Abstract
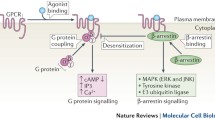
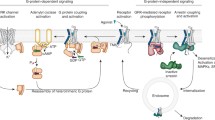
The complex of G protein-coupled receptors (GPCR) and G proteins is the core assembly in GPCR signaling in eukaryotes. With the recent development of cryo-electron microscopy, there has been a rapid growth in structures of GPCR–G protein complexes solved to near-atomic resolution, giving important insights into this signaling complex. Here we describe the biochemical protocol to study the interaction between GPCRs and G proteins before preparation of GPCR–G protein complexes for structural studies. We use gel filtration to analyze the binding properties between GPCR and G protein with the presence of agonist or antagonist, as well as the complex dissociation in the presence of GTP analogue. Methods used in the protocol are affinity purification and gel filtration, which are also commonly used in protein sample preparation for structural work. Therefore, the protocol can be easily adapted for large-scale sample preparation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Stevens RC, Cherezov V, Katritch V et al (2013) The GPCR network: a large-scale collaboration to determine human GPCR structure and function. Nat Rev Drug Discov 12:25–34. https://doi.org/10.1038/nrd3859
Pándy-Szekeres G, Munk C, Tsonkov TM et al (2018) GPCRdb in 2018: adding GPCR structure models and ligands. Nucleic Acids Res 46:D440–D446. https://doi.org/10.1093/nar/gkx1109
Hilger D, Masureel M, Kobilka BK (2018) Structure and dynamics of GPCR signaling complexes. Nat Struct Mol Biol 25:4–12. https://doi.org/10.1038/s41594-017-0011-7
Kühlbrandt W (2014) Biochemistry. The resolution revolution. Science 343:1443–1444. https://doi.org/10.1126/science.1251652
Bai X, McMullan G, Scheres SH (2015) How cryo-EM is revolutionizing structural biology. Trends Biochem Sci 40:49–57. https://doi.org/10.1016/j.tibs.2014.10.005
Zhao DY, Pöge M, Morizumi T et al (2019) Cryo-EM structure of the native rhodopsin dimer in nanodiscs. J Biol Chem 294:14215–14230. https://doi.org/10.1074/jbc.RA119.010089
Koehl A, Hu H, Feng D et al (2019) Structural insights into the activation of metabotropic glutamate receptors. Nature 566:79–84. https://doi.org/10.1038/s41586-019-0881-4
Liang Y-L, Khoshouei M, Radjainia M et al (2017) Phase-plate cryo-EM structure of a class B GPCR–G-protein complex. Nature:1–18. https://doi.org/10.1038/nature22327
Zhang Y, Sun B, Feng D et al (2017) Cryo-EM structure of the activated GLP-1 receptor in complex with a G protein. Nature 546:248–253. https://doi.org/10.1038/nature22394
Liang Y-L, Khoshouei M, Glukhova A et al (2018) Phase-plate cryo-EM structure of a biased agonist-bound human GLP-1 receptor-Gs complex. Nature 555:121–125. https://doi.org/10.1038/nature25773
García-Nafría J, Lee Y, Bai X et al (2018) Cryo-EM structure of the adenosine A2A receptor coupled to an engineered heterotrimeric G protein. elife 7:e35946. https://doi.org/10.7554/eLife.35946
Koehl A, Hu H, Maeda S et al (2018) Structure of the μ opioid receptor-G i protein complex. Nature 558:1–23. https://doi.org/10.1038/s41586-018-0219-7
Kang Y, Kuybeda O, de Waal PW et al (2018) Cryo-EM structure of human rhodopsin bound to an inhibitory G protein. Nature 558:553–558. https://doi.org/10.1038/s41586-018-0215-y
Draper-Joyce CJ, Khoshouei M, Thal DM et al (2018) Structure of the adenosine-bound human adenosine A1 receptor–Gi complex. Nature 558:559–563. https://doi.org/10.1038/s41586-018-0236-6
García-Nafría J, Nehmé R, Edwards PC, Tate CG (2018) Cryo-EM structure of the serotonin 5-HT1B receptor coupled to heterotrimeric Go. Nature 558:620–623. https://doi.org/10.1038/s41586-018-0241-9
Liang Y-L, Khoshouei M, Deganutti G et al (2018) Cryo-EM structure of the active, Gs-protein complexed, human CGRP receptor. Nature 561:492–497. https://doi.org/10.1038/s41586-018-0535-y
Krishna Kumar K, Shalev-Benami M, Robertson MJ et al (2019) Structure of a signaling cannabinoid receptor 1-G protein complex. Cell:1–11. https://doi.org/10.1016/J.CELL.2018.11.040
Zhao L-H, Ma S, Sutkeviciute I et al (2019) Structure and dynamics of the active human parathyroid hormone receptor-1. Science 364:148–153. https://doi.org/10.1126/science.aav7942
Maeda S, Qu Q, Robertson MJ et al (2019) Structures of the M1 and M2 muscarinic acetylcholine receptor/G-protein complexes. Science 364:552–557. https://doi.org/10.1126/science.aaw5188
Qi X, Liu H, Thompson B et al (2019) Cryo-EM structure of oxysterol-bound human smoothened coupled to a heterotrimeric Gi. Nature. https://doi.org/10.1038/s41586-019-1286-0
Kato HE, Zhang Y, Hu H et al (2019) Conformational transitions of a neurotensin receptor 1-Gi1 complex. Nature 572:80–85. https://doi.org/10.1038/s41586-019-1337-6
Tsai C-J, Marino J, Adaixo R et al (2019) Cryo-EM structure of the rhodopsin-Gαi-βγ complex reveals binding of the rhodopsin C-terminal tail to the gβ subunit. elife 8:547919. https://doi.org/10.7554/eLife.46041
Gao Y, Hu H, Ramachandran S et al (2019) Structures of the rhodopsin-transducin complex: insights into G-protein activation. Mol Cell 75:781–790.e3. https://doi.org/10.1016/j.molcel.2019.06.007
Zhao P, Liang Y-L, Belousoff MJ et al (2020) Activation of the GLP-1 receptor by a non-peptidic agonist. Nature 577:432–436. https://doi.org/10.1038/s41586-019-1902-z
Yin W, Li Z, Jin M et al (2019) A complex structure of arrestin-2 bound to a G protein-coupled receptor. Cell Res 29:971–983. https://doi.org/10.1038/s41422-019-0256-2
Nguyen AH, Thomsen ARB, Cahill TJ et al (2019) Structure of an endosomal signaling GPCR–G protein–β-arrestin megacomplex. Nat Struct Mol Biol 26:1123–1131. https://doi.org/10.1038/s41594-019-0330-y
Simon MI, Strathmann MP, Gautam N (1991) Diversity of G proteins in signal transduction. Science 252:802–808. https://doi.org/10.1126/science.1902986
Flock T, Hauser AS, Lund N et al (2017) Selectivity determinants of GPCR-G-protein binding. Nature 545:317–322. https://doi.org/10.1038/nature22070
Deupi X, Edwards P, Singhal A et al (2012) Stabilized G protein binding site in the structure of constitutively active metarhodopsin-II. Proc Natl Acad Sci 109:119–124. https://doi.org/10.1073/pnas.1114089108
Sun D, Flock T, Deupi X et al (2015) Probing Gα i1 protein activation at single-amino acid resolution. Nat Struct Mol Biol 22:686–694. https://doi.org/10.1038/nsmb.3070
Maeda S, Sun D, Singhal A et al (2014) Crystallization scale preparation of a stable GPCR signaling complex between constitutively active rhodopsin and G-protein. PLoS One 9:e98714. https://doi.org/10.1371/journal.pone.0098714
Molday LL, Molday RS (2014) 1D4: a versatile epitope tag for the purification and characterization of expressed membrane and soluble proteins. Methods Mol Biol 1177:1–15. https://doi.org/10.1007/978-1-4939-1034-2_1
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Pamula, F., Tsai, CJ. (2021). Biochemical Characterization of GPCR–G Protein Complex Formation. In: Schmidt-Krey, I., Gumbart, J.C. (eds) Structure and Function of Membrane Proteins. Methods in Molecular Biology, vol 2302. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1394-8_3
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1394-8_3
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1393-1
Online ISBN: 978-1-0716-1394-8
eBook Packages: Springer Protocols