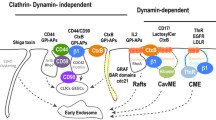
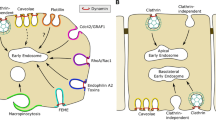
Abstract
Endocytosis and intracellular retrograde trafficking from endosomes to the Golgi apparatus are key cellular processes. Endocytosis is directly or indirectly involved in many if not all cellular functions ranging from nutrient uptake and receptor signaling to mitosis, cell division, and migration (Scita, Di Fiore. Nature 463(7280):464–473, 2010; McMahon, Boucrot. Nat Rev Mol Cell Biol 12(8):517–533, 2011). Retrograde trafficking is emerging as a key driver for cell polarity. Robust methods are needed to quantify these processes. At the example of the bacterial Shiga toxin and the endogenous α5β1 integrin, we here describe generic methods to differentiate (1) internalized from cell surface-accessible cargo proteins and (2) endocytic cargo proteins that have reached the Golgi apparatus via the retrograde route from those that have not. The choice of antibodies or natural ligands allows to adjust these methods to virtually any chosen biological system.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Scita G, Di Fiore PP (2010) The endocytic matrix. Nature 463(7280):464–473. https://doi.org/10.1038/nature08910
McMahon HT, Boucrot E (2011) Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat Rev Mol Cell Biol 12(8):517–533. https://doi.org/10.1038/nrm3151
Kumari S, Mg S, Mayor S (2010) Endocytosis unplugged: multiple ways to enter the cell. Cell Res 20(3):256–275. https://doi.org/10.1038/cr.2010.19
Doherty GJ, McMahon HT (2009) Mechanisms of endocytosis. Annu Rev Biochem 78:857–902. https://doi.org/10.1146/annurev.biochem.78.081307.110540
Moreno-Layseca P, Icha J, Hamidi H, Ivaska J (2019) Integrin trafficking in cells and tissues. Nat Cell Biol 21(2):122–132. https://doi.org/10.1038/s41556-018-0223-z
Sandvig K, Grimmer S, Lauvrak SU, Torgersen ML, Skretting G, van Deurs B, Iversen TG (2002) Pathways followed by ricin and Shiga toxin into cells. Histochem Cell Biol 117(2):131–141. https://doi.org/10.1007/s00418-001-0346-2
Jovic M, Sharma M, Rahajeng J, Caplan S (2010) The early endosome: a busy sorting station for proteins at the crossroads. Histol Histopathol 25(1):99–112. https://doi.org/10.14670/HH-25.99
Johannes L, Popoff V (2008) Tracing the retrograde route in protein trafficking. Cell 135(7):1175–1187. https://doi.org/10.1016/j.cell.2008.12.009
Nonnenmacher ME, Cintrat JC, Gillet D, Weber T (2015) Syntaxin 5-dependent retrograde transport to the trans-Golgi network is required for adeno-associated virus transduction. J Virol 89(3):1673–1687. https://doi.org/10.1128/JVI.02520-14
Schelhaas M, Ewers H, Rajamaki ML, Day PM, Schiller JT, Helenius A (2008) Human papillomavirus type 16 entry: retrograde cell surface transport along actin-rich protrusions. PLoS Pathog 4(9):e1000148. https://doi.org/10.1371/journal.ppat.1000148
Carpier JM, Zucchetti AE, Bataille L, Dogniaux S, Shafaq-Zadah M, Bardin S, Lucchino M, Maurin M, Joannas LD, Magalhaes JG, Johannes L, Galli T, Goud B, Hivroz C (2018) Rab6-dependent retrograde traffic of LAT controls immune synapse formation and T cell activation. J Exp Med 215(4):1245–1265. https://doi.org/10.1084/jem.20162042
Shafaq-Zadah M, Gomes-Santos CS, Bardin S, Maiuri P, Maurin M, Iranzo J, Gautreau A, Lamaze C, Caswell P, Goud B, Johannes L (2016) Persistent cell migration and adhesion rely on retrograde transport of beta(1) integrin. Nat Cell Biol 18(1):54–64. https://doi.org/10.1038/ncb3287
Su GF, Brahmbhatt HN, Wehland J, Rohde M, Timmis KN (1992) Construction of stable LamB-Shiga toxin B subunit hybrids: analysis of expression in salmonella typhimurium aroA strains and stimulation of B subunit-specific mucosal and serum antibody responses. Infect Immun 60(8):3345–3359
Shi G, Azoulay M, Dingli F, Lamaze C, Loew D, Florent JC, Johannes L (2012) SNAP-tag based proteomics approach for the study of the retrograde route. Traffic 13(7):914–925. https://doi.org/10.1111/j.1600-0854.2012.01357.x
Johannes L, Shafaq-Zadah M (2013) SNAP-tagging the retrograde route. Methods Cell Biol 118:139–155. https://doi.org/10.1016/B978-0-12-417164-0.00009-4
Keppler A, Gendreizig S, Gronemeyer T, Pick H, Vogel H, Johnsson K (2003) A general method for the covalent labeling of fusion proteins with small molecules in vivo. Nat Biotechnol 21(1):86–89. https://doi.org/10.1038/nbt765
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Shafaq-Zadah, M., Dransart, E., Johannes, L. (2021). Quantitative Methods to Study Endocytosis and Retrograde Transport of Cargo Proteins. In: Niedergang, F., Vitale, N., Gasman, S. (eds) Exocytosis and Endocytosis. Methods in Molecular Biology, vol 2233. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1044-2_4
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1044-2_4
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1043-5
Online ISBN: 978-1-0716-1044-2
eBook Packages: Springer Protocols