nigrosin as a dye for differentiating live and dead ascites cells. Exp Cell Res 15:112–117
Kerenyi MA (2014) LT-HSC methylcellulose assay. Bio Protoc 4(5). https://doi.org/10.21769/BioProtoc.1067
Wognum B, Yuan N, Lai B et al (2013) Colony forming cell assays for human hematopoietic progenitor cells. Methods Mol Biol 946:267–283
Kuo CY, Long JD, Campo-Fernandez B et al (2018) Site-specific gene editing of human hematopoietic stem cells for X-linked Hyper-IgM syndrome. Cell Rep 23:2606–2616
Thomas KR, Folger KR, Capecchi MR (1986) High frequency targeting of genes to specific sites in the mammalian genome. Cell 44:419–428
Chen F, Pruett-Miller HY et al (2011) High-frequency genome editing using ssDNA oligonucleotides with zinc-finger nucleases. Nat Methods 8:753–755
Davis L, Maizels N (2014) Homology-directed repair of DNA nicks via pathways distinct from canonical double-strand break repair. Proc Natl Acad Sci U S A 111:E924–E932
Laustsen A, Bak RO (2019) Electroporation-based CRISPR/Cas9 gene editing using Cas9 protein and chemically modified sgRNAs. Methods Mol Biol 1961:127–134
Jinek M, Chylinski K, Fonfara I et al (2012) A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337:816–821
Mali P, Yang L, Esvelt KM et al (2013) RNA-guided human genome engineering via Cas9. Science 339:823–826
Cong L, Ran FA, Cox D et al (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339:819–823
Slaymaker IM, Gao L, Zetsche B et al (2016) Rationally engineered Cas9 nucleases with improved specificity. Science 351:84–88
Kleinstiver BP, Pattanayak V, Prew MS et al (2016) High-fidelity CRISPR-Cas9 nucleases with no detectable genome-wide off-target effects. Nature 529:490–495
Chen JS, Dagdas YS, Kleinstiver BP et al (2017) Enhanced proofreading governs CRISPR-Cas9 targeting accuracy. Nature 550:407–410
Fonfara I, Richter H, Bratovic M et al (2016) The CRISPR-associated DNA-cleaving enzyme Cpf1 also processes precursor CRISPR RNA. Nature 532:517–521
Kleinstiver BP, Tsai SQ, Prew MS et al (2016) Genome-wide specificities of CRISPR-Cas Cpf1 nucleases in human cells. Nat Biotechnol 34:869–874
Kim D, Kim J, Hur JK et al (2016) Genome-wide analysis reveals specificities of Cpf1 endonucleases in human cells. Nat Biotechnol 34:863–868
Safari F, Zare K, Negahdaripour M et al (2019) CRISPR Cpf1 proteins: structure, function and implications for genome editing. Cell Biosci 9:36
Abudayyeh OO, Gootenberg JS, Konermann S et al (2016) C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science 353:aaf5573
Cox DBT, Gootenberg JS, Abudayyeh OO et al (2017) RNA editing with CRISPR-Cas13. Science 358:1019–1027
Abudayyeh OO, Gootenber JS, Essletzbichler et al (2017) RNA targeting with CRISPR-Cas13. Nature 550:280–284
Konermann S, Lotfy P, Brideau NJ et al (2018) Transcriptome engineering with RNA-targeting type VI-D CRISPR effectors. Cell 173:665–676
Granados-Riveron JT, Aquino-Jarquin G (2018) CRISPR-Cas13 precision transcriptome engineering in cancer. Cancer Res 78:4107–4113
Makarova KS, Wolf YI, Alkhnbashi OS et al (2015) An updated evolutionary classification of CRISPR-Cas systems. Nat Rev Microbiol 13:722–736
Harrington LB, Burstein D, Chen JS et al (2018) Programmed DNA destruction by miniature CRISPR-Cas14 enzymes. Science 362:839–842
Moon SB, Kim DY, Ko J-H et al (2019) Recent advances in the CRISPR genome editing tool set. Exp Mol Med 51:130
Al-Shayeb B, Sachdeva R, Chen L-X et al (2020) Clades of huge phages from across Earth’s ecosystems. Nature 578:425–431
Pausch P, Al-Shayeb B, Bisom-Rapp E et al (2020) CRISPR-CasPhi from huge phages is a hypercompact genome editor. Science 369:333–337
Dolan AE, Hou Z, Xiao Y et al (2019) Introducing a spectrum of long-range genomic deletions in human embryonic stem cells using Type I CRISPR-Cas. Mol Cell 74:936–950 e5
Morisaka H, Yoshimi K, Okuzaki Y et al (2019) CRISPR-Cas3 induces broad and unidirectional genome editing in human cells. Nat Commun 10:5302
Ran FA, Hsu PD, Lin C-Y et al (2013) Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 154:1380–1389
Mal P, Aach J, Stranges PB et al (2013) CAS9 transcriptional activators for target specificity screening and paired nickases for cooperative genome engineering. Nat Biotechnol 31:833–838
Guilinger JP, Thompson DB, Liu DR (2014) Fusion of catalytically inactive Cas9 to FokI nuclease improves the specificity of genome modification. Nat Biotechnol 32:577–582
Rees HA, Liu DR (2018) Base editing: precision chemistry on the genome and transcriptome of living cells. Nat Rev Genet 19:770–788
Gaudelli NM, Komar AC, Rees HA (2017) Programmable base editing of A*T to G*C in genomic DNA without DNA cleavage. Nature 551:464–471
Strecker J, Ladha A, Gardner Z et al (2019) RNA-guided DNA insertion with CRISPR-associated transposases. Science 365:48–53
Hess GT, Fresard L, Han K et al (2016) Directed evolution using dCas9-targeted somatic hypermutation in mammalian cells. Nat Methods 13:1036–1042
Bae S, Park J, Kim JS (2014) Cas-OFFinder: a fast and versatile algorithm that searches for potential off-target sites of Cas9 RNA-guided endonucleases. Bioinformatics 30:1473–1475
Aach J, Mali P, Church GM (2014) CasFinder: flexible algorithm for identifying specific Cas9 targets in genomes (unpublished). bioRxiv. https://doi.org/10.1101/005074
Heigwer F, Kerr FG, Boutros M (2014) E-CRISP: fast CRISPR target site identification. Nat Methods 11:122–123
Oliveros JC, Franch M, Tabas-Madrid D et al (2016) Breaking-Cas-interactive design of guide RNAs for CRISPR-Cas experiments for ENSEMBL genomes. Nucleic Acids Res 44:W267–W271
Haeussler M, Schonig K, Eckert H et al (2016) Evaluation of off-target and on-target scoring algorithms and integration into the guide RNA selection tool CRISPOR. Genome Biol 17:148
Labun K, Montague TG, Krause M et al (2019) CHOPCHOP v3: expanding the CRISPR web toolbox beyond genome editing. Nucleic Acids Res 47:W171–W174
Sanson KR, Hanna RE, Hegde M et al (2018) Optimized libraries for CRISPR-Cas9 genetic screens with multiple modalities. Nat Commun 9:416
Stemmer M, Thumberger T, Keyer M et al (2017) Correction: CCTop: an intuitive, flexible and reliable CRISPR/Cas9 target prediction tool. PLoS One 12:e0176619
O’Brien A, Bailey TL (2014) GT-scan: identifying unique genomic targets. Bioinformatics 30:2673–2675
Naito Y, Hino K, Bono H et al (2015) CRISPRdirect: software for designing CRISPR/Cas guide RNA with reduced off-target sites. Bioinformatics 31:1120–1123
Xie S, Shen B, Zhang C et al (2014) sgRNAcas9: a software package for designing CRISPR sgRNA and evaluating potential off-target cleavage sites. PLoS One 9:e100448
Tsai SQ, Nguyen NT, Malagon-Lopez J et al (2017) CIRCLE-seq: a highly sensitive in vitro screen for genome-wide CRISPR-Cas9 nuclease off-targets. Nat Methods 1:607–614
Kim D, Bae S, Park J et al (2015) Digenome-seq: genome-wide profiling of CRISPR-Cas9 off-target effects in human cells. Nat Methods 12:37–43
Cameron P, Fuller CK, Donohoue PD et al (2017) Mapping the genomic landscape of CRISPR-Cas9 cleavage. Nat Methods 14:600–606
Yan WX, Mirzazadeh R, Garnerone S et al (2017) BLISS is a versatile and quantitative method for genome-wide profiling of DNA double-strand breaks. Nat Commun 8:15058
Tsai SQ, Zheng Z, Ngguyen NT et al (2015) GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nat Biotechnol 33:187–189
Hu J, Meyers RM, Dong J et al (2016) Detecting DNA double-stranded breaks in mammalian genomes by linear amplification-mediated high-throughput genome-wide translocation sequencing. Nat Protoc 11:53–871
Wienert B, Wyman SK, Richardson CD et al (2019) Unbiased detection of CRISPR off-targets in vivo using DISCOVER-Seq. Science 364:286–289
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Shahbazi, R., Lipson, P., Gottimukkala, K.S.V., Lane, D.D., Adair, J.E. (2023). CRISPR Gene Editing of Hematopoietic Stem and Progenitor Cells. In: Pelus, L.M., Hoggatt, J. (eds) Hematopoietic Stem Cells. Methods in Molecular Biology, vol 2567. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2679-5_4
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2679-5_4
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2678-8
Online ISBN: 978-1-0716-2679-5
eBook Packages: Springer Protocols