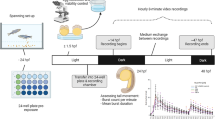
Abstract
Zebrafish (Danio rerio) has rapidly become a popular model species for behavioral studies that may be relevant to drug screening and safety toxicology. Zebrafish embryos show a complex behavioral repertoire already a few hours after fertilization. Particularly, early stage zebrafish show characteristic behavioral features such as spontaneous tail coiling (STC) or induced movements when exposed to a short and bright light flash (called photomotor response—PMR). In this chapter, we provide the methods for assessing STC and PMR in zebrafish embryos and to detect changes provoked by chemicals. One of the protocols uses video analysis suitable for automated high-throughput screening. Moreover, both protocols describe the use of automated video analysis by using an open-source integration platform (KNIME® analytics platform), providing a flexible workflow system that can be adapted to a diversity of video recordings. We also provide a toxicological validation of this assay and show that these protocols can be used to provide an automated, high data-content readout for zebrafish behavioral responses.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bailey JM, Oliveri AN, Levin ED (2015) Pharmacological analyses of learning and memory in zebrafish (Danio rerio). Pharmacol Biochem Behav 139(Pt B):103–111
Crofton KM, Mundy WR, Lein PJ et al (2010) Developmental neurotoxicity testing: recommendations for developing alternative methods for the screening and prioritization of chemicals. ALTEX 28:9–15
Coecke S, Goldberg AM, Allen S et al (2007) Workgroup report: incorporating in vitro alternative methods for developmental neurotoxicity into international hazard and risk assessment strategies. Environ Health Perspect 115:924–931
Tropepe V, Sive HL (2003) Can zebrafish be used as a model to study the neurodevelopmental causes of autism? Genes Brain Behav 2:268–281
Brustein E, Saint-Amant L, Buss RR et al (2003) Steps during the development of the zebrafish locomotor network. J Physiol 97:77–86
Saint-Amant L, Drapeau P (1998) Time course of the development of motor behaviors in the zebrafish embryo. J Neurobiol 37:622–632
Zindler F, Beedgen F, Braunbeck T (2019) Time-course of coiling activity in zebrafish (Danio rerio) embryos exposed to ethanol as an endpoint for developmental neurotoxicity (DNT) – hidden potential and underestimated challenges. Chemposphere 235:12–20
Ogungbemi AO, Teixido E, Massei R et al (2020) Optimization of the spontaneous tail coiling test for fast assessment of neurotoxic effects in the zebrafish embryo using an automated workflow in KNIME®. Neurotoxicol Teratol 81:106918
Drapeau P, Saint-Amant L, Buss RR et al (2002) Development of the locomotor network in zebrafish. Prog Neurobiol 68:85–111
Kokel D, Bryan J, Laggner C et al (2010) Rapid behavior-based identification of neuroactive small molecules in the zebrafish. Nat Chem Biol 6:231–227
Kokel D, Peterson RT (2011) Using the Zebrafish Photomotor response for psychotropic drug screening. Methods Cell Biol 105:517–524
Selderslaghs IWT, Hooyberghs J, Blust R et al (2013) Assessment of the developmental neurotoxicity of compounds by measuring locomotor activity in zebrafish embryos and larvae. Neurotoxicol Teratol 37:44–56
Vliet SM, Ho TC, Volz DC (2017) Behavioral screening of the LOPAC1280 library in zebrafish embryos. Toxicol Appl Pharmacol 329:241–248
Legradi J, el Abdellaoui N, van Pomeren M et al (2015) Comparability of behavioural assays using zebrafish larvae to assess neurotoxicity. Environ Sci Pollut Res Int 22:16277–16289
Ogungbemi A, Leuthold D, Scholz S et al (2019) Hypo- or hyperactivity of zebrafish embryos provoked by neuroactive substances: a review on how experimental parameters impact the predictability of behavior changes. Environ Sci Eur 31:88
Berthold MR, Cebron N, Dill F et al (2008) KNIME: the Konstanz information miner. In: Preisach C, Burkhardt H, Schmidt-Thieme L et al (eds) Data analysis, machine learning and applications. Springer, Berlin, Heidelberg, pp 319–326
Lammer E, Carr GJ, Wendler K et al (2009) Is the fish embryo toxicity test (FET) with the zebrafish (Danio rerio) a potential alternative for the fish acute toxicity test? Comp Biochem Physiol C Toxicol Pharmacol 149:196–209
Westerfield M (1995) The zebrafish book: a guide for the laboratory use of zebrafish (Brachydanio rerio). Oregon press, University of
ISO International Standards (1996) Water quality – Determination of the acute lethal toxicity of substance to a freshwater fish [Brachydanio rerio Hamilton-Buchanan] ISO 7346-3: Flow-through method. http://www.iso.org
OECD (2013) Test no. 236: fish embryo acute toxicity (FET) test. OECD guidelines for the testing of chemicals, subheading 2. Paris, France. 1–22
Fischer FC, Cirpka OA, Goss K-U et al (2018) Application of experimental polystyrene partition constants and diffusion coefficients to predict the sorption of neutral organic chemicals to multiwell plates in in vivo and in vitro bioassays. Environ Sci Technol 52:13511–13522
Riedl J, Altenburger R (2007) Physicochemical substance properties as indicators for unreliable exposure in microplate-based bioassays. Chemosphere 67:2210–2220
Teixidó E, Klüver N, Ogungbemi A, et al (2020) KNIME workflows for the evaluation of neurotoxic effects in zebrafish embryos by automatic measurement of early motor behaviours [Data set]. https://doi.org/10.5281/zenodo.3835947
Copmans D, Meinl T, Dietz C et al (2016) A KNIME-based analysis of the Zebrafish photomotor response clusters the phenotypes of 14 classes of neuroactive molecules. J Biomol Screen 21:427–436
Developers FFmpeg (2016) FFmpeg tool. http://ffmpeg.org
Ryan JA, Ulrich JN (2018) Quantmod: Quantitative Financial Modelling Framework. R package version 0.4-13.
Wahlby C, Sintorn I-M, Erlandsson F et al (2004) Combining intensity, edge and shape information for 2D and 3D segmentation of cell nuclei in tissue sections. J Microsc 215:67–76
Ritz C, Baty F, Streibig JC et al (2015) Dose-response analysis using R. PLoS One 10:e0146021
Croen LA, Connors SL, Matevia M et al (2011) Prenatal exposure to β2-adrenergic receptor agonists and risk of autism spectrum disorders. J Neurodev Disord 3:307–315
Kudo C, Kori M, Matsuzaki K et al (2003) Diclofenac inhibits proliferation and differentiation of neural stem cells. Biochem Pharmacol 66:289–295
Andreasson KI, Savonenko A, Vidensky S et al (2001) Age-dependent cognitive deficits and neuronal apoptosis in cyclooxygenase-2 transgenic mice. J Neurosci 21:8198–8209
Biol TJ, Elibol B, Aritan Oğur B et al (2019) Prenatal exposure of diclofenac sodium alters the behavioral development of young Wistar rats. Turk J Biol 43:305–313
Teixidó E, Kießling TR, Krupp E et al (2019) Automated morphological feature assessment for Zebrafish embryo developmental toxicity screens. Toxicol Sci 167:438–449
Bittner L, Teixidó E, Keddi I et al (2019) pH-dependent uptake and sublethal effects of antihistamines in zebrafish (Danio rerio) embryos. Aquat Toxicol 38:1012–1022
Maes J, Verlooy L, Buenafe OE et al (2012) Evaluation of 14 organic solvents and carriers for screening applications in Zebrafish embryos and larvae. PLoS One 7:e43850
Padilla S, Hunter DL, Padnos B et al (2011) Assessing locomotor activity in larval zebrafish: influence of extrinsic and intrinsic variables. Neurotoxicol Teratol 33:624–630
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
1 Electronic Supplementary Material
Figure S1
Mean frequency of spontaneous tail coiling (STC) of untreated zebrafish embryos over time. Error bars indicate the standard deviation of n=3 biological replicates of 13-16 embryos each. Solid line describes the polynomial fit of the data (DOCX 49 kb)
Table S1
Configuration of the communication with ffmpeg and external tool node (DOCX 70 kb)
Table S2
Example of plate layout (saved as excel file) for 96 well plate analysis. The layout can be generated using the supplementary file supplied with the KNIME workflow (excel file named animal list). A layout should be generated for each video recording for the PMR and STC analysis in 96 well-plates. (DOCX 17 kb)
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Teixidó, E., Klüver, N., Ogungbemi, A.O., Küster, E., Scholz, S. (2021). Evaluation of Neurotoxic Effects in Zebrafish Embryos by Automatic Measurement of Early Motor Behaviors. In: Llorens, J., Barenys, M. (eds) Experimental Neurotoxicology Methods. Neuromethods, vol 172. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1637-6_17
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1637-6_17
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1636-9
Online ISBN: 978-1-0716-1637-6
eBook Packages: Springer Protocols