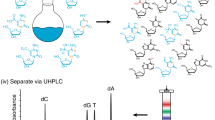
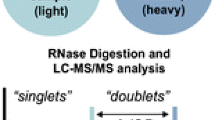
Abstract
Recent progress in epitranscriptome research shows an interplay of enzymes modifying RNAs and enzymes dedicated for RNA modification removal. One of the main techniques to study RNA modifications is liquid chromatography-coupled tandem mass spectrometry (LC-MS/MS) as it allows sensitive detection of modified nucleosides. Although RNA modifications have been found to be highly dynamic, state-of-the-art LC-MS/MS analysis only gives a static view on modifications and does not allow the investigation of temporal modification placement. Here, we present the principles of nucleic acid isotope labeling coupled with mass spectrometry, termed NAIL-MS, which overcomes these limitations by stable isotope labeling in human cell culture and gives detailed instructions on how to label cells and process samples in order to get reliable results. For absolute quantification in the context of NAIL-MS, we explain the production of internal standards in detail. Furthermore, we outline the requirements for stable isotope labeling in cell culture and all subsequent steps to receive nucleoside mixtures of native RNA for NAIL-MS analysis. In the final section of this chapter, we describe the distinctive features of NAIL-MS data analysis with a special focus toward absolute quantification of modified nucleosides.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Heiss M, Kellner S (2016) Detection of nucleic acid modifications by chemical reagents. RNA Biol 0. https://doi.org/10.1080/15476286.2016.1261788
Motorin Y, Helm M (2019) Methods for RNA modification mapping using deep sequencing: established and new emerging technologies. Genes (Basel) 10(1). https://doi.org/10.3390/genes10010035
Helm M, Lyko F, Motorin Y (2019) Limited antibody specificity compromises epitranscriptomic analyses. Nat Commun 10(1):5669. https://doi.org/10.1038/s41467-019-13684-3
Brandmayr C, Wagner M, Bruckl T, Globisch D, Pearson D, Kneuttinger AC, Reiter V, Hienzsch A, Koch S, Thoma I, Thumbs P, Michalakis S, Muller M, Biel M, Carell T (2012) Isotope-based analysis of modified tRNA nucleosides correlates modification density with translational efficiency. Angew Chem Int Ed Engl 51(44):11162–11165. https://doi.org/10.1002/anie.201203769
Kellner S, Ochel A, Thuring K, Spenkuch F, Neumann J, Sharma S, Entian KD, Schneider D, Helm M (2014) Absolute and relative quantification of RNA modifications via biosynthetic isotopomers. Nucleic Acids Res 42(18):e142. https://doi.org/10.1093/nar/gku733
Thuring K, Schmid K, Keller P, Helm M (2017) LC-MS analysis of methylated RNA. Methods Mol Biol 1562:3–18. https://doi.org/10.1007/978-1-4939-6807-7_1
Borland K, Diesend J, Ito-Kureha T, Heissmeyer V, Hammann C, Buck AH, Michalakis S, Kellner S (2019) Production and application of stable isotope-labeled internal standards for RNA modification analysis. Genes (Basel) 10(1). https://doi.org/10.3390/genes10010026
Reichle VF, Petrov DP, Weber V, Jung K, Kellner S (2019) NAIL-MS reveals the repair of 2-methylthiocytidine by AlkB in E. coli. Nat Commun 10(1):5600. https://doi.org/10.1038/s41467-019-13565-9
Heiss M, Reichle VF, Kellner S (2017) Observing the fate of tRNA and its modifications by nucleic acid isotope labeling mass spectrometry: NAIL-MS. RNA Biol 14(9):1260–1268. https://doi.org/10.1080/15476286.2017.1325063
Heiss M, Hagelskamp F, Kellner S (2020) Cell culture NAIL-MS allows insight into human RNA modification dynamics in vivo. bioRxiv. https://doi.org/10.1101/2020.04.28.067314
Thumbs P, Ensfelder TT, Hillmeier M, Wagner M, Heiss M, Scheel C, Schon A, Muller M, Michalakis S, Kellner S, Carell T (2020) Synthesis of galactosyl-queuosine and distribution of hypermodified Q-nucleosides in mouse tissues. Angew Chem Int Ed Engl. https://doi.org/10.1002/anie.202002295
Chionh YH, Ho CH, Pruksakorn D, Ramesh Babu I, Ng CS, Hia F, McBee ME, Su D, Pang YL, Gu C, Dong H, Prestwich EG, Shi PY, Preiser PR, Alonso S, Dedon PC (2013) A multidimensional platform for the purification of non-coding RNA species. Nucleic Acids Res 41(17):e168. https://doi.org/10.1093/nar/gkt668
Hagelskamp F, Borland K, Ramos J, Hendrick AG, Fu D, Kellner S (2020) Broadly applicable oligonucleotide mass spectrometry for the analysis of RNA writers and erasers in vitro. Nucleic Acids Res 48(7):e41. https://doi.org/10.1093/nar/gkaa091
Hauenschild R, Tserovski L, Schmid K, Thuring K, Winz ML, Sharma S, Entian KD, Wacheul L, Lafontaine DL, Anderson J, Alfonzo J, Hildebrandt A, Jaschke A, Motorin Y, Helm M (2015) The reverse transcription signature of N-1-methyladenosine in RNA-Seq is sequence dependent. Nucleic Acids Res 43(20):9950–9964. https://doi.org/10.1093/nar/gkv895
Collart MA, Oliviero S (2001) Preparation of yeast RNA. Curr Protoc Mol Biol Chapter 13:Unit13 12. https://doi.org/10.1002/0471142727.mb1312s23
Acknowledgments
This study was funded through the Deutsche Forschungsgemeinschaft (KE1943/3-1, KE1943/4-1–SPP1784, and Project-ID 325871075–SFB 1309). We are grateful to Prof. Peter Dedon, Prof. Mark Helm, and Prof. Thomas Carell for generous donation of synthetic standards of modified nucleosides.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Heiss, M., Borland, K., Yoluç, Y., Kellner, S. (2021). Quantification of Modified Nucleosides in the Context of NAIL-MS. In: McMahon, M. (eds) RNA Modifications. Methods in Molecular Biology, vol 2298. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1374-0_18
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1374-0_18
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1373-3
Online ISBN: 978-1-0716-1374-0
eBook Packages: Springer Protocols