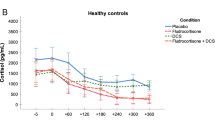
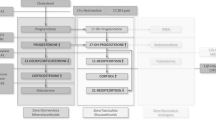
Abstract
Glucocorticoids mediate their actions, including feedback regulation of the hypothalamic-pituitary-adrenal (HPA) axis, through two distinct corticosteroid receptor subtypes: mineralocorticoid receptors (MR) and glucocorticoid receptors (GR). Dexamethasone, specifically the dexamethasone suppression test (DST), was the first and most studied glucocorticoid for assessing HPA axis activity; unfortunately, it has pharmacodynamic and pharmacokinetic features that are very distinct from cortisol. We have developed a suppressive test using prednisolone, which is pharmacologically more similar to cortisol than dexamethasone and can therefore obtain a more physiological assessment of HPA axis regulation. We focus here the prednisolone suppression test (PST), summarizing the preclinical work, studies in healthy control subjects, and applications to date in clinical populations. We propose that prednisolone at the 5-mg dosage, together with the assessment of salivary cortisol, can be used to investigate both impaired and enhanced glucocorticoid-mediated negative feedback. The work undertaken to date suggests that, in depression, results using the PST differ from those of the DST, in that patients who are non-suppressors to dexamethasone show normal suppressive responses to prednisolone. Furthermore, preserved suppressive responses to prednisolone predict treatment response in previously difficult-to-treat depression, whereas prednisolone non-suppression is predictive of severe treatment resistance even to the most intensive treatments. We argue that the different results using the PST, which probes both MR and GR, and the DST, which probes only GR, suggest a dissociation between GR and MR function in depressive patients. Thus, we propose that the prednisolone suppression test may offer specific biological and clinical information related to its action at both the GR and the MR. Therefore, the relevance of HPA axis dysfunction in psychiatric disorders and the ability to distinguish “true” from “pseudo-” treatment-resistant depression suggest that the PST holds an important clinical tool.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Nemeroff CB, Evans DL (1984) Correlation between the dexamethasone suppression test in depressed patients and clinical response. Am J Psychiatry 141(2):247–249
Gold PW, Goodwin FK, Chrousos GP (1988) Clinical and biochemical manifestations of depression. Relation to the neurobiology of stress. N Engl J Med 319(7):413–420
de Kloet ER, Vreugdenhil E, Oitzl MS, Joels M (1998) Brain corticosteroid receptor balance in health and disease. Endocr Rev 19(3):269–301
McEwen BS (2000) Allostasis and allostatic load: implications for neuropsychopharmacology. Neuropsychopharmacology 22(2):108–124
Reul JM, van den Bosch FR, de Kloet ER (1987) Relative occupation of type-I and type-II corticosteroid receptors in rat brain following stress and dexamethasone treatment: functional implications. J Endocrinol 115(3):459–467
Arriza JL, Simerly RB, Swanson LW, Evans RM (1988) The neuronal mineralocorticoid receptor as a mediator of glucocorticoid response. Neuron 1(9):887–900
Fuxe K, Harfstrand A, Agnati LF et al (1985) Immunocytochemical studies on the localization of glucocorticoid receptor immunoreactive nerve cells in the lower brain stem and spinal cord of the male rat using a monoclonal antibody against rat liver glucocorticoid receptor. Neurosci Lett 60(1):1–6
Modell S, Yassouridis A, Huber J, Holsboer F (1997) Corticosteroid receptor function is decreased in depressed patients. Neuroendocrinology 65(3):216–222
Juruena MF, Cleare AJ, Bauer ME, Pariante CM (2003) Molecular mechanisms of glucocorticoid receptor sensitivity and relevance to affective disorders. Acta Neuropsychiatrica 15(6):354–367
Carroll BJ, Martin FI, Davies B (1968) Pituitary–adrenal function in depression. Lancet 1(7556):1373–1374
Arana GW, Baldessarini RJ, Ornsteen M (1985) The dexamethasone suppression test for diagnosis and prognosis in psychiatry: commentary and review. Arch Gen Psychiatry 42(12):1193–1204
Ribeiro SC, Tandon R, Grunhaus L, Greden JF (1993) The DST as a predictor of outcome in depression: a meta-analysis. Am J Psychiatry 150(11):1618–1629
Carroll BJ, Feinberg M, Greden JF et al (1981) A specific laboratory test for the diagnosis of melancholia. Standardization, validation, and clinical utility. Arch Gen Psychiatry 38(1):15–22
Carroll BJ (1982) The dexamethasone suppression test for melancholia. Br J Psychiatry 140:292–304
Rush AJ, Weissenburger JE (1994) Melancholic symptom features and DSM-IV. Am J Psychiatry 151(4):489–498
Rush AJ, Giles DE, Schlesser MA et al (1996) The dexamethasone suppression test in patients with mood disorders. J Clin Psychiatry 57(10):470–484
Juruena MF, Agustini B, Cleare AJ, Young AH. A translational approach to clinical practice via stress-responsive glucocorticoid receptor signalling. Stem cell investigation. 2017;(16) 4:13
Nelson JC, Davis JM (1997) DST studies in psychotic depression: a metaanalysis. Am J Psychiatry 154(11):1497–1503
Pariante CM, Papadopoulos AS, Poon L et al (2002) A novel prednisolone suppression test for the hypothalamic-pituitary-adrenal axis. Biol Psychiatry 51(11):922–930
Orth D, Kovacs W (1998) The adrenal cortex. In: Wilson J, Foster D, Kronenberg H, Larsen P (eds) Williams textbook of endocrinology, 9th edn. WB Saunders Company, Philadelphia, pp 517–664
Rupprecht R, Reul JM, van Steensel B et al (1993) Pharmacological and functional characterization of human mineralocorticoid and glucocorticoid receptor ligands. Eur J Pharmacol 247(2):145–154
Reul JM, Gesing A, Droste S et al (2000) The brain mineralocorticoid receptor: greedy for ligand, mysterious in function. Eur J Pharmacol 405(1–3):235–249
von Bardeleben U, Holsboer F (1991) Effect of age on the cortisol response to human corticotropin-releasing hormone in depressed patients pretreated with dexamethasone. Biol Psychiatry 29(10):1042–1050
von Bardeleben U, Holsboer F, Stalla GK, Muller OA (1985) Combined administration of human corticotropin-releasing factor and lysine vasopressin induces cortisol escape from dexamethasone suppression in healthy subjects. Life Sci 37(17):1613–1618
Heuser I, Yassouridis A, Holsboer F (1994) The combined dexamethasone/CRH test: a refined laboratory test for psychiatric disorders. J Psychiatr Res 28(4):341–356
Watson S, Gallagher P, Smith MS, Ferrier IN, Young AH (2006) The dex/CRH test—is it better than the DST? Psychoneuroendocrinology 31(7):889–894
Pariante CM, Papadopoulos AS, Poon L et al (2004) Four days of citalopram increase suppression of cortisol secretion by prednisolone in healthy volunteers. Psychopharmacology 177(1–2):200–206
Lan NC, Matulich DT, Morris JA, Baxter JD (1981) Mineralocorticoid receptor like aldosterone-binding protein in cell culture. Endocrinology 109(6):1963–1970
Liddle GW (1958) Aldosterone antagonists. AMA Arch Intern Med 102(6):998–1004
Slater JD, Moxham A, Hurter R, Nabarro J (1959) Clinical and metabolic effects of aldosterone antagonism. Lancet 2:931–934
Ballard PL, Carter JP, Graham BS, Baxter JD (1975) A radioreceptor assay for evaluation of the plasma glucocorticoid activity of natural and synthetic steroids in man. J Clin Endocrinol Metab 41(2):290–304
Lan NC, Graham B, Bartter FC, Baxter JD (1982) Binding of steroids to mineralocorticoid receptors: implications for in vivo occupancy by glucocorticoids. J Clin Endocrinol Metab 54(2):332–342
Grossmann C, Scholz T, Rochel M et al (2004) Transactivation via the human glucocorticoid and mineralocorticoid receptor by therapeutically used steroids in CV-1 cells: a comparison of their glucocorticoid and mineralocorticoid properties. Eur J Endocrinol 151(3):397–406
Kirschbaum C, Hellhammer DH (1994) Salivary cortisol in psychoneuroendocrine research: recent developments and applications. Psychoneuroendocrinology 19(4):313–333
Reynolds RM, Bendall HE, Whorwood CB, Wood PJ, Walker BR, Phillips DI (1998) Reproducibility of the low dose dexamethasone suppression test: comparison between direct plasma and salivary cortisol assays. Clin Endocrinol 49(3):307–310
Juruena MF, Cleare AJ, Papadopoulos AS, Poon L, Lightman S, Pariante CM (2010 Nov) The prednisolone suppression test in depression: dose-response and changes with antidepressant treatment. Psychoneuroendocrinology 35(10):1486–1491
Juruena MF, Pariante CM, Papadopoulos AS, Poon L, Lightman S, Cleare AJ (2009) Prednisolone suppression test in depression: prospective study of the role of HPA axis dysfunction in treatment resistance. Br J Psychiatry 194(4):342–349
Jerjes WK, Cleare AJ, Wood PJ, Taylor NF (2006) Assessment of subtle changes in glucocorticoid negative feedback using prednisolone: comparison of salivary free cortisol and urinary cortisol metabolites as endpoints. Clin Chim Acta 364(1–2):279–286
Seidegard J, Simonsson M, Edsbacker S (2000) Effect of an oral contraceptive on the plasma levels of budesonide and prednisolone and the influence on plasma cortisol. Clin Pharmacol Ther 67(4):373–381
Juruena MF, Cleare AJ, Papadopoulos AS, Poon L, Lightman S, Pariante CM (2006) Different responses to dexamethasone and prednisolone in the same depressed patients. Psychopharmacology 189(2):225–235
Baldessarini RJ, Arana GW (1985) Does the dexamethasone suppression test have clinical utility in psychiatry? J Clin Psychiatry 46(2 Pt 2):25–29
Young EA, Lopez JF, Murphy-Weinberg V, Watson SJ, Akil H (2003) Mineralocorticoid receptor function in major depression. Arch Gen Psychiatry 60(1):24–28
Gold PW, Chrousos GP (2002) Organization of the stress system and its dysregulation in melancholic and atypical depression: high vs low CRH/ NE states. Mol Psychiatry 7(3):254–275
Holsboer F (2000) The corticosteroid receptor hypothesis of depression. Neuropsychopharmacology 23(5):477–501
Juruena M. The neurobiology of treatment resistant depression: role of the hypothalamic-pituitary-adrenal axis and glucocorticoid and mineralocorticoid receptor function PhD thesis, University of London, Institute of Psychiatry/King’s College London; 2007
Coryell W, Tsuang D (1992) Hypothalamic–pituitary–adrenal axis hyperactivity and psychosis: recovery during an 8-year follow-up. Am J Psychiatry 149(8):1033–1039
Van VC, Winokur G, Behar D, Lowry M (1984) Depressed women with panic attacks. J Clin Psychiatry 45(9):367–369
Brown C, Schulberg HC, Shear MK (1996) Phenomenology and severity of major depression and comorbid lifetime anxiety disorders in primary medical care practice. Anxiety 2(5):210–218
Joffe RT, Bagby RM, Levitt A (1993) Anxious and nonanxious depression. Am J Psychiatry 150(8):1257–1258
Kara S, Yazici KM, Gulec C, Unsal I (2000) Mixed anxiety–depressive disorder and major depressive disorder: comparison of the severity of illness and biological variables. Psychiatry Res 94(1):59–66
Young EA, Abelson JL, Cameron OG (2004) Effect of comorbid anxiety disorders on the hypothalamic–pituitary–adrenal axis response to a social stressor in major depression. Biol Psychiatry 56(2):113–120
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Juruena, M.F., Cleare, A.J., Young, A.H. (2022). The Role of Hypothalamic-Pituitary-Adrenal Axis in the Pathophysiology of Major Depression: A Translational Research Perspective of the Prednisolone Suppression Test. In: Kim, YK., Amidfar, M. (eds) Translational Research Methods for Major Depressive Disorder. Neuromethods, vol 179. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2083-0_1
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2083-0_1
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2082-3
Online ISBN: 978-1-0716-2083-0
eBook Packages: Springer Protocols