Abstract
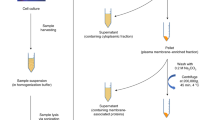
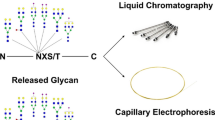
Glycans on viruses are major molecules that mediate the communication of the viruses with host cells. Investigating the structure of glycans may be important in the characterization of viruses. In this regard, the high-performance liquid chromatography (HPLC) is a widely used analytical technique. We developed a multidimensional HPLC mapping method that identifies glycan structures. This is achieved by separating and purifying glycans using three types of HPLC columns and comparing the elution time data with known data. This method enables the easy profiling of glycans at the molecular, cellular, and tissue levels and can be used to study the glycans of virological samples such as virus and their hosts. It also enables the discrimination of isomers, which is difficult via mass spectrometry. In this paper, we describe in detail the principle and method of the structural analysis of N-type glycans using the proposed method.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Li Y, Liu D, Wang Y et al (2021) The importance of Glycans of viral and host proteins in enveloped virus infection. Front Immunol 12. https://doi.org/10.3389/fimmu.2021.638573The
Raman R, Tharakaraman K, Sasisekharan V, et al (2016) Glycan–protein interactions in viral pathogenesis
Zaia J (2008) Mass spectrometry and the emerging field of Glycomics. Chem Biol 15:881–892
Takahashi N, Yagi H, Kato K et al (2008) The two-/three-dimensional HPLC mapping method for the identification of N-glycan structures. In: Comprehensive glycoscience
Yagi H, Kato K (2009) Multidimensional HPLC mapping method for the structural analysis of anionic N-glycans. Trends Glycosci Glycotechnol 21:95–104
Takahashi N, Kato K (2003) GALAXY(Glycoanalysis by the three axes of MS and chromatography): a web application that assists structural analyses of N-glycans. Trends Glycosci Glycotechnol 15:235–251
Yagi H, Watanabe S, Suzuki T et al (2012) Comparative analyses of N-glycosylation profiles of influenza a viruses grown in different host cells. Open Glycosci 5:2–12
Tomiya N, Takahashi N (1998) Contribution of component monosaccharides to the coordinates of neutral and sialyl pyridylaminated N-glycans on a two-dimensional sugar map. Anal Biochem 264:204–210
Yagi H, Takahashi N, Yamaguchi Y et al (2005) Development of structural analysis of sulfated N-glycans by multidimensional high-performance liquid chromatography mapping methods. Glycobiology 15:1051–1060
Yagi H, Yamada K, Ohno E et al (2008) Development and application of high-performance liquid chromatography map of Glucuronyl N-glycans. Open Glycosci 1:8–18
Yagi H, Takahashi N, Yamaguchi Y et al (2004) Temperature-dependent isologous Fab-Fab interaction that mediates cryocrystallization of a monoclonal immunoglobulin G. Mol Immunol 41:1211–1215
Guo C-T, Takahashi T, Bukawa W et al (2006) Edible bird’s nest extract inhibits influenza virus infection. Antivir Res 70:140–146
Holland M, Yagi H, Takahashi N et al (2006) Differential glycosylation of polyclonal IgG, IgG-Fc and IgG-Fab isolated from the sera of patients with ANCA-associated systemic vasculitis. Biochim Biophys Acta - Gen Subj 1760:669–677
Guo C-T, Takahashi N, Yagi H et al (2007) The quail and chicken intestine have sialyl-galactose sugar chains responsible for the binding of influenza A viruses to human type receptors. Glycobiology 17:713–724
Yagi H, Yasukawa N, Yu S-Y et al (2008) The expression of sialylated high-antennary N-glycans in edible bird’s nest. Carbohydr Res 343:1373–1377
Sriwilaijaroen N, Kondo S, Yagi H et al (2009) Analysis of N-glycans in embryonated chicken egg chorioallantoic and amniotic cells responsible for binding and adaptation of human and avian influenza viruses. Glycoconj J 26:433–443
Thongratsakul S, Songserm T, Poolkhet C et al (2009) Determination of N-linked Sialyl-sugar chains in the lungs of domestic cats and dogs in Thailand susceptible to the highly pathogenic avian influenza virus (H5N1). Open Glycosci 2:28–36
Sriwilaijaroen N, Kondo S, Yagi H et al (2011) N-Glycans from porcine trachea and lung: predominant NeuAcα2-6Gal could be a selective pressure for influenza variants in favor of human-type receptor. PLoS One 6:e16302
Yagi H, Ohno E, Kondo S et al (2011) Development and application of multidimensional HPLC mapping method for O-linked oligosaccharides. Biomol Ther 1:48–62
Acknowledgments
We would like to express our profound respect and gratitude to Dr. Noriko Takahashi, Professor Emeritus, Nagoya City University, for developing the multidimensional HPLC method and laying the foundation for its various applications and to Prof. Koichi Kato for his support of this study. This study was supported by a Grant-in-Aid for Scientific Research (21H02625, 18H03942, and 17H06414 to H.Y.) from the Ministry of Education, Culture, Sports, Science and Technology and the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Saito, T., Yagi, H. (2022). High-Performance Liquid Chromatography Mapping Method for the Structural Analysis of N-Glycans at the Molecular, Cellular, and Tissue Levels. In: Suzuki, Y. (eds) Glycovirology. Methods in Molecular Biology, vol 2556. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2635-1_2
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2635-1_2
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2634-4
Online ISBN: 978-1-0716-2635-1
eBook Packages: Springer Protocols