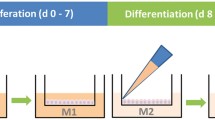
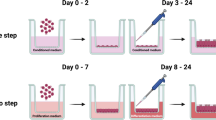
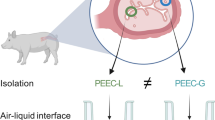
Abstract
Oviduct and uterus are key female reproductive organs lined by ciliated simple columnar epithelia, which are the first line of maternal contact with gametes and the developing embryo during reproduction and which warrant the optimal developmental environment for the conceptus. A major challenge for modeling these epithelia in vitro is the preservation of apical–basal polarization and cilia formation. The air–liquid interface (ALI) culture approach is a technology originally invented for modeling epidermal and airway epithelia. It has recently been shown that it also allows the establishment of highly differentiated in vitro models of epithelia that do not have access to ambient air in vivo. In this chapter, we present a comprehensive ALI procedure to model female reproductive tract (FRT) epithelia of different mammalian species in vitro over extended time periods. As a working example, the protocol focuses on primary oviductal epithelial cells (OEC) isolated from domestic pig. Hints on protocol variations for the culture of OEC from other species are provided in the Subheading 4.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Wira CR, Grant-Tschudy KS, Crane-Godreau MA (2005) Epithelial cells in the female reproductive tract: a central role as sentinels of immune protection. Am J Reprod Immunol 53(2):65–76. https://doi.org/10.1111/j.1600-0897.2004.00248.x
Kurita T (2011) Normal and abnormal epithelial differentiation in the female reproductive tract. Differentiation 82(3):117–126. https://doi.org/10.1016/j.diff.2011.04.008
Haycock JW (2011) 3D cell culture: a review of current approaches and techniques. Methods Mol Biol 695:1–15. https://doi.org/10.1007/978-1-60761-984-0_1
Jensen C, Teng Y (2020) Is it time to start transitioning from 2D to 3D cell culture? Front Mol Biosci 7:33. https://doi.org/10.3389/fmolb.2020.000334
Kessler M, Hoffmann K, Brinkmann V, Thieck O, Jackisch S, Toelle B, Berger H, Mollenkopf HJ, Mangler M, Sehouli J, Fotopoulou C, Meyer TF (2015) The Notch and Wnt pathways regulate stemness and differentiation in human fallopian tube organoids. Nat Commun 6:8989. https://doi.org/10.1038/ncomms9989
Turco MY, Gardner L, Hughes J, Cindrova-Davies T, Gomez MJ, Farrell L, Hollinshead M, Marsh SGE, Brosens JJ, Critchley HO, Simons BD, Hemberger M, Koo BK, Moffett A, Burton GJ (2017) Long-term, hormone-responsive organoid cultures of human endometrium in a chemically defined medium. Nat Cell Biol 19(5):568–577. https://doi.org/10.1038/ncb3516
Chen S, Schoen J (2019) Air-liquid interface cell culture: from airway epithelium to the female reproductive tract. Reprod Domest Anim 54(Suppl 3):38–45. https://doi.org/10.1111/rda.13481
Chen S, Einspanier R, Schoen J (2015) Transepithelial electrical resistance (TEER): a functional parameter to monitor the quality of oviduct epithelial cells cultured on filter supports. Histochem Cell Biol 144(5):509–515. https://doi.org/10.1007/s00418-015-1351-1
Chen S, Einspanier R, Schoen J (2013) In vitro mimicking of estrous cycle stages in porcine oviduct epithelium cells: estradiol and progesterone regulate differentiation, gene expression, and cellular function. Biol Reprod 89(3):54. https://doi.org/10.1095/biolreprod.113.108829
Chen S, Palma-Vera SE, Kempisty B, Rucinski M, Vernunft A, Schoen J (2018) In vitro mimicking of estrous cycle stages: dissecting the impact of estradiol and progesterone on oviduct epithelium. Endocrinology 159(9):3421–3432. https://doi.org/10.1210/en.2018-00567
Du S, Trakooljul N, Schoen J, Chen S (2020) Does maternal stress affect the early embryonic microenvironment? Impact of long-term cortisol stimulation on the oviduct epithelium. Int J Mol Sci 21(2):443. https://doi.org/10.3390/ijms21020443
Miessen K, Sharbati S, Einspanier R, Schoen J (2011) Modelling the porcine oviduct epithelium: a polarized in vitro system suitable for long-term cultivation. Theriogenology 76(5):900–910. https://doi.org/10.1016/j.theriogenology.2011.04.021
Palma-Vera SE, Schoen J, Chen S (2017) Periovulatory follicular fluid levels of estradiol trigger inflammatory and DNA damage responses in oviduct epithelial cells. PLoS One 12(2):e0172192. https://doi.org/10.1371/journal.pone.0172192
Chen S, Palma-Vera SE, Langhammer M, Galuska SP, Braun BC, Krause E, Lucas-Hahn A, Schoen J (2017) An air-liquid interphase approach for modeling the early embryo-maternal contact zone. Sci Rep 7:42298. https://doi.org/10.1038/srep42298
van der Weijden VA, Chen S, Bauersachs S, Ulbrich SE, Schoen J (2017) Gene expression of bovine embryos developing at the air-liquid interface on oviductal epithelial cells (ALI-BOEC). Reprod Biol Endocrinol 15(1):91. https://doi.org/10.1186/s12958-017-0310-1
Simopoulou M, Sfakianoudis K, Rapani A, Giannelou P, Anifandis G, Bolaris S, Pantou A, Lambropoulou M, Pappas A, Deligeoroglou E, Pantos K, Koutsilieris M (2018) Considerations regarding embryo culture conditions: from media to epigenetics. In Vivo 32(3):451–460. https://doi.org/10.21873/invivo.11261
Acknowledgments
We thank Lisa Speck, Caterina Poeppel, Petra Reckling, and Bianka Drawert, Leibniz Institute for Farm Animal Biology (FBN) Dummerstorf, Germany, for excellent technical assistance. We appreciate Dagmar Viertel, Leibniz Institute for Zoo and Wildlife research (IZW) Berlin, Germany, for providing electron microscopy pictures. The authors are active members of the COST Action CA16119 (In vitro 3D total cell guidance and fitness). The original work mentioned in this chapter was partially supported by grants from the German Research Foundation (DFG Scho1231/7-1; CH2321/1-1). The Authors are members of the COST Action CA16119 In vitro 3-D total cell guidance and fitness (CellFit).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Science+Business Media, LLC , part of Springer Nature
About this protocol
Cite this protocol
Chen, S., Schoen, J. (2021). Using the Air–Liquid Interface Approach to Foster Apical–Basal Polarization of Mammalian Female Reproductive Tract Epithelia In Vitro. In: Brevini, T.A., Fazeli, A., Turksen, K. (eds) Next Generation Culture Platforms for Reliable In Vitro Models . Methods in Molecular Biology, vol 2273. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1246-0_18
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1246-0_18
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1245-3
Online ISBN: 978-1-0716-1246-0
eBook Packages: Springer Protocols