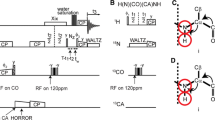
Abstract
Determining hydrogen exchange kinetics in proteins can shed light on their structure and dynamics. Nuclear magnetic resonance (NMR) spectroscopy is an important analytical technique to determine exchange rates. In this chapter, we describe a new method (Paris-DÉCOR) to determine fast protein amide backbone hydrogen exchange rates in the range 10 to 104 s−1. Measuring fast exchange rates is particularly important for the study of intrinsically disordered proteins, where there is very little protection from exchange to the solvent by the formation of persistent structure. We provide a protocol to set up the experiment as well as MATLAB scripts for numerical simulation that is needed to determine the exchange rates.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Dass R, Corlianò E, Mulder FAA (2019) Measurement of very fast exchange rates of individual amide protons in proteins by NMR spectroscopy. ChemPhysChem 20(2):231–235
Skrynnikov NR, Ernst RR (1999) Detection of intermolecular chemical exchange through decorrelation of two-spin order. J Magn Reson 137:276–280
Kateb F, Pelupessy P, Bodenhausen G (2007) Measuring fast hydrogen exchange rates by NMR spectroscopy. J Magn Reson 184(1):108–113
Carr HY, Purcell EM (1954) Effects of diffusion on free precession in nuclear magnetic resonance experiments. Phys Rev 94(3):630–638
Meiboom S, Gill D (1958) Modified spin-Echo method for measuring nuclear relaxation times. Rev Sci Instrum 29(8):688–691
Teng Q (2013) Structural biology: practical NMR applications, 2nd edn. Springer, Dordrecht
Delaglio F, Grzesiek S, Vuister G, et al (1995) NMRPipe: a multidimensional spectral processing system based on UNIX pipes. J Biomol NMR 6(3):277–293
The MathWorks, Inc. MATLAB and Statistics Toolbox Release 2012b. Natick, Massachusetts, United States
Goddard TD, Kneller DG SPARKY 3. University of California: San Francisco
Lerche I, Mudford BS (2005) How many Monte Carlo simulations does one need to do? Energy Explor Exploit 23(6):405–427
Acknowledgments
This work was supported by a postdoctoral fellowship to R.D. from the VILLUM Foundation.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Dass, R., Mulder, F.A.A. (2020). Paris-DÉCOR: A Protocol for the Determination of Fast Protein Backbone Amide Hydrogen Exchange Rates. In: Kragelund, B.B., Skriver, K. (eds) Intrinsically Disordered Proteins. Methods in Molecular Biology, vol 2141. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0524-0_17
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0524-0_17
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0523-3
Online ISBN: 978-1-0716-0524-0
eBook Packages: Springer Protocols