Abstract
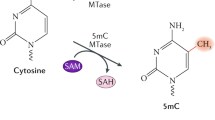
DNA methylations are one of the most well-known epigenetic modifications along with histone modifications and noncoding RNAs. They are found at specific sites along the DNA in all domains of life, with 5-mC and 6-mA/4-mC being well-characterized in eukaryotes and bacteria respectively, and they have not only been described as contributing to the structure of the double helix itself but also as regulators of DNA-based processes such as replication, transcription, and recombination. Different methods have been developed to accurately identify and/or map methylated motifs to decipher the involvement of DNA methylations in regulatory networks that affect the cellular state.
Although DNA methylations have been detected along archaeal genomes, their involvement as regulators of DNA-based processes remains the least known. To highlight the importance of DNA methylations in the control of key cellular mechanisms and their dynamics in archaea cells, we have used single-molecule real-time (SMRT) sequencing. This sequencing technology allows the identification and direct mapping of the methylated motifs along the genome of an organism. In this chapter, we present a step-by-step protocol for detecting DNA methylations in the hyperthermophilic crenarchaeon Sulfolobus acidocaldarius using SMRT sequencing. This protocol can easily be adapted to other prokaryotes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Jeltsch A (2002) Beyond Watson and Crick: DNA methylation and molecular enzymology of DNA methyltransferases. Chembiochem 3:274–293. https://doi.org/10.1002/1439-7633(20020402)3:4<274::AID-CBIC274>3.0.CO;2-S
Sánchez-Romero MA, Casadesús J (2020) The bacterial epigenome. Nat Rev Microbiol 18:7–20. https://doi.org/10.1038/s41579-019-0286-2
Severin PMD, Zou X, Gaub HE, Schulten K (2011) Cytosine methylation alters DNA mechanical properties. Nucleic Acids Res 39:8740–8751. https://doi.org/10.1093/nar/gkr578
Geahigan KB, Meints GA, Hatcher ME, Orban J, Drobny GP (2000) The dynamic impact of CpG methylation in DNA. Biochemistry 39:4939–4946. https://doi.org/10.1021/bi9917636
Derreumaux S, Chaoui M, Tevanian G, Fermandjian S (2001) Impact of CpG methylation on structure, dynamics and solvation of cAMP DNA responsive element. Nucleic Acids Res 29:2314–2326. https://doi.org/10.1093/nar/29.11.2314
Diekmann S (1987) DNA methylation can enhance or induce DNA curvature. EMBO J 6:4213–4217. https://doi.org/10.1002/j.1460-2075.1987.tb02769.x
Polaczek P, Kwan K, Campbell JL (1998) GATC motifs may alter the conformation of DNA depending on sequence context and N 6 -adenine methylation status: possible implications for DNA-protein recognition. Mol Gen Genet MGG 258:488–493. https://doi.org/10.1007/s004380050759
Wion D, Casadesús J (2006) N6-methyl-adenine: an epigenetic signal for DNA–protein interactions. Nat Rev Microbiol 4:183–192. https://doi.org/10.1038/nrmicro1350
Camacho EM, Casadesús J (2005) Regulation of traJ transcription in the Salmonella virulence plasmid by strand-specific DNA adenine hemimethylation: regulation of traJ transcription by Dam methylation. Mol Microbiol 57:1700–1718. https://doi.org/10.1111/j.1365-2958.2005.04788.x
Kumar S, Karmakar BC, Nagarajan D, Mukhopadhyay AK, Morgan RD, Rao DN (2018) N4-cytosine DNA methylation regulates transcription and pathogenesis in Helicobacter pylori. Nucleic Acids Res 46:3429–3445. https://doi.org/10.1093/nar/gky126
Clemens AW, Wu DY, Moore JR, Christian DL, Zhao G, Gabel HW (2019) MeCP2 represses enhancers through chromosome topology-associated DNA methylation. Mol Cell 77:1–15. https://doi.org/10.1016/j.molcel.2019.10.033
Nye TM, van Gijtenbeek LA, Stevens AG, Schroeder JW, Randall JR, Matthews LA, Simmons LA (2020) Methyltransferase DnmA is responsible for genome-wide N6-methyladenosine modifications at non-palindromic recognition sites in Bacillus subtilis. Nucleic Acids Res 48:5332–5348. https://doi.org/10.1093/nar/gkaa266
Fu Y, Luo G-Z, Chen K, Deng X, Yu M, Han D, Hao Z, Liu J, Lu X, Doré LC, Weng X, Ji Q, Mets L, He C (2015) N6-Methyldeoxyadenosine marks active transcription start sites in Chlamydomonas. Cell 161:879–892. https://doi.org/10.1016/j.cell.2015.04.010
Beh LY, Debelouchina GT, Clay DM, Thompson RE, Lindblad KA, Hutton ER, Bracht JR, Sebra RP, Muir TW, Landweber LF (2019) Identification of a DNA N6-adenine methyltransferase complex and its impact on chromatin organization. Cell 177:1781–1796. https://doi.org/10.1016/j.cell.2019.04.028
Lizarraga A, O’Brown ZK, Boulias K, Roach L, Greer EL, Johnson PJ, Strobl-Mazzulla PH, de Miguel N (2020) Adenine DNA methylation, 3D genome organization, and gene expression in the parasite Trichomonas vaginalis. Proc Natl Acad Sci U S A 117:13033–13043. https://doi.org/10.1073/pnas.1917286117
Roberts RJ, Vincze T, Posfai J, Macelis D (2010) REBASE – a database for DNA restriction and modification: enzymes, genes and genomes. Nucleic Acids Res 38:D234–D236. https://doi.org/10.1093/nar/gkp874
Roberts RJ, Vincze T, Posfai J, Macelis D (2015) REBASE – a database for DNA restriction and modification: enzymes, genes and genomes. Nucleic Acids Res 43:D298–D299. https://doi.org/10.1093/nar/gku1046
Greer EL, Blanco MA, Gu L, Sendinc E, Liu J, Aristizábal-Corrales D, Hsu C-H, Aravind L, He C, Shi Y (2015) DNA methylation on N6-Adenine in C. elegans. Cell 161:868–878. https://doi.org/10.1016/j.cell.2015.04.005
Zhang G, Huang H, Liu D, Cheng Y, Liu X, Zhang W, Yin R, Zhang D, Zhang P, Liu J, Li C, Liu B, Luo Y, Zhu Y, Zhang N, He S, He C, Wang H, Chen D (2015) N6-Methyladenine DNA modification in drosophila. Cell 161:893–906. https://doi.org/10.1016/j.cell.2015.04.018
Weber M, Davies JJ, Wittig D, Oakeley EJ, Haase M, Lam WL, Schübeler D (2005) Chromosome-wide and promoter-specific analyses identify sites of differential DNA methylation in normal and transformed human cells. Nat Genet 37:853–862. https://doi.org/10.1038/ng1598
Down TA, Rakyan VK, Turner DJ, Flicek P, Li H, Kulesha E, Gräf S, Johnson N, Herrero J, Tomazou EM, Thorne NP, Bäckdahl L, Herberth M, Howe KL, Jackson DK, Miretti MM, Marioni JC, Birney E, Hubbard TJP, Durbin R, Tavaré S, Beck S (2008) A Bayesian deconvolution strategy for immunoprecipitation-based DNA methylome analysis. Nat Biotechnol 26:779–785. https://doi.org/10.1038/nbt1414
Flusberg BA, Webster DR, Lee JH, Travers KJ, Olivares EC, Clark TA, Korlach J, Turner SW (2010) Direct detection of DNA methylation during single-molecule, real-time sequencing. Nat Methods 7:461–465. https://doi.org/10.1038/nmeth.1459
Clark TA, Murray IA, Morgan RD, Kislyuk AO, Spittle KE, Boitano M, Fomenkov A, Roberts RJ, Korlach J (2012) Characterization of DNA methyltransferase specificities using single-molecule, real-time DNA sequencing. Nucleic Acids Res 40:e29–e29. https://doi.org/10.1093/nar/gkr1146
Laszlo AH, Derrington IM, Brinkerhoff H, Langford KW, Nova IC, Samson JM, Bartlett JJ, Pavlenok M, Gundlach JH (2013) Detection and mapping of 5-methylcytosine and 5-hydroxymethylcytosine with nanopore Msp A. Proc Natl Acad Sci 110:18904–18909. https://doi.org/10.1073/pnas.1310240110
Rand AC, Jain M, Eizenga JM, Musselman-Brown A, Olsen HE, Akeson M, Paten B (2017) Mapping DNA methylation with high-throughput nanopore sequencing. Nat Methods 14:411–413. https://doi.org/10.1038/nmeth.4189
Prangishvili DA, Vashakidze RP, Chelidze MG, Gabriadze IY (1985) A restriction endonuclease Sua I from the thermoacidophilic archaebacterium Sulfolobus acidocaldarius. FEBS Lett 192:57–60. https://doi.org/10.1016/0014-5793(85)80042-9
Grogan DW (2003) Cytosine methylation by the suaI restriction-modification system: implications for genetic fidelity in a hyperthermophilic archaeon. J Bacteriol 185:4657–4661. https://doi.org/10.1128/JB.185.15.4657-4661.2003
Ouellette M, Jackson L, Chimileski S, Papke RT (2015) Genome-wide DNA methylation analysis of Haloferax volcanii H26 and identification of DNA methyltransferase related PD-(D/E)XK nuclease family protein HVO_A0006. Front Microbiol 6. https://doi.org/10.3389/fmicb.2015.00251
Blow MJ, Clark TA, Daum CG, Deutschbauer AM, Fomenkov A, Fries R, Froula J, Kang DD, Malmstrom RR, Morgan RD, Posfai J, Singh K, Visel A, Wetmore K, Zhao Z, Rubin EM, Korlach J, Pennacchio LA, Roberts RJ (2016) The epigenomic landscape of prokaryotes. PLoS Genet 12:e1005854. https://doi.org/10.1371/journal.pgen.1005854
Couturier M, Lindås A-C (2018) The DNA methylome of the hyperthermoacidophilic crenarchaeon Sulfolobus acidocaldarius. Front Microbiol 9. https://doi.org/10.3389/fmicb.2018.00137
Chimileski S, Dolas K, Naor A, Gophna U, Papke RT (2014) Extracellular DNA metabolism in Haloferax volcanii. Front Microbiol 5. https://doi.org/10.3389/fmicb.2014.00057
Acknowledgments
The authors would like to thank the staff at the Uppsala Genome Center, National Genomics Infrastructure, Science for Life Laboratory for helpful comments on the DNA quality check protocol.
Funding
Funding was provided by the Carl R. Woese Institute for Genomic Biology postdoctoral fellowship (to M.C.).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Tellgren-Roth, C., Couturier, M. (2022). Detecting DNA Methylations in the Hyperthermoacidophilic Crenarchaeon Sulfolobus acidocaldarius Using SMRT Sequencing. In: Peeters, E., Bervoets, I. (eds) Prokaryotic Gene Regulation. Methods in Molecular Biology, vol 2516. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2413-5_3
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2413-5_3
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2412-8
Online ISBN: 978-1-0716-2413-5
eBook Packages: Springer Protocols