Abstract
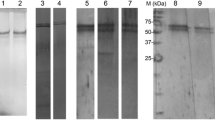
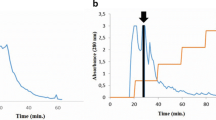
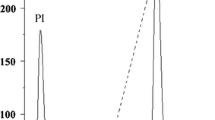
Tachylectin-5, a 41-kDa protein with a common fold of the C-terminal globular domain of the γ-chain of fibrinogen, is purified from horseshoe crab hemolymph plasma by affinity column chromatography, using acetyl-group-immobilized resin. Two types of isolectins, tachylectin-5A and tachylectin-5B, are obtained by stepwise elution with GlcNAc at 25 and 250 mM, respectively. Tachylectins-5A and -5B exhibit extraordinarily strong hemagglutinating activity against all types of human erythrocytes (the minimum agglutinating concentration of 0.004–0.008 μg/mL for tachylectin-5A and 0.077–0.27 μg/mL for tachylectin-5B). Their hemagglutinating activities are inhibited by acetyl group-containing sugars and noncarbohydrates such as sodium acetate, acetylcholine, and acetyl CoA (the minimum inhibitory concentrations of 1.3–1.6 mM), indicating that the acetyl group is required and sufficient for recognition by tachylectins-5A and -5B. EDTA inhibits their hemagglutinating activity, whereas the inhibition is overcome by adding an excess amount of Ca2+. Tachylectins-5A and -5B also exhibit bacterial agglutinating activity against both Gram-negative bacteria (the minimum agglutinating concentrations of 0.04–0.08 μg/mL for tachylectin-5A and 0.05–0.11 μg/mL for tachylectin-5B) and Gram-positive bacteria (the minimum agglutinating concentrations of 0.3–2.4 μg/mL for tachylectin-5A and 15.1–26.8 μg/mL for tachylectin-5B). Interestingly, tachylectins-5A and -5B enhance the antimicrobial activity of a hemocyte-derived peptide, big defensin.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Gokudan S, Muta T, Tsuda R et al (1999) Horseshoe crab acetyl group-recognizing lectins involved in innate immunity are structurally related to fibrinogen. Proc Natl Acad Sci U S A 96:10086–10091
Kawabata S, Tsuda R (2002) Molecular basis of non-self recognition by the horseshoe crab tachylectins. Biochim Biophys Acta 1572:414–421
Iwanaga S (2002) The molecular basis of innate immunity in the horseshoe crab. Curr Opin Immunol 14:87–95
Kawabata S (2011) Immunocompetent molecules and their response network in horseshoe crabs. In: Söderhäll K (ed) Invertebrate immunity. Springer Science + Business Media, New York, pp 122–136
Kairies N, Beisel H-G, Fuentes-Prior P et al (2001) The 2.0-Å crystal structure of tachylectin 5 provides evidence for the common origin of the innate immunity and the blood coagulation systems. Proc Natl Acad Sci U S A 98:13519–13524
Xu X, Doolittle RF (1990) Presence of vertebrate fibrinogen-like sequence in an echinoderm. Proc Natl Acad Sci U S A 87:2097–2101
Adema CM, Hertel LA, Miller RD, Loker ES (1997) A family of fibrinogen-related proteins that precipitates parasite-derived molecule is produced by an invertebrate after infection. Proc Natl Acad Sci U S A 94:8691–8696
Kurachi S, Song Z, Takagaki M et al (1998) Sialic-acid binding lectin from the slug Limax flavus: cloning, expression of the polypeptide, and tissue localization. Eur J Biochem 254:217–222
Dimopoulos G, Casavant TL, Chang SR et al (2000) Anopheles gambiae pilot gene discovery project: identification of mosquito innate immunity genes from expressed sequence tags generated from immune-competent cell lines. Proc Natl Acad Sci U S A 97:6619–6624
Kenjo A, Takahashi M, Matsushita M et al (2001) Cloning and characterization of novel ficolins from the solitary ascidian, Halocynthia roretzi. J Biol Chem 276:19959–19965
Leonard PM, Adema CM, Zhang S-MLoker ES (2001) Structure of two FREP genes that combine IgSF and fibrinogen domains, with comments on diversity of the FREP gene family in the snail. Biomphalaria glabrata. Gene 269:155–165
Angthong P, Roytrakul S, Jarayabhand P, Jiravanichpaisal P (2017) Characterization and function of a tachylectin 5-like immune molecule in Penaeus monodon. Dev Comp Immunol 76:120–131
Saito T, Kawabata S, Shigenaga T et al (1995) A novel big defensin identified in horseshoe crab hemocytes: isolation, amino acid sequence, and antibacterial activity. J Biochem 117:1131–1137
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Kawabata, Si., Shibata, T. (2020). Purification and Assays of Tachylectin-5. In: Hirabayashi, J. (eds) Lectin Purification and Analysis. Methods in Molecular Biology, vol 2132. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0430-4_27
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0430-4_27
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0429-8
Online ISBN: 978-1-0716-0430-4
eBook Packages: Springer Protocols