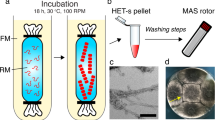
Abstract
Amyloid-beta (Aβ) aggregation into soluble oligomers and fibril formation are associated with Alzheimer’s disease (AD) pathogenesis. Aβ1–42 is the major form of the Aβ peptide present in neuritic plaques and shown to be neurotoxic both in vivo and in vitro. However, understanding the mechanism of its toxicity, aggregation, and other biochemical properties is limited because of its difficult production (recombinant or synthetic) and irreproducibility issues attributed to batch-to-batch preparation differences. Chemically synthetic Aβ1–42 is now well established, but it always introduces up to 5% D-isomers along with its L-isomeric form, and thus it is not fruitful for biochemical/structural studies. Here, we optimized an efficient published method for expression and purification of Aβ1–42 upon overexpression in Escherichia coli (E. coli) that provides a satisfactory yield as well as minimizes the variability between batch preparations. With the present protocol, ~7–8 mg/liter of unlabeled peptide and ~3.5–4 mg/liter for 13C,15N-labeled (double-labeled) Aβ1–42 were obtained.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Parihar MS, Hemnani T (2004) Alzheimer’s disease pathogenesis and therapeutic interventions. J Clinical Neurosci 11:456–467
Perl DP (2010) Neuropathology of Alzheimer’s disease. Mt Sinai J Med 77:32–42
Selkoe DJ (1994) Alzheimer’s disease: a central role for amyloid. J Neuropathol Exp Neurol 53:438–447
O’Brien RJ, Wong PC (2011) Amyloid precursor protein processing and Alzheimer’s disease. Ann Rev Neurosci 34:185–204
Selkoe DJ (1991) The molecular pathology of Alzheimer’s disease. Neuron 6:487–498
Finder VH, Vodopivec I, Nitsch RM et al (2010) The recombinant amyloid-beta peptide Abeta1-42 aggregates faster and is more neurotoxic than synthetic Abeta1-42. J Mol Biol 396:9–18
Abelein A, Chen G, Kitoka K et al (2020) High-yield production of Amyloid-beta peptide enabled by a customized spider silk domain. Sci Rep 10:235
LeVatte MA, Lipfert M, Ladner-Keay C et al (2019) Preparation and characterization of a highly soluble Abeta1-42 peptide variant. Protein Expr Purif 164:105480
Wälti MA, Ravotti F, Arai H et al (2016) Atomic-resolution structure of a disease-relevant Abeta(1-42) amyloid fibril. Proc Natl Acad Sci USA 113:E4976–E4984
Ravotti F, Wälti MA, Guntert P et al (2016) Solid-state NMR sequential assignment of an Amyloid-beta(1-42) fibril polymorph. Biomol NMR Assign 10:269–276
Wälti MA, Orts J, Riek R (2017) Quenched hydrogen-deuterium exchange NMR of a disease-relevant Abeta(1-42) amyloid polymorph. PLoS One 12:e0172862
Tropea JE, Cherry S, Waugh DS (2009) Expression and purification of soluble His(6)-tagged TEV protease. Methods Mol Biol 498:297–307
Kopanic JL, Al-Mugotir M, Zach S et al (2013) An Escherichia coli strain for expression of the connexin45 carboxyl terminus attached to the 4th transmembrane domain. Front Pharmacol 4:106
Bitan G, Fradinger EA, Spring SM et al (2005) Neurotoxic protein oligomers–what you see is not always what you get. Amyloid 2:88–95
Bitan G, Kirkitadze MD, Lomakin A et al (2003) Amyloid beta -protein (Abeta) assembly: Abeta 40 and Abeta 42 oligomerize through distinct pathways. Proc Natl Acad Sci USA 100:330–335
Pujol-Pina R, Vilaprinyo-Pascual S, Mazzucato R et al (2015) SDS-PAGE analysis of Abeta oligomers is disserving research into Alzheimer s disease: appealing for ESI-IM-MS. Sci Rep 5:14809
Acknowledgment
The work was supported by the Sinergia grant (177195), Swiss National Science Foundation. The authors would like to acknowledge Dr. Witold Kwiatkowski, the Bio NMR group, and the Laboratory of Physical chemistry, ETH Zürich, for providing the infrastructure to carry out the project.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Ghosh, D., Wälti, M.A., Riek, R. (2023). An Efficient Method of Expression and Purification of Amyloid-Beta (Aβ1–42) Peptide from E. coli. In: Cieplak, A.S. (eds) Protein Aggregation. Methods in Molecular Biology, vol 2551. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2597-2_4
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2597-2_4
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2596-5
Online ISBN: 978-1-0716-2597-2
eBook Packages: Springer Protocols