Abstract
In order to study the molecular pathways of Parkinson’s disease (PD) and to develop novel therapeutic strategies, scientific investigators rely on animal models. The identification of PD-associated genes has led to the development of genetic PD models as an alternative to toxin-based models. Viral vector-mediated loco-regional gene delivery provides an attractive way to express transgenes in the central nervous system. Several vector systems based on various viruses have been developed. In this chapter, we give an overview of the different viral vector systems used for targeting the CNS. Further, we describe the different viral vector-based PD models currently available based on overexpression strategies for autosomal dominant genes such as α-synuclein and LRRK2, and knockout or knockdown strategies for autosomal recessive genes, such as parkin, DJ-1, and PINK1. Models based on overexpression of α-synuclein are the most prevalent and extensively studied, and therefore the main focus of this chapter. Many efforts have been made to increase the expression levels of α-synuclein in the dopaminergic neurons. The best α-synuclein models currently available have been developed from a combined approach using newer AAV serotypes and optimized vector constructs, production, and purification methods. These third-generation α-synuclein models show improved face and predictive validity, and therefore offer the possibility to reliably test novel therapeutics.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Parkinson’s disease (PD) is the second most common age-related movement disorder. It is estimated that PD affects 1 % of the population at 65 years of age, which increases to 4–5 % in 85-year olds. To date, more than 4.5 million people suffer from PD worldwide, and this number is expected to double to 9 million in the next 20 years due to the improved living conditions and the increased life expectancy (Wirdefeldt et al. 2011). PD is a slowly progressing disorder that affects the dopaminergic neurons (DN) of the substantia nigra pars compacta (SNpc), a small nucleus in the midbrain. The loss of dopaminergic innervation leads to hyperactivity of the subthalamic nucleus and the globus pallidus, resulting in the progressive decline of movement control, which closely correlates with the degree of SNpc cell loss (Ma et al. 1997). At the onset of the motor symptoms, the striatal dopamine (DA) levels are already depleted by 80 % and approximately 50–70 % of the DN in the SNpc has been lost (Dauer and Przedborski 2003). However, the traditional view that the neuropathology of PD is restricted to nigral degeneration has been abandoned. For instance, Braak and colleagues define a six-stage pathological process where PD pathology starts in the olfactory bulb and the dorsal motor nucleus of the vagal nerve and extends to the midbrain and other brainstem regions in the later stages of the disease process (Braak et al. 2003; Hawkes et al. 2010). Next to the selective loss of the DN in the SN, PD is characterized by the presence of Lewy bodies (LBs) and Lewy neurites (LNs) in the surviving neurons. These cytoplasmic aggregates are predominantly composed of fibrillar forms of the protein α-synuclein , and can be found in several affected brain regions of most PD patients (Spillantini et al. 1997; Shults 2006). Currently, most treatments of PD are focused on the symptomatic improvement of motor symptoms related to the loss of the DN in the SNpc. These symptomatic treatments (pharmacological and surgical) significantly reduce PD-associated motor symptoms and improve quality of life and life expectancy. Still, the development of therapeutic approaches that slow or halt the disease progression, as well as the identification of reliable biomarkers for early diagnosis remains crucial.
Historically, PD has been considered a purely sporadic disorder without a clear etiology. Environmental factors (e.g., pesticides exposure, living in a rural area, and well water consumption) have been proposed as important risk factors or triggers for PD in the context of an aging brain. However, in the past decade, genetic studies of PD families from different geographical regions worldwide have strengthened the hypothesis that PD has a substantial genetic compound. The first gene linked to PD (SNCA, encoding for α-synuclein ) was discovered through analysis of a large multigenerational Greek/Italian family, in which PD segregated in an autosomal dominant pattern (Polymeropoulos et al. 1997). Since then, 18 PD loci have been nominated through linkage analysis (PARK 1–15) or genome-wide association studies (PARK 16–18) (Farrer 2006; Belin and Westerlund 2008; Satake et al. 2009; Simon-Sanchez et al. 2009; Pankratz et al. 2009; Hamza et al. 2010). Mutations within the genes of six of these loci (SNCA, LRRK2, PRKN, PINK1, DJ-1, and ATP13A2) have conclusively been demonstrated to cause familial PD (Lesage and Brice 2009). In addition, common polymorphisms within two of the same genes (SNCA and LRRK2) are well validated risk factors for PD (Paisan-Ruiz 2009; Edwards et al. 2010). Although these familial forms of PD account for only 5–10 % of all PD patients, unraveling of the molecular pathways underlying familial forms of PD will greatly contribute to our understanding of sporadic PD since both forms share clinical and neuropathological features.
To study the molecular pathways of PD and to develop novel therapeutic strategies, investigators rely on animal models . However, to reliably translate the results from animal experiments to the human disease, one has to consider “the validity” of the animal model being used. In general, three criteria are taken into consideration: The face, predictive, and construct validity. The “face validity” refers to a similar pathophysiology; do the symptoms and pathology observed in the animal model resemble the ones seen in human patients? Face validity can sometimes be distracting because behavioral manifestations in rodents and people might differ. The “predictive validity” suggests that a treatment that is effective in the model will also be successful in patients. A model based on an established cause of the disease has “construct validity” and is useful in the understanding of the pathophysiological mechanism. A perfect animal model for PD should display age-dependent and progressive degeneration of DN, the cardinal motor symptoms of PD as well as nonmotor dysfunctions and LB pathology in surviving nigral cells and eventually other brain regions (high face validity). Furthermore, it should be responsive to dopamine replacement strategies (high predictive validity) and would be based on a known cause of the disease, e.g., genetic mutation causing familial PD (high construct validity). Although such a perfect PD model does not exist, significant progress has been made over the last 15 years. Currently, PD models are based on toxicological, transgenic, or viral vector-based stereotactic gene delivery approaches.
2 Toxin-Based Animal Models
Toxin models are the classical and the oldest experimental PD models. They aim at reproducing the pathological and behavioral changes of the human disease in rodents or primates by using neurotoxins (e.g., 6-OHDA, MPTP), which selectively accumulate in the substantia nigra DN, causing cellular dysfunction and cell death (Ungerstedt and Arbuthnott 1970; Betarbet et al. 2002). These toxins can be administered either locally or systemically depending on the type of toxin used and the species involved (Bezard and Przedborski 2011). The unilateral 6-OHDA rat model has been used extensively as a preclinical model to assess the antiparkinsonian effects of new pharmacological treatments (Jiang et al. 1993; Ilijic et al. 2011; Bjorklund et al. 2002; Kirik et al. 2002). Studies using MPTP have led to concepts such as “environmental toxicity” as a potential cause of dysfunction in sporadic PD and “mitochondrial dysfunction” as a potential pathogenic mechanism (Fox and Brotchie 2010). However, a major limitation of these toxin models is that they induce rather acute effects, which differ significantly from the slowly progressive pathology of human PD. Moreover, LB pathology is not present in the surviving neurons, and no other brain areas involved in PD are affected. Later, a variety of other toxic compounds (pesticides) like rotenone, paraquat, and maneb were tested in vivo in an attempt to find additional and improved toxin models (Hoglinger et al. 2006; Thiruchelvam et al. 2000). Interestingly, ubiquitine-positive LB-like inclusions were observed in the surviving nigral neurons of rotenone-treated animals (Betarbet et al. 2000). Despite these positive features, the substantial variability observed in all of these pesticide models limits their use for therapeutical development.
3 Transgenic Mouse Models
The discovery of α-synuclein as the first PARK gene in 1997 led to the development of the first genetic PD models. The A53T mutation in α-synuclein was identified first in a Greek/Italian family (Polymeropoulos et al. 1997). Later, the A30P mutation and later the E46K mutation were identified in a German and Spanish family (Kruger et al. 1998; Zarranz et al. 2004). Shortly after these findings, it was realized that an aggregated form of α-synuclein forms the main constituent of LBs in both sporadic and familial PD cases (Spillantini et al. 1997). In addition, duplications and triplications of the entire gene locus have been reported in different families (Chartier-Harlin et al. 2004; Ibanez et al. 2004; Singleton et al. 2003). Interestingly, gene triplication leads to earlier onset and faster progression of disease than duplication, indicating that disease severity is dependent on α-synuclein expression levels. Altogether, these findings provide strong evidence that α-synuclein plays an important role in the pathogenesis of PD. For these reasons, it was hypothesized that overexpression of α-synuclein in the brain may lead to pathological features reminiscent of PD such as neuronal cell death and the accumulation of cellular inclusions. Many transgenic mice overexpressing human wild-type (WT) or mutant (A30P, A53T) α-synuclein have been generated over the last 10 years. The promoter driving α-synuclein expression has proven to be crucial in the development of the pathology. Other significant variations are the mouse strain, the presence or absence of endogenous α-synuclein, and whether the full length or the truncated form is expressed (detailed review by Magen and Chesselet 2010). Overexpression of both WT and several clinical mutants of human α-synuclein in transgenic mice have been shown to induce pathological accumulation of α-synuclein and neuronal dysfunction (Masliah et al. 2000; Freichel et al. 2007; Fleming et al. 2005; Kahle et al. 2001; Chesselet and Richter 2011). However, until now most transgenic α-synuclein mouse models failed to display clear dopaminergic cell loss and dopamine-dependent behavioral deficits. Some transgenic models expressing the double mutated (Thiruchelvam et al. 2004; Richfield et al. 2002; Chen et al. 2006) or truncated (Tofaris et al. 2006; Shelkovnikova et al. 2011; Wakamatsu et al. 2008) α-synuclein gene have reported dopaminergic cell loss, but the clinical relevance of these models is questionable.
Later, the identification of other PD-associated genes has led to the development of more genetic PD models. Overexpression strategies for autosomal dominant genes such as LRRK2 and knockout strategies for autosomal recessive genes such as Parkin, DJ-1, and PINK1 were used. Current transgenic LRRK2 mouse models are not very robust PD models. BAC transgenic mice expressing R1441G, G2019S LRRK2 present minimal evidence of neurodegeneration (Li et al. 2009, 2010). When the R1441C mutation is expressed under the control of endogenous regulatory elements, by knock-in of the R1441C mutation, no degeneration of DN is observed (Tong et al. 2009). For one R1441G-LRRK2 transgenic mouse abnormalities in the nigrostriatal system such as stimulated DA neurotransmission and l-dopa responsive behavioral defects have been described, but no DN loss was reported (Li et al. 2009). Recently, mice expressing the G2019S mutant showed loss of DN and dendrites at 19–20 months of age (Ramonet et al. 2011).
None of the parkin KO mice have substantial dopaminergic or behavioral abnormalities (Itier et al. 2003; Perez and Palmiter 2005; Von Coelln et al. 2004; Goldberg et al. 2003; Zhu et al. 2007). Some have subtle abnormalities in their DA neurotransmission or in the noradrenergic system of the locus coeruleus (Goldberg et al. 2003). Interestingly, overexpression of truncated human parkin (Q311X) led to progressive degeneration of DN (Lu et al. 2009), supporting the idea that some parkin mutants might act in a dominant negative fashion. Similar to the parkin KO mice, PINK-1 KO mice do not show any loss of DN, have normal levels of striatal DA and DA receptors are unchanged in most KO mice (Gautier et al. 2008; Kitada et al. 2007). Mild abnormalities in DA neurotransmission have been described (Kitada et al. 2007). DJ-1 KO mice do not exhibit any major abnormalities, and the number of DN and levels of striatal DA are unchanged. Abnormalities in DA neurotransmission in the nigrostriatal circuit and mitochondrial dysfunction were observed in some DJ-1 KO animals (Goldberg et al. 2005; Kim et al. 2005; Andres-Mateos et al. 2007). To summarize, all parkin, PINK1, and DJ-1 KO animals present only a mild phenotype without clear neurodegeneration.
In conclusion, in some transgenic models based on mutations causing PD, loss of DN has been reported, specifically in mice expressing the double mutated (Thiruchelvam et al. 2004; Richfield et al. 2002; Chen et al. 2006) or truncated (Tofaris et al. 2006; Shelkovnikova et al. 2011; Wakamatsu et al. 2008) α-synuclein gene, a truncated form of parkin (Lu et al. 2009), or the G2019S mutation in LRRK2 (Ramonet et al. 2011). Nevertheless, most transgenic α-synuclein mouse models develop gradual α-synuclein pathology, but fail to display clear dopaminergic cell loss and dopamine-dependent behavioral deficits. This hurdle was overcome by direct targeting of the SN with viral vectors overexpressing PD-associated genes.
4 Viral Vector-Based PD Models
Viral vector-mediated loco-regional gene delivery provides an alternative way to express transgenes in the CNS with several advantages: Local transgene delivery allows for specific targeting of brain regions; transgene expression can be induced during adulthood, bypassing the risk of compensatory mechanisms during development; different doses can be applied; models can be created in multiple species and strains, ranging from rodents to nonhuman primates and finally different combinations of genes can easily be made. Since the first proof of principle of this technique, a continuously growing number of publications have proven the value of viral vector-mediated loco-regional transduction. Before we discuss the different viral vector-based PD models currently available, we will give an overview of the different viral vector systems used for delivery of transgenes to the brain.
4.1 Viral Vectors for Gene Transfer
Viruses are efficient vehicles to infect cells to introduce genetic material and force the cell to replicate the viral genome in order to produce new virus particles. Viruses can be engineered to nonreplicating viral vectors that retain their ability of entering cells and introducing genes. By deleting parts of the viral genome and replacing them by the genes of interest, application of the vector will result in a single-round infection without replication in the host cell. The general design of viral vectors is based on the physical separation in different plasmids of the cis-elements involved in the transfer of the vector genome from trans-acting elements encoding for the viral (structural) proteins. These plasmids deliver the necessary components to assemble a vector particle during the vector production in producer cells. Viral vectors can be used for both overexpression and silencing of certain genes. The transgene encoded by the vector can be a reporter protein for visualization of the labeled cells by immunohistochemical or non-invasive imaging techniques such as bioluminescence, nuclear medicine techniques, or magnetic resonance imaging (Deroose et al. 2009). The expressed protein can be a therapeutic protein for gene therapy applications (Manfredsson et al. 2007; Vercammen et al. 2006; Winklhofer 2007) or as we will focus on in this chapter, a disease-related protein used for disease modeling (Kirik et al. 2002, 2003; Lauwers et al. 2003; Klein et al. 2002). Several vector systems based on various viruses have been developed. The choice of the vector system depends on the size of the gene of interest, the required duration of gene expression, the target cells and biosafety issues (see Table 1). For stable gene transfer in the brain, LV and rAAV vectors are the vector systems of choice since they lead to efficient and long-term gene expression in the rodent brain. The different vector systems will be described in more detail in the following section.
4.1.1 Lentiviral Vectors
LVs are derived from lentiviruses, a family of complex retroviruses that have the ability to replicate in non-dividing cells as opposed to simple retroviruses. Vectors derived from these viruses are an attractive means for gene transfer to the central nervous system (CNS) because they are capable of efficiently transducing non-dividing mature neurons. The prototypical lentivirus is the human immunodeficiency virus type 1 (HIV-1), which has a diameter of 110 nm and contains two copies of a 9200 bp ss RNA genome. The genome has three major coding regions in common with simple retroviruses (gag, pol, and env), flanked by two long terminal repeats (LTR). Gag encodes the structural core protein matrix, capsid, and nucleocapsid, pol encodes the viral enzymes [protease, reverse transcriptase (RT), and integrase] and env encodes the glycoprotein components of the viral envelope. In addition, complex retroviruses have six accessory genes (tat, rev, vpr, vpu, vif, and nef) that encode for additional regulatory proteins. These accessory genes are not essential for viral replication in cell culture, but determine the pathogenicity of viral infection in vivo. Several studies have shown that LVs derived from HIV-1 are indeed capable of efficient and stable expression of the transgene in vivo without induction of significant immune response (Baekelandt et al. 2002; Naldini et al. 1996a, b; Zufferey et al. 1997).
Design of Lentiviral Vectors
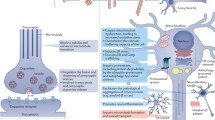
The first generation of LVs derived from HIV-1 was developed in 1996, by Naldini et al. (1996a, b). LVs are usually produced by triple transfection of HEK293T cells with three different plasmids, providing the cis- and trans-elements necessary for the production of vector particles (Fig. 1). The first plasmid, the packaging plasmid encodes structural viral proteins from gag and enzymes from pol under the control of a constitutive promoter. For biosafety reasons, the packaging signal (Ψ) and the env gene have been deleted. The envelope plasmid mostly codes for the envelope glycoprotein of the vesicular stomatitis virus (VSV-G). This VSV-G envelope results in a broad cellular tropism and efficiently targets neurons and astrocytes in the CNS and ensures a better stability of the vector particle compared to HIV enveloped vectors. The third plasmid, the transfer plasmid encodes the transgene of interest under the control of a heterologous promoter. It contains cis-acting sequences necessary for encapsidation, reverse transcription, and integration flanked by two LTRs. LTRs are necessary for integration of the proviral DNA mediated by the viral integrase and also contain a promoter for proviral DNA transcription. After triple transfection, the vector particles produced by the HEK293T cells are released into the medium and can be purified and concentrated.
After the initial validation of the LV system, additional elements were introduced to increase the specificity and enhance the efficiency of these vectors. A first improvement was the insertion of the central polypurine tract (cPPT), which is thought to facilitate reverse transcription and nuclear import of the lentiviral pre-integration complex prior to vector integration. Inclusion of this element in the LV increased the transduction efficiency and gene expression 3- to 10-fold (De Rijck et al. 2005; Zennou et al. 2000). A second improvement was the introduction of the Woodchuck hepatitis virus post-transcriptional regulatory element (WPRE) that enhanced expression levels about 5- to 8-fold in a number of different cell lines and tissues due to an increased mRNA stability in the cytoplasm (Zufferey et al. 1999). LV containing both the cPPT and the WPRE has higher in vivo transduction efficiency than the sum of the expression levels reached by LV containing only the cPPT or the WPRE (Baekelandt et al. 2002).
Biosafety of Lentiviral Vectors
Lentiviral vectors derived from HIV-1 may provoke biosafety concerns because of the well-known pathogenicity of the parental virus. An important issue is the potential occurrence of replication competent retroviruses (RCR), which can theoretically be generated during vector production by homologous recombination. A lot of effort has been undertaken to improve the biosafety of lentiviral vectors. In addition to the removal of accessory genes in the transfer plasmid, thereby reducing or eliminating the pathogenicity of HIV-1, safer LVs have been developed by splitting the cis- and trans-acting viral sequences over separate expression plasmids and deleting promoter and enhancer elements in the transfer vector itself. This reduces the probability of homologous recombination.
In the first generation of vectors, the env gene coding for the natural HIV envelope was replaced by the VSG-G gene. As a result, no WT HIV-1 could ever arise even if the three plasmids recombined. The encapsidation signal was removed from the packaging plasmid and the viral LTRs were replaced by a heterologous promoter at the 5′-end and a polyadenylation signal at the 3′-end. In the second-generation vectors, the virulence genes vpr, vpu, vif, and nef were deleted from the packaging construct without loss of transduction capacity (Zufferey et al. 1997). Since it is known that WT HIV-1 loses its pathogenicity when devoid of these proteins, deletion of these genes further increased biosafety. Vectors from the third generation do not require tat anymore since the U3 region of the 5′-LTR, which enhances viral transcription in the presence of tat was replaced by a constitutive promoter (Dull et al. 1998). This allowed tat gene deletion from the packaging plasmids. In addition, rev, an accessory protein mediating nuclear export of viral RNA was supplied from a fourth plasmid, further reducing the chance of recombination.
Another strategy to improve the biosafety of LVs involves deletion of the promoter and enhancer elements located in the 3′-LTR U3 region of the transfer plasmid. Vectors containing this deletion are called self-inactivating (SIN) vectors because the deletion is copied from the 3′-LTR to the 5′-LTR during reverse transcription, resulting in modified LTRs with only weak promoter activity (10 % of original activity). In addition, this deletion in the 3′-LTR prevents potential transcriptional activation of any (onco)gene upstream or downstream of the integration site. Moreover, SIN vectors cannot be rescued by WT HIV-1 (Bukovsky et al. 1999).
4.1.2 Recombinant Adeno-Associated Viral Vectors
Recombinant adeno-associated viral (rAAV) vectors derived from the adeno-associated virus (AAV) have been an attractive gene delivery vehicle since they were first engineered almost three decades ago (Samulski et al. 1982). The main reason for this is the unique combination of attractive properties: rAAV vectors ensure efficient gene transfer in different tissues. rAAV vectors are considered safe because the AAV virus they are derived from, is a non-pathogenic parvovirus whose replication depends on co-infection with a lytic helper virus, usually a member of the adenovirus (Atchison et al. 1965) or herpes virus family (Buller et al. 1981). rAAV vectors are also devoid of all WT AAV genes, making reversion to replication competent forms virtually impossible. In addition, compared to adenoviral vectors, the administration of rAAV vectors in vivo usually does not elicit a host immune response resulting in destruction of the transduced cells as shown in preclinical studies in small animal models .
rAAV vectors are capable of packaging gene cassettes of 4.5–5 kb (Baekelandt et al. 2000; Grieger and Samulski 2005). rAAV gene cassettes do not integrate into the host genome, but they readily persist for months to years in slowly dividing or nondividing cells. Indeed, studies have demonstrated that rAAV vectors can efficiently transduce a number of somatic tissues, including muscle (Xiao et al. 1996), liver (Miao et al. 1998), heart (Chu et al. 2004), retina (Hellstrom et al. 2009), and the CNS (Tenenbaum et al. 2004). To date, the most widely used rAAV vector is based on the AAV2 serotype, which has however been shown to contain some drawbacks. rAAV2 transduces neurons efficiently in the immediate vicinity of the injection site (Passini et al. 2004), but requires multiple injections, convection-enhanced delivery (Bankiewicz et al. 2000; Cunningham et al. 2000) or addition of agents such as mannitol, basic fibroblast growth factor (bFGF) or heparin to transduce larger brain volumes (Mastakov et al. 2001, 2002; Burger et al. 2005; Hadaczek et al. 2004). Furthermore, the clinical application of rAAV2/2 may be limited by pre-existing immunity to AAV2, which is present in most humans (Moskalenko et al. 2000). Finally, rAAV2 mainly targets neurons, although other cell types in the CNS might also be transduced. The limitations of rAAV2 have advanced the evaluation of alternative/artificial serotypes with broader cellular targets, higher transduction efficiencies and potential to evade pre-existing immunity to the AAV2 capsid (Paterna et al. 2004; Burger et al. 2004; Taymans et al. 2007; McFarland et al. 2009a; Dodiya et al. 2009).
A possible route to find the ideal rAAV tropism, next to modification of the rAAV capsid structure by chemical, immunological, or genetic means (Rabinowitz and Samulski 2000), is exploiting the natural capsid diversity of newly isolated serotypes by packaging rAAV2 genomes into capsids derived from other human or non-human AAV isolates (Grimm and Kay 2003). To this end, up until now, most researchers employ hybrid trans-complementing constructs that encode rep from AAV2, whereas cap is derived from the serotype displaying the cell tropism of choice. These hybrid vectors are therefore indicated as rAAV2/5, rAAV2/7, etc., where the second number refers to the capsid serotype. Among the more than 120 identified AAV variants that have been isolated from adenovirus stock or from human/non-human primate tissues, AAV2/1 to AAV2/10 are currently being developed as recombinant vectors for brain applications (Grimm and Kay 2003; Rutledge et al. 1998; Gao et al. 2002, 2003, 2004, 2005; Schmidt et al. 2006; Wu et al. 2006). This major advance in rAAV vectorology has significantly broadened potential applications of rAAV vectors for clinical gene therapy or disease modeling, and offers more options for the selection of a suitable rAAV variant per specific application. Recombinant AAV vectors are currently considered as a first choice option for brain applications both to generate preclinical models of neurodegeneration and for gene therapy. Indeed, rAAV vectors have proven useful to model diseases such as PD and have also been tested in various phases of clinical development of gene therapy for PD (Christine et al. 2009; Kaplitt et al. 2007) and Alzheimer’s disease (Mandel 2010)
Design of rAAV Vectors
Historically, most recombinant AAV vectors were based on serotype 2 (rAAV2/2) that constitutes the prototype of the genus, and was produced by means of two plasmids (the transfer and the packaging plasmid) and an infectious adenovirus. The transfer plasmid carries a transgene expression cassette flanked by the AAV2 inverted terminal repeats (ITRs), which are the only cis-acting elements required for replication and packaging of the recombinant genome (Samulski et al. 1987). The ends of the AAV2 genome consist of a 145 nucleotide-long ITR that, due to the multipalindromic nature of the terminal 125 bases, can fold on itself via complementary base pairing and form a characteristic T-shaped hairpin structure. The AAV2 nonstructural (rep) gene and a specific structural (cap) gene that depends on the serotype used are supplied in trans on the second plasmid, the so-called packaging plasmid. The adenoviral (Ad) helper functions were originally supplied by infection of rAAV producer cells with a WT adenovirus. The finding that Ad helper functions are provided by expression of E1A, E1B, E2A, E4ORF6, and VA RNAs, enabled subsequent Ad-free production of rAAV vector stocks by incorporating these sequences into a plasmid (referred to as adeno helper plasmid) and transfecting it together with the two above-mentioned plasmids. Upon introduction of all these constructs into the producer cells, vector particles are generated (Fig. 1).
Extensive efforts have been focused on developing versatile and scalable manufacturing processes for rAAV vector production with attention to compatibility with good manufacturing practice (GMP) (Gao et al. 2000; Clark et al. 1995; Blouin et al. 2004; Wright 2009). In our research group, we optimized a scalable and flexible serum-free rAAV vector production system, allowing a swift adaptation for production of different serotypes (Lock et al. 2010; Van der Perren et al. 2011, 2014; Toelen et al. 2014).
4.1.3 Adenoviral Vectors
Adenoviral vectors are derived from adenoviruses, DNA viruses with a linear double- stranded genome of 36 kb. The viral genome encodes for about 50 different proteins, 11 of which are structural and used to build the virion. These viruses have been isolated from a large number of species, and in humans, they primarily infect the respiratory airways and the gut causing mild respiratory and gastroenteric diseases. More than 40 AdV serotypes have been described, some more widespread than others (Davison et al. 2003). Because of their low pathogenicity and wide tropism, AdV-based vectors could be good candidates for gene delivery transfer. Unfortunately, the broad pre-existing immunity in the population prevents the use of vectors derived from the most common serotypes. Furthermore, the high immunogenicity of AdV proteins severely limits re-administration. To bypass these problems, AdV are mostly derived from rare serotypes, usually AdV2 or 5. AdV proved to be able to transduce a great variety of post-mitotic cells in the tissues like lung, skeletal muscle, heart, and brain (Howarth et al. 2010).
Design of Adenoviral Vectors
In first-generation AdV vectors, the early gene 1A (E1A), a regulatory gene essential for replication, was deleted (Graham and Prevec 1995). To further reduce the risk of the occurrence of a replication competent virus, E1B and E3, genes that play a role in modulating AdV-specific immunity, as well as E2 and E4 were additionally deleted (Campos and Barry 2007). This resulted in a vector system with a capacity to introduce up to 30 kb of DNA. To produce vector particles, these deleted genes essential for replication are provided by a helper virus or DNA construct that provides the missing functions in trans. Vector particles are generated by transfection of the vector construct and a helper virus, or by double transfection with a packaging construct. Alternatively, AdV vectors can also be produced using a packaging cell line, which stably expresses the structural and regulatory proteins required for vector assembly (Wang et al. 2009). Efforts are being made to circumvent the broad pre-existing immunity, which currently limits the use of this vector type.
4.1.4 Herpes Simplex Vectors
Herpes virus vectors are mainly derived from HSV type1, a very large doublestranded DNA virus of 152 kb. The viral genome encodes for more than 80 different proteins, which can be divided into essential and nonessential genes whether they are required for viral replication. HSV-1 is spread by contact, infects, and replicates in the skin or mucous membranes, and is taken up by sensory nerve terminals where it establishes a latent state, from which the virus can subsequently be reactivated and spread to other individuals. HSV-1 is endemic and more than 70 % of the people have a specific immune response that is maintained active due to intermittent reactivation of the infection. The main limitation in the use of HSV-1 vectors is the pre-existing immunity in humans, eliminating vector particles and transduced cells exposing HSV proteins on their surface. A second safety concern is the presence of latently infected cells, which may, upon transduction, offer a suitable environment for the HSV vector to recombine with the WT genome. These limitations have severely limited the range of applications of HSV vectors. On the other hand, these limitations can be considered as an advantage when using them as oncolytic vectors, targeting proliferating tumor cells.
Design of Herpes Simplex Vectors
The HSV-1 genome contains a significant portion of viral genes that are considered “non-essential,” which have been removed during vector construction. This elimination makes room for up to 50–150 kb of DNA depending on the type of HSV vector, making them the largest carriers among all viral vectors. The genetic complexibility of the virus genome allows the production of different types of vectors with different properties. Currently, three classes of vectors are derived from HSV-1: Conditionally replication competent vectors, replication incompetent vectors, and amplicon-based vectors (de Silva and Bowers 2009). The first class is designed by deleting the genes not essential for replication, but important for pathogenicity. These vectors are capable of replicating only in certain cell types and tissues in vivo, and are typically used in the development of therapies for malignant brain tumors (Markert et al. 2000). They are referred as oncolytic HSV-1 vectors. For the second class, the replication incompetent vectors, one or more immediate-early genes essential for lytic replication and reactivation have been deleted, but they retain the ability to establish latency. For the production of vector particles, this genetic information is provided in trans by a replication competent HSV strain, a packaging construct or by a specific packaging cell line that stable expresses the required proteins. These replication incompetent vectors have been used for preclinical studies of neurodegenerative diseases and chronic pain (Burton et al. 2002). To generate the third class, the amplicon-based vectors, a single origin of replication and a single packaging/cleavage signal from the WT HSV-1 was incorporated into a standard bacterial plasmid, called the amplicon. A transgene can be cloned into the amplicon plasmid and defective viral vector particles can be produced by complementing the necessary genes by a defective helper virus, a packaging construct, or specific packaging cells (de Silva and Bowers 2009). This class is the safest as it carries minimal viral sequences, but requires optimization to increase vector titers.
4.2 From Viral Vector to Animal Models
4.2.1 Viral Vector-Based α-Synuclein Overexpression Models
Overexpression of α-synuclein by direct targeting of the SN of rats, mice, or non-human primates with viral vectors offers a valuable alternative approach to the α-synuclein transgenic mice. Using viral vectors, high transgene expression levels can be achieved, which might be crucial since the disease onset and severity depends on the level of α-synuclein expression. Two vector systems have been explored for this purpose: rAAV and LV (Table 2).
The viral vector approach was initially explored almost simultaneously by a number of different groups using either rAAV2/2 or LV vectors (Kirik et al. 2002; Klein et al. 2002; Lauwers et al. 2003; Lo Bianco et al. 2002). In rats injection of rAAV2/2 vectors expressing either WT, A30P, or A53T mutant human α-synuclein unilaterally into the SN (Kirik et al. 2002; Klein et al. 2002) induced efficient expression of α-synuclein in nigral DN, accompanied by cellular and axonal pathologies and nigral DN loss that developed progressively over time. These first-generation rAAV2/2-based α-synuclein models displayed progressive neurodegeneration, but the loss of the nigral TH-positive neurons (25–80 %) as well as the time course described was quite variable (6 weeks up to 1 year) (Kirik et al. 2002; Klein et al. 2002). LV vectors encoding WT, A30P, or A53T mutant α-synuclein were also capable of inducing neuronal cell loss in rats, but less pronounced and more delayed (24–35 % cell loss at 5 months) (Lo Bianco et al. 2002). Dystrophic neurites and swollen perikarya were detected in the remaining DNs and cytoplasmic accumulations of α-synuclein were found in cell bodies as well as in neurites. Many attempts were made to reproduce and improve these rat models using rAAV2/2 (Yamada et al. 2004; Maingay et al. 2006; Mochizuki et al. 2006; Chung et al. 2009) or LV (Lauwers et al. 2007), but similar results were obtained.
Both LV and rAAV vectors have also been used in mice. Work from our own group showed that injection of LV-WT and A30P α-synuclein in different mouse brain regions (striatum, amygdala, SN) induced time-dependent neuropathological changes including neuritic enlargements and cytoplasmic inclusions. Furthermore, nigral overexpression of A30P α-synuclein resulted in a 10–25 % cell loss at 10–12 months (Lauwers et al. 2003). In another study, nigral injection of rAAV2/2 WT α-synuclein induced a 25 % cell loss at 24 weeks; however, no obvious cytoplasmic inclusions were detected in the α-synuclein expressing cells (St Martin et al. 2007). Overall, the pathology observed in mice, both the onset and the severity, appeared less severe compared to the described rat models. rAAV vectors were also applied in non-human primates. Sixteen weeks after nigral injection of rAAV2/2 WT or A53T α-synuclein, 30–60 % of the nigral cells were lost, which resulted in mild motor deficits (Kirik et al. 2003).
The limitations of rAAV2/2 have prompted the evaluation of alternative serotypes with broader cellular targets and higher transduction efficiencies. As a result, other serotypes, notably rAAV2/5, rAAV2/6, rAAV2/8, and rAAV1/2, have been used in rats and non-human primates to overexpress α-synuclein (Eslamboli et al. 2007; Gorbatyuk et al. 2008; Azeredo da Silveira et al. 2009; Sanchez-Guajardo et al. 2010; Koprich et al. 2011; McFarland et al. 2009a). Since phosphorylation of α-synuclein at serine 129 is a common feature in patients suffering of synucleinopathies, mutations mimicking α-synuclein phosphorylation might also enhance dopaminergic pathology. Mutations mimicking (S129D) or preventing phosphorylation (S129A) have been expressed using rAAV2/5, rAAV2/6, and rAAV2/8 in the rat SN (Gorbatyuk et al. 2008; Azeredo da Silveira et al. 2009; McFarland et al. 2009b). Interestingly, in the studies using rAAV2/5 and rAAV2/6, the S129A mutation, which can no longer be phosphorylated, showed enhanced toxicity (70 % cell loss at 4 weeks) compared to WT (60 % cell loss at 26 weeks), while the S129D mutant showed reduced or no toxicity (Gorbatyuk et al. 2008; Azeredo da Silveira et al. 2009). With rAAV2/8, the α-synuclein mediated toxicity was equivalent for WT as well as both S129 mutants (26 % at 6 weeks) (McFarland et al. 2009b), an observation potentially explained by different expression levels obtained by rAAV2/8 compared to rAAV2/5 or rAAV2/6 in the substantia nigra (Van der Perren et al. 2011). rAAV1/2 (chimeric vector) driven overexpression of A53T α-synuclein in the SN of rats caused a dopaminergic cell loss of 28 % at 6 weeks (Koprich et al. 2011). In this study, the choice of the vector titer was crucial since too high vector titers caused unspecific toxic effects. These second-generation rAAV-based α-synuclein rat models display a more reproducible neurodegeneration (15–60 % loss 6–26 weeks p.i.) (Gorbatyuk et al. 2008; Azeredo da Silveira et al. 2009; Sanchez-Guajardo et al. 2010; Koprich et al. 2011). However, even in these rat models, the time course and loss of the nigral TH-positive neurons still suffered from variability, which hindered clear motor deficits.
Different species of α-synuclein could also influence the levels of pathology. Winner et al. evaluated two artificial mutants of α-synuclein (E57K, E35K) with the propensity to form oligomeric aggregates and one faster fibril forming mutant of α-synuclein (α-SYN 30-110) in vivo (Winner et al. 2011). E57K and E35K mutant α-synuclein were delivered in the rat SN using LV and resulted in a dopaminergic loss of, respectively, 51 % and 50 %, which turned out to be more toxic than WT (32 %), A30P (38 %), E46K (40 %), and A53T (17 %) α-synuclein at 3 weeks p.i. Remarkably, the faster fibril forming mutant A53T did not show a significant decrease in dopaminergic cell number. These data further support the idea that the oligomeric species and not the fibrillar form of synuclein are toxic and contribute to dopaminergic degeneration. Although being very instrumental, the dopaminergic cell loss of 17–50 % in these LV-based rat models is insufficient for behavioral manifestations, which require at least 60–70 % cell loss. In non-human primates, better results were obtained. Overexpression of WT or A53T mutant α-synuclein using rAAV2/5 resulted in a clear difference at the behavioral and morphological level (Eslamboli et al. 2007). Stronger motor impairments were observed in the A53T group compared to the WT group 42 weeks after injection. After 1 year, TH-positive fiber loss was observed in the striatum of both groups, but it was more pronounced in the A53T group. In both groups, the remaining fibers were dystrophic and contained α-synuclein positive insoluble inclusions. Many α-synuclein positive aggregates were found to be phosphorylated at serine 129. When analyzing the SN, a loss of up to 40 % of the DNs was observed in the A53T α-synuclein animals in contrast to WT α-synuclein animals, which only differed in two out of eight animals from the eGFP control animals. Furthermore, at 1 year p.i. ubiquitin positive inclusions were detected in the surviving nigral neurons of both groups.
To summarize, viral vector-mediated α-synuclein models display synuclein pathology and clear dopaminergic neurodegeneration in contrast to α-synuclein transgenic mice. The transgene expression in DN achieved with the novel rAAV serotypes (rAAV2/5, 2/6, 2/8, and 2/1) is improved compared to rAAV2/2 and substantially higher compared to LV probably due to their high titers and tropism for DN neurons, resulting in higher levels of neurodegeneration. Although promising, these models still suffer from a certain degree of variability, a slow progression of the phenotype and lack overt behavioral impairments, hindering their usefulness for testing novel therapeutics.
To address these issues, a third generation of α-synuclein models has been developed by optimizing the promoter, the rAAV serotype, the vector construct, as well as the vector titer and purity of the vector preparation. Decressac et al. reported progressive dopaminergic neurodegeneration up to 75 % at 8–16 weeks after nigral injection of rAAV2/6 using the neuron specific synapsin-1 promoter to drive the expression of WT α-synuclein (Decressac et al. 2012). The cell loss was preceded by degenerative changes of striatal axons and terminals, and the presence of α-synuclein positive inclusions in dystrophic axons and dendrites. This high level of neurodegeneration resulted in clear behavioral deficits seen in the cylinder test and the amphetamine-induced rotation test. Our group has recently shown that rAAV2/7 outperformed most serotypes in terms of transduction efficiency and expression levels in rat SN (Van der Perren et al. 2011). Based on these data, we developed a rat model for PD by injection of rAAV2/7 encoding A53T α-synuclein into the SN (Fig. 2) (Van der Perren et al. 2014). High α-synuclein expression was observed four days post-injection. At least 90 % of the DNs were transduced, which resulted in progressive nigrostriatal pathology up to 80 % and behavioral deficits in 4 weeks’ time period. Levodopa (l-dopa) was found to reverse the behavioral phenotype. Non-invasive PET and MR imaging allowed longitudinal monitoring of neurodegeneration. In addition, ubiquitin positive α-synuclein aggregates were detected. This rat PD model successfully recapitulates the progressive degeneration of DN and associated motor deficits together with the formation of α-synuclein inclusions. In parallel to this study, we also showed that strong and significant α-synuclein-induced neuropathology and progressive dopaminergic neurodegeneration can be achieved in mouse brain by means of rAAV2/7 (Oliveras-Salva et al. 2013). We noted a significant and dose-dependent α-synucleinopathy over time upon nigral viral vector-mediated α-synuclein overexpression. We obtained a strong, progressive, and dose-dependent loss of DNs in the SN, reaching a maximum of 82 % after 8 weeks. When comparing wild-type to A53T mutant α-synuclein at the same vector dose, both induced a similar degree of dopaminergic cell death (Oliveras-Salva et al. 2013). These major improvements can probably be explained by the choice of the rAAV7 serotype, the use of the CMV enhanced synapsin-1 promoter, and the vector titer combined with an optimized vector preparation. We believe that these improved rodent models will be of great value for further development and testing of neuroprotective strategies.
In vivo validation of an rAAV vector encoding α-synuclein using behavioral, PET, MRI, and IHC analysis. (a) Stereotactic injection of rAAV vector encoding α-synuclein (α-SYN) in the rat SN. (b) Cylinder test, PET imaging (dopamine transporter), and MR imaging. (c) Immunohistochemical staining for tyrosine hydroxylase (TH) in the substantia nigra (SN) and the striatum (STR) 4 and 29 days after vector injection in the SN, and for α-synuclein 29 days after injection
4.2.2 Viral Vector-Based LRRK2 Overexpression Models
rAAV are not suitable for the delivery of the 7581 bp LRRK2 cDNA to the nigrostriatal system due to their limited packaging capacity. Even for LV, the size of the LRRK2 cDNA is approaching the packaging limitations. Our group has succeeded to generate functional LRRK2 LV (Civiero et al. 2012), however, with reduced titers, which compromises the chances to induce phenotypic effects in vivo. To date, two groups have reported the generation of viral vector-mediated LRRK2 PD models using either recombinant adenoviral (rAdv) or herpes simplex vectors (HSV) (Table 3) (Dusonchet et al. 2011; Lee et al. 2010). rAd vectors expressing WT or G2019S mutant LRRK2 were injected into six sites within the rat striatum and nigral transduction relied on retrograde transport of the vector. Quantification of the expression levels revealed that 31 % of the nigral DN were transduced. Injection of Adv encoding mutant G2019S LRRK2 resulted in progressive loss of nigral DN up to 10 % after 10 days and 21 % after 42 days. WT LRRK2 rAdv did not induce any degeneration. No abnormal α-synuclein accumulation, ubiquitinated, or phosphorylated aggregates were found in WT or G2019S LRRK2 expressing neurons in the SN (Dusonchet et al. 2011). Lee et al. generated an LRRK2 mouse model using HSV vectors (Lee et al. 2010). HSV vectors encoding WT LRRK2, G2019S, or G2019S/D1994A LRRK2 were injected into the mouse striatum resulting in a retrograde transduction of 75 % of the nigral neurons. Three weeks after injection, overexpression of G2019S LRRK2 induced a progressive loss of 50 % of the nigral DN. G2019S-D1994A (kinase dead) LRRK2 caused no neuronal loss, similar to WT LRRK2 and GFP control vectors. These data link LRRK2 toxicity to elevated kinase activity in vivo.
4.2.3 Viral Vector-Based Parkin, DJ-1, and PINK1 Knockdown Models
Since parkin, PINK1, and DJ-1 KO mice do not display a clear phenotype, viral vector-mediated gene silencing in the adult brain could be explored as an alternative strategy. Stable knockdown of gene expression can be achieved by viral vector-mediated RNA interference after stereotactic injection into the brain (Ulusoy et al. 2009). Adult knockdown with viral vector technology has not been published for parkin or DJ-1. Haque et al. achieved a shRNA-mediated knockdown of PINK1 of 71 % in the striatum and 68 % in the SN by stereotactic striatal injection using an adenoviral vector in adult rats (Haque et al. 2012). This knockdown of PINK1 in the SN increased the sensitivity to MPTP administration, resulting in an increased loss of DN in the SN and terminal dopaminergic fibers in the striatum. Knockdown of PINK1 by itself, however, did not affect the number of DN in this 3-week time frame (Table 3) (Haque et al. 2012). Recent observations from our own group have confirmed that knockdown of PINK1 (Oliveras-Salvá et al. 2014) or even parkin using rAAV2/7 in adult mouse or rat SN up to 10 months does not elicit significant dopaminergic cell death (unpublished data).
However, an elegant alternative approach was recently pursued by adult knockout of parkin after injection of an LV expressing GFP-Cre into the SN of 6- to 8-week-old conditional parkin knockout mice (exon 7 flanked by loxP sites, parkinFLX/FLX). Injection of LV GFP-Cre led to a near complete loss of parkin in the ventral midbrain of parkinFlx/Flx animals, resulting in a significant reduction in nigral DN up to 45 % after 10 months (Shin et al. 2011).
5 Conclusions and Perspectives
Much effort has been spent over the last 10 years on the development and improvement of viral vector-based PD models. So far, models based on overexpression of α-synuclein are the most prevalent and extensively studied. The first-generation α-synuclein models have learned that the levels of α-synuclein expression crucially determine the disease onset and severity, and are important to elicit reliable motor impairments. Since then, efforts have been focused on increasing expression levels of α-synuclein in the DN, which has resulted in second- and third-generation models. rAAV vectors have gradually outcompeted LV vectors because of their higher titers and transduction efficiency of DN. The best models currently available have been developed from a combined approach using newer AAV serotypes (rAAV1, 5, 6, 7, 8) and optimized vector constructs, titer, and purity. These third-generation α-synuclein models show improved face and predictive validity and therefore offer the possibility to reliably test novel therapeutics.
Besides being useful for preclinical drug testing, the viral vector-based α-synuclein models have allowed to examine important aspects of α-synuclein pathophysiology, such as α-synuclein phosphorylation, toxicity of α-synuclein oligomers versus fibrils, etc. Furthermore, differences in sensitivity of DN among animal species have been detected. Compared to rats, mice DN seem to be less sensitive to α-synuclein overexpression, resulting in a delayed manifestation of neurodegeneration. This is in line with observations in α-synuclein transgenic mice where dopaminergic degeneration is largely absent. However, a comparison with α-synuclein transgenic rats would be required to rule out whether this is genuinely due to different protective mechanisms between mice and rats rather than to differences in levels or age-of-onset of the α-synuclein overexpression.
Comparing models created across different laboratories remains difficult since different vector systems, serotypes, and production methods are being used. The vector titer as well as the vector purity directly influences the phenotypic outcome of the model. Excessive vector titers or insufficiently purified vector batches may result in a specific toxicity. Therefore, appropriate control vectors are indispensable. Considerable time investment in the viral vector production, upscaling, and purification procedures has also proven essential to obtain reproducible and high quality vector batches.
Rodent models based on overexpression of G2019S LRRK2 display mild (10 %) to severe (50 %) neurodegeneration depending on the vector system (AdV5 or HSV) used. A disadvantage of the AdV5-based rat model is the low percentage of retrogradely transduced nigral neurons (31 %) compared to the 75 % obtained in mice using HSV. Importantly, both models have provided in vivo evidence for the link between G2019S LRRK2 toxicity and elevated kinase activity, which reinforces the concept of LRRK2 kinase inhibition as a possible therapeutic strategy.
RNAi-mediated viral vector-based knockdown offers in principle an interesting tool to study recessive genes like parkin, DJ-1, and PINK1. However, until now, only one study related to PINK1 has been published applying this approach. This might partially be explained by difficulties to detect and quantify knockdown levels in vivo due to the lack of reliable antibodies or other detection methods of endogenous protein. Nevertheless, the knockdown approach with viral vectors still represents an attractive and flexible strategy to combine with overexpression/knockdown of other PD-related genes or also in combination with transgenic animals in order to determine whether these proteins act in similar pathways in vivo.
References
Andres-Mateos E, Perier C, Zhang L, Blanchard-Fillion B, Greco TM, Thomas B, Ko HS, Sasaki M, Ischiropoulos H, Przedborski S, Dawson TM, Dawson VL (2007) DJ-1 gene deletion reveals that DJ-1 is an atypical peroxiredoxin-like peroxidase. Proc Natl Acad Sci U S A 104(37):14807–14812
Atchison RW, Casto BC, Hammon WM (1965) Adenovirus-associated defective virus particles. Science 149:754–756
Azeredo da Silveira S, Schneider BL, Cifuentes-Diaz C, Sage D, Abbas-Terki T, Iwatsubo T, Unser M, Aebischer P (2009) Phosphorylation does not prompt, nor prevent, the formation of alpha-synuclein toxic species in a rat model of Parkinson’s disease. Hum Mol Genet 18(5):872–887
Baekelandt V, De Strooper B, Nuttin B, Debyser Z (2000) Gene therapeutic strategies for neurodegenerative diseases. Curr Opin Mol Ther 2(5):540–554
Baekelandt V, Claeys A, Eggermont K, Lauwers E, De Strooper B, Nuttin B, Debyser Z (2002) Characterization of lentiviral vector-mediated gene transfer in adult mouse brain. Hum Gene Ther 13(7):841–853
Bankiewicz KS, Eberling JL, Kohutnicka M, Jagust W, Pivirotto P, Bringas J, Cunningham J, Budinger TF, Harvey-White J (2000) Convection-enhanced delivery of AAV vector in parkinsonian monkeys; in vivo detection of gene expression and restoration of dopaminergic function using pro-drug approach. Exp Neurol 164(1):2–14
Belin AC, Westerlund M (2008) Parkinson’s disease: a genetic perspective. FEBS J 275(7):1377–1383
Betarbet R, Sherer TB, MacKenzie G, Garcia-Osuna M, Panov AV, Greenamyre JT (2000) Chronic systemic pesticide exposure reproduces features of Parkinson’s disease. Nat Neurosci 3(12):1301–1306
Betarbet R, Sherer TB, Greenamyre JT (2002) Animal models of Parkinson’s disease. BioEssays 24(4):308–318
Bezard E, Przedborski S (2011) A tale on animal models of Parkinson’s disease. Mov Disord 26(6):993–1002
Bjorklund LM, Sanchez-Pernaute R, Chung S, Andersson T, Chen IY, McNaught KS, Brownell AL, Jenkins BG, Wahlestedt C, Kim KS, Isacson O (2002) Embryonic stem cells develop into functional dopaminergic neurons after transplantation in a Parkinson rat model. Proc Natl Acad Sci U S A 99(4):2344–2349
Blouin V, Brument N, Toublanc E, Raimbaud I, Moullier P, Salvetti A (2004) Improving rAAV production and purification: towards the definition of a scaleable process. J Gene Med 6(Suppl 1):S223–S228
Braak H, Del Tredici K, Rub U, de Vos RA, Jansen Steur EN, Braak E (2003) Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 24(2):197–211
Bukovsky AA, Song JP, Naldini L (1999) Interaction of human immunodeficiency virus-derived vectors with wild-type virus in transduced cells. J Virol 73(8):7087–7092
Buller RM, Janik JE, Sebring ED, Rose JA (1981) Herpes simplex virus types 1 and 2 completely help adenovirus-associated virus replication. J Virol 40(1):241–247
Burger C, Gorbatyuk OS, Velardo MJ, Peden CS, Williams P, Zolotukhin S, Reier PJ, Mandel RJ, Muzyczka N (2004) Recombinant AAV viral vectors pseudotyped with viral capsids from serotypes 1, 2, and 5 display differential efficiency and cell tropism after delivery to different regions of the central nervous system. Mol Ther 10(2):302–317
Burger C, Nguyen FN, Deng J, Mandel RJ (2005) Systemic mannitol-induced hyperosmolality amplifies rAAV2-mediated striatal transduction to a greater extent than local co-infusion. Mol Ther 11(2):327–331
Burton EA, Fink DJ, Glorioso JC (2002) Gene delivery using herpes simplex virus vectors. DNA Cell Biol 21(12):915–936
Campos SK, Barry MA (2007) Current advances and future challenges in adenoviral vector biology and targeting. Curr Gene Ther 7 (3):189-204
Chartier-Harlin MC, Kachergus J, Roumier C, Mouroux V, Douay X, Lincoln S, Levecque C, Larvor L, Andrieux J, Hulihan M, Waucquier N, Defebvre L, Amouyel P, Farrer M, Destee A (2004) Alpha-synuclein locus duplication as a cause of familial Parkinson’s disease. Lancet 364(9440):1167–1169
Chen L, Thiruchelvam MJ, Madura K, Richfield EK (2006) Proteasome dysfunction in aged human alpha-synuclein transgenic mice. Neurobiol Dis 23(1):120–126
Chesselet MF, Richter F (2011) Modelling of Parkinson’s disease in mice. Lancet Neurol 10(12):1108–1118
Christine CW, Starr PA, Larson PS, Eberling JL, Jagust WJ, Hawkins RA, VanBrocklin HF, Wright JF, Bankiewicz KS, Aminoff MJ (2009) Safety and tolerability of putaminal AADC gene therapy for Parkinson disease. Neurology 73(20):1662–1669
Chu D, Thistlethwaite PA, Sullivan CC, Grifman MS, Weitzman MD (2004) Gene delivery to the mammalian heart using AAV vectors. Methods Mol Biol 246:213–224
Chung CY, Koprich JB, Siddiqi H, Isacson O (2009) Dynamic changes in presynaptic and axonal transport proteins combined with striatal neuroinflammation precede dopaminergic neuronal loss in a rat model of AAV alpha-synucleinopathy. J Neurosci 29(11):3365–3373
Civiero L, Vancraenenbroeck R, Belluzzi E, Beilina A, Lobbestael E, Reyniers L, Gao F, Micetic I, De Maeyer M, Bubacco L, Baekelandt V, Cookson MR, Greggio E, Taymans JM (2012) Biochemical characterization of highly purified leucine-rich repeat kinases 1 and 2 demonstrates formation of homodimers. PLoS One 7(8)
Clark KR, Voulgaropoulou F, Fraley DM, Johnson PR (1995) Cell lines for the production of recombinant adeno-associated virus. Hum Gene Ther 6(10):1329–1341
Cunningham J, Oiwa Y, Nagy D, Podsakoff G, Colosi P, Bankiewicz KS (2000) Distribution of AAV-TK following intracranial convection-enhanced delivery into rats. Cell Transplant 9(5):585–594
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39(6):889–909
Davison AJ, Benko M, Harrach B (2003) Genetic content and evolution of adenoviruses. J Gen Virol 84(Pt 11):2895–2908
De Rijck J, Van Maele B, Debyser Z (2005) Positional effects of the central DNA flap in HIV-1-derived lentiviral vectors. Biochem Biophys Res Commun 328(4):987–994
de Silva S, Bowers WJ (2009) Herpes virus amplicon vectors. Viruses 1(3):594–629
Decressac M, Mattsson B, Lundblad M, Weikop P, Bjorklund A (2012) Progressive neurodegenerative and behavioural changes induced by AAV-mediated overexpression of alpha-synuclein in midbrain dopamine neurons. Neurobiol Dis 45(3):939–953
Deroose CM, Reumers V, Debyser Z, Baekelandt V (2009) Seeing genes at work in the living brain with non-invasive molecular imaging. Curr Gene Ther 9(3):212–238
Dodiya HB, Bjorklund T, Stansell Iii J, Mandel RJ, Kirik D, Kordower JH (2009) Differential transduction following basal ganglia administration of distinct pseudotyped AAV capsid serotypes in nonhuman primates. Mol Ther 18(3):579–587
Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D, Naldini L (1998) A third-generation lentivirus vector with a conditional packaging system. J Virol 72(11):8463–8471
Dusonchet J, Kochubey O, Stafa K, Young SM Jr, Zufferey R, Moore DJ, Schneider BL, Aebischer P (2011) A rat model of progressive nigral neurodegeneration induced by the Parkinson’s disease-associated G2019S mutation in LRRK2. J Neurosci 31(3):907–912
Edwards TL, Scott WK, Almonte C, Burt A, Powell EH, Beecham GW, Wang L, Zuchner S, Konidari I, Wang G, Singer C, Nahab F, Scott B, Stajich JM, Pericak-Vance M, Haines J, Vance JM, Martin ER (2010) Genome-wide association study confirms SNPs in SNCA and the MAPT region as common risk factors for Parkinson disease. Ann Hum Genet 74(2):97–109
Eslamboli A, Romero-Ramos M, Burger C, Bjorklund T, Muzyczka N, Mandel RJ, Baker H, Ridley RM, Kirik D (2007) Long-term consequences of human alpha-synuclein overexpression in the primate ventral midbrain. Brain 130(Pt 3):799–815
Farrer MJ (2006) Genetics of Parkinson disease: paradigm shifts and future prospects. Nat Rev Genet 7(4):306–318
Fleming SM, Fernagut PO, Chesselet MF (2005) Genetic mouse models of parkinsonism: strengths and limitations. NeuroRx 2(3):495–503
Fox SH, Brotchie JM (2010) The MPTP-lesioned non-human primate models of Parkinson’s disease. Past, present, and future. Prog Brain Res 184:133–157
Freichel C, Neumann M, Ballard T, Muller V, Woolley M, Ozmen L, Borroni E, Kretzschmar HA, Haass C, Spooren W, Kahle PJ (2007) Age-dependent cognitive decline and amygdala pathology in alpha-synuclein transgenic mice. Neurobiol Aging 28(9):1421–1435
Gao G, Qu G, Burnham MS, Huang J, Chirmule N, Joshi B, Yu QC, Marsh JA, Conceicao CM, Wilson JM (2000) Purification of recombinant adeno-associated virus vectors by column chromatography and its performance in vivo. Hum Gene Ther 11(15):2079–2091
Gao GP, Alvira MR, Wang L, Calcedo R, Johnston J, Wilson JM (2002) Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc Natl Acad Sci U S A 99(18):11854–11859
Gao G, Alvira MR, Somanathan S, Lu Y, Vandenberghe LH, Rux JJ, Calcedo R, Sanmiguel J, Abbas Z, Wilson JM (2003) Adeno-associated viruses undergo substantial evolution in primates during natural infections. Proc Natl Acad Sci U S A 100(10):6081–6086
Gao G, Vandenberghe LH, Alvira MR, Lu Y, Calcedo R, Zhou X, Wilson JM (2004) Clades of Adeno-associated viruses are widely disseminated in human tissues. J Virol 78(12):6381–6388
Gao G, Vandenberghe LH, Wilson JM (2005) New recombinant serotypes of AAV vectors. Curr Gene Ther 5(3):285–297
Gautier CA, Kitada T, Shen J (2008) Loss of PINK1 causes mitochondrial functional defects and increased sensitivity to oxidative stress. Proc Natl Acad Sci U S A 105(32):11364–11369
Goldberg MS, Fleming SM, Palacino JJ, Cepeda C, Lam HA, Bhatnagar A, Meloni EG, Wu N, Ackerson LC, Klapstein GJ, Gajendiran M, Roth BL, Chesselet MF, Maidment NT, Levine MS, Shen J (2003) Parkin-deficient mice exhibit nigrostriatal deficits but not loss of dopaminergic neurons. J Biol Chem 278(44):43628–43635
Goldberg MS, Pisani A, Haburcak M, Vortherms TA, Kitada T, Costa C, Tong Y, Martella G, Tscherter A, Martins A, Bernardi G, Roth BL, Pothos EN, Calabresi P, Shen J (2005) Nigrostriatal dopaminergic deficits and hypokinesia caused by inactivation of the familial Parkinsonism-linked gene DJ-1. Neuron 45(4):489–496
Gorbatyuk OS, Li S, Sullivan LF, Chen W, Kondrikova G, Manfredsson FP, Mandel RJ, Muzyczka N (2008) The phosphorylation state of Ser-129 in human alpha-synuclein determines neurodegeneration in a rat model of Parkinson disease. Proc Natl Acad Sci U S A 105(2):763–768
Graham FL, Prevec L (1995) Methods for construction of adenovirus vectors. Mol Biotechnol 3 (3):207–220
Grieger JC, Samulski RJ (2005) Packaging capacity of adeno-associated virus serotypes: impact of larger genomes on infectivity and postentry steps. J Virol 79(15):9933–9944
Grimm D, Kay MA (2003) From virus evolution to vector revolution: use of naturally occurring serotypes of adeno-associated virus (AAV) as novel vectors for human gene therapy. Curr Gene Ther 3(4):281–304
Hadaczek P, Mirek H, Bringas J, Cunningham J, Bankiewicz K (2004) Basic fibroblast growth factor enhances transduction, distribution, and axonal transport of adeno-associated virus type 2 vector in rat brain. Hum Gene Ther 15(5):469–479
Hamza TH, Zabetian CP, Tenesa A, Laederach A, Montimurro J, Yearout D, Kay DM, Doheny KF, Paschall J, Pugh E, Kusel VI, Collura R, Roberts J, Griffith A, Samii A, Scott WK, Nutt J, Factor SA, Payami H (2010) Common genetic variation in the HLA region is associated with late-onset sporadic Parkinson’s disease. Nat Genet 42(9):781–785
Haque ME, Mount MP, Safarpour F, Abdel-Messih E, Callaghan S, Mazerolle C, Kitada T, Slack RS, Wallace V, Shen J, Anisman H, Park DS (2012) Inactivation of Pink1 gene in vivo sensitizes dopamine-producing neurons to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and can be rescued by autosomal recessive Parkinson disease genes, Parkin or DJ-1. J Biol Chem 287(27):23162–23170
Hawkes CH, Del Tredici K, Braak H (2010) A timeline for Parkinson’s disease. Parkinsonism Relat Disord 16(2):79–84
Hellstrom M, Ruitenberg MJ, Pollett MA, Ehlert EM, Twisk J, Verhaagen J, Harvey AR (2009) Cellular tropism and transduction properties of seven adeno-associated viral vector serotypes in adult retina after intravitreal injection. Gene Ther 16(4):521–532
Hoglinger GU, Oertel WH, Hirsch EC (2006) The rotenone model of parkinsonism—the five years inspection. J Neural Transm Suppl 70:269–272
Howarth JL, Lee YB, Uney JB (2010) Using viral vectors as gene transfer tools (Cell Biology and Toxicology Special Issue: ETCS-UK 1 day meeting on genetic manipulation of cells). Cell Biol Toxicol 26(1):1–20
Ibanez P, Bonnet AM, Debarges B, Lohmann E, Tison F, Pollak P, Agid Y, Durr A, Brice A (2004) Causal relation between alpha-synuclein gene duplication and familial Parkinson’s disease. Lancet 364(9440):1169–1171
Ilijic E, Guzman JN, Surmeier DJ (2011) The L-type channel antagonist isradipine is neuroprotective in a mouse model of Parkinson’s disease. Neurobiol Dis 43(2):364–371
Itier JM, Ibanez P, Mena MA, Abbas N, Cohen-Salmon C, Bohme GA, Laville M, Pratt J, Corti O, Pradier L, Ret G, Joubert C, Periquet M, Araujo F, Negroni J, Casarejos MJ, Canals S, Solano R, Serrano A, Gallego E, Sanchez M, Denefle P, Benavides J, Tremp G, Rooney TA, Brice A, Garcia de Yebenes J (2003) Parkin gene inactivation alters behaviour and dopamine neurotransmission in the mouse. Hum Mol Genet 12(18):2277–2291
Jiang H, Jackson-Lewis V, Muthane U, Dollison A, Ferreira M, Espinosa A, Parsons B, Przedborski S (1993) Adenosine receptor antagonists potentiate dopamine receptor agonist-induced rotational behavior in 6-hydroxydopamine-lesioned rats. Brain Res 613(2):347–351
Kahle PJ, Neumann M, Ozmen L, Muller V, Odoy S, Okamoto N, Jacobsen H, Iwatsubo T, Trojanowski JQ, Takahashi H, Wakabayashi K, Bogdanovic N, Riederer P, Kretzschmar HA, Haass C (2001) Selective insolubility of alpha-synuclein in human Lewy body diseases is recapitulated in a transgenic mouse model. Am J Pathol 159(6):2215–2225
Kaplitt MG, Feigin A, Tang C, Fitzsimons HL, Mattis P, Lawlor PA, Bland RJ, Young D, Strybing K, Eidelberg D, During MJ (2007) Safety and tolerability of gene therapy with an adeno-associated virus (AAV) borne GAD gene for Parkinson’s disease: an open label, phase I trial. Lancet 369(9579):2097–2105
Kim RH, Smith PD, Aleyasin H, Hayley S, Mount MP, Pownall S, Wakeham A, You-Ten AJ, Kalia SK, Horne P, Westaway D, Lozano AM, Anisman H, Park DS, Mak TW (2005) Hypersensitivity of DJ-1-deficient mice to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyrindine (MPTP) and oxidative stress. Proc Natl Acad Sci U S A 102(14):5215–5220
Kirik D, Georgievska B, Burger C, Winkler C, Muzyczka N, Mandel RJ, Bjorklund A (2002a) Reversal of motor impairments in parkinsonian rats by continuous intrastriatal delivery of L-dopa using rAAV-mediated gene transfer. Proc Natl Acad Sci U S A 99(7):4708–4713
Kirik D, Rosenblad C, Burger C, Lundberg C, Johansen TE, Muzyczka N, Mandel RJ, Bjorklund A (2002b) Parkinson-like neurodegeneration induced by targeted overexpression of alpha-synuclein in the nigrostriatal system. J Neurosci 22(7):2780–2791
Kirik D, Annett LE, Burger C, Muzyczka N, Mandel RJ, Bjorklund A (2003) Nigrostriatal alpha-synucleinopathy induced by viral vector-mediated overexpression of human alpha-synuclein: a new primate model of Parkinson’s disease. Proc Natl Acad Sci U S A 100(5):2884–2889
Kitada T, Pisani A, Porter DR, Yamaguchi H, Tscherter A, Martella G, Bonsi P, Zhang C, Pothos EN, Shen J (2007) Impaired dopamine release and synaptic plasticity in the striatum of PINK1-deficient mice. Proc Natl Acad Sci U S A 104(27):11441–11446
Klein RL, King MA, Hamby ME, Meyer EM (2002) Dopaminergic cell loss induced by human A30P alpha-synuclein gene transfer to the rat substantia nigra. Hum Gene Ther 13(5):605–612
Koprich JB, Johnston TH, Huot P, Reyes MG, Espinosa M, Brotchie JM (2011) Progressive neurodegeneration or endogenous compensation in an animal model of Parkinson’s disease produced by decreasing doses of alpha-synuclein. PLoS ONE 6(3):e17698
Kruger R, Kuhn W, Muller T, Woitalla D, Graeber M, Kosel S, Przuntek H, Epplen JT, Schols L, Riess O (1998) Ala30Pro mutation in the gene encoding alpha-synuclein in Parkinson’s disease. Nat Genet 18(2):106–108
Lauwers E, Debyser Z, Van Dorpe J, De Strooper B, Nuttin B, Baekelandt V (2003) Neuropathology and neurodegeneration in rodent brain induced by lentiviral vector-mediated overexpression of alpha-synuclein. Brain Pathol 13(3):364–372
Lauwers E, Beque D, Van Laere K, Nuyts J, Bormans G, Mortelmans L, Casteels C, Vercammen L, Bockstael O, Nuttin B, Debyser Z, Baekelandt V (2007) Non-invasive imaging of neuropathology in a rat model of alpha-synuclein overexpression. Neurobiol Aging 28(2):248–257
Lee BD, Shin JH, VanKampen J, Petrucelli L, West AB, Ko HS, Lee YI, Maguire-Zeiss KA, Bowers WJ, Federoff HJ, Dawson VL, Dawson TM (2010) Inhibitors of leucine-rich repeat kinase-2 protect against models of Parkinson’s disease. Nat Med 16(9):998–1000
Lesage S, Brice A (2009) Parkinson’s disease: from monogenic forms to genetic susceptibility factors. Hum Mol Genet 18(R1):R48–R59
Li Y, Liu W, Oo TF, Wang L, Tang Y, Jackson-Lewis V, Zhou C, Geghman K, Bogdanov M, Przedborski S, Beal MF, Burke RE, Li C (2009) Mutant LRRK2(R1441G) BAC transgenic mice recapitulate cardinal features of Parkinson’s disease. Nat Neurosci 12(7):826–828
Li X, Patel JC, Wang J, Avshalumov MV, Nicholson C, Buxbaum JD, Elder GA, Rice ME, Yue Z (2010) Enhanced striatal dopamine transmission and motor performance with LRRK2 overexpression in mice is eliminated by familial Parkinson’s disease mutation G2019S. J Neurosci 30(5):1788–1797
Lo Bianco C, Ridet JL, Schneider BL, Deglon N, Aebischer P (2002) alpha-Synucleinopathy and selective dopaminergic neuron loss in a rat lentiviral-based model of Parkinson’s disease. Proc Natl Acad Sci U S A 99(16):10813–10818
Lock M, Alvira M, Vandenberghe LH, Samanta A, Toelen J, Debyser Z, Wilson JM (2010) Rapid, simple and versatile manufacturing of recombinant adeno-associated virus vectors at scale. Hum Gene Ther 21(10):1259–1271
Lu XH, Fleming SM, Meurers B, Ackerson LC, Mortazavi F, Lo V, Hernandez D, Sulzer D, Jackson GR, Maidment NT, Chesselet MF, Yang XW (2009) Bacterial artificial chromosome transgenic mice expressing a truncated mutant parkin exhibit age-dependent hypokinetic motor deficits, dopaminergic neuron degeneration, and accumulation of proteinase K-resistant alpha-synuclein. J Neurosci 29(7):1962–1976
Ma SY, Roytta M, Rinne JO, Collan Y, Rinne UK (1997) Correlation between neuromorphometry in the substantia nigra and clinical features in Parkinson’s disease using dissector counts. J Neurol Sci 151(1):83–87
Magen I, Chesselet MF (2010) Genetic mouse models of Parkinson’s disease—the state of the art. Prog Brain Res 184:53–87
Maingay M, Romero-Ramos M, Carta M, Kirik D (2006) Ventral tegmental area dopamine neurons are resistant to human mutant alpha-synuclein overexpression. Neurobiol Dis 23(3):522–532
Mandel RJ (2010) CERE-110, an adeno-associated virus-based gene delivery vector expressing human nerve growth factor for the treatment of Alzheimer’s disease. Curr Opin Mol Ther 12(2):240–247
Manfredsson FP, Burger C, Sullivan LF, Muzyczka N, Lewin AS, Mandel RJ (2007) rAAV-mediated nigral human parkin over-expression partially ameliorates motor deficits via enhanced dopamine neurotransmission in a rat model of Parkinson’s disease. Exp Neurol 207(2):289–301
Markert JM, Medlock MD, Rabkin SD, Gillespie GY, Todo T, Hunter WD, Palmer CA, Feigenbaum F, Tornatore C, Tufaro F, Martuza RL (2000) Conditionally replicating herpes simplex virus mutant, G207 for the treatment of malignant glioma: results of a phase I trial. Gene Ther 7(10):867–874
Masliah E, Rockenstein E, Veinbergs I, Mallory M, Hashimoto M, Takeda A, Sagara Y, Sisk A, Mucke L (2000) Dopaminergic loss and inclusion body formation in alpha-synuclein mice: implications for neurodegenerative disorders. Science 287(5456):1265–1269
Mastakov MY, Baer K, Xu R, Fitzsimons H, During MJ (2001) Combined injection of rAAV with mannitol enhances gene expression in the rat brain. Mol Ther 3(2):225–232
Mastakov MY, Baer K, Kotin RM, During MJ (2002) Recombinant adeno-associated virus serotypes 2- and 5-mediated gene transfer in the mammalian brain: quantitative analysis of heparin co-infusion. Mol Ther 5(4):371–380
McFarland NR, Lee JS, Hyman BT, McLean PJ (2009a) Comparison of transduction efficiency of recombinant AAV serotypes 1, 2, 5, and 8 in the rat nigrostriatal system. J Neurochem 109(3):838–845
McFarland NR, Fan Z, Xu K, Schwarzschild MA, Feany MB, Hyman BT, McLean PJ (2009b) Alpha-synuclein S129 phosphorylation mutants do not alter nigrostriatal toxicity in a rat model of Parkinson disease. J Neuropathol Exp Neurol 68(5):515–524
Miao CH, Snyder RO, Schowalter DB, Patijn GA, Donahue B, Winther B, Kay MA (1998) The kinetics of rAAV integration in the liver. Nat Genet 19(1):13–15
Mochizuki H, Yamada M, Mizuno Y (2006) Alpha-synuclein overexpression model. J Neural Transm Suppl 70:281–284
Moskalenko M, Chen L, van Roey M, Donahue BA, Snyder RO, McArthur JG, Patel SD (2000) Epitope mapping of human anti-adeno-associated virus type 2 neutralizing antibodies: implications for gene therapy and virus structure. J Virol 74(4):1761–1766
Naldini L, Blomer U, Gage FH, Trono D, Verma IM (1996a) Efficient transfer, integration, and sustained long-term expression of the transgene in adult rat brains injected with a lentiviral vector. Proc Natl Acad Sci U S A 93(21):11382–11388
Naldini L, Blomer U, Gallay P, Ory D, Mulligan R, Gage FH, Verma IM, Trono D (1996b) In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 272(5259):263–267
Oliveras-Salvá M, Macchi F, Coessens V, Deleersnijder A, Gérard M, Van der Perren A, Van den Haute C, Baekelandt V (2014) Total but not partial loss of PINK1 enhances α-synuclein-induced neurodegeneration in mouse substantia nigra. Neurobiol Aging
Oliveras-Salvá M, Van der Perren A, Casadei N, Nuber S, D'Hooge R, Van den Haute C, Baekelandt V (2013) rAAV vector-mediated overexpression of α-synuclein in mouse substantia nigra induces protein aggregation and progressive dose-dependent neurodegeneration. Mol Neurodegener 8:44
Paisan-Ruiz C (2009) LRRK2 gene variation and its contribution to Parkinson disease. Hum Mutat 30(8):1153–1160
Pankratz N, Wilk JB, Latourelle JC, DeStefano AL, Halter C, Pugh EW, Doheny KF, Gusella JF, Nichols WC, Foroud T, Myers RH (2009) Genomewide association study for susceptibility genes contributing to familial Parkinson disease. Hum Genet 124(6):593–605
Passini MA, Watson DJ, Wolfe JH (2004) Gene delivery to the mouse brain with adeno-associated virus. Methods Mol Biol 246:225–236
Paterna JC, Feldon J, Bueler H (2004) Transduction profiles of recombinant adeno-associated virus vectors derived from serotypes 2 and 5 in the nigrostriatal system of rats. J Virol 78(13):6808–6817
Perez FA, Palmiter RD (2005) Parkin-deficient mice are not a robust model of parkinsonism. Proc Natl Acad Sci U S A 102(6):2174–2179
Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, Pike B, Root H, Rubenstein J, Boyer R, Stenroos ES, Chandrasekharappa S, Athanassiadou A, Papapetropoulos T, Johnson WG, Lazzarini AM, Duvoisin RC, Di Iorio G, Golbe LI, Nussbaum RL (1997) Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease. Science 276(5321):2045–2047
Rabinowitz JE, Samulski RJ (2000) Building a better vector: the manipulation of AAV virions. Virology 278(2):301–308
Ramonet D, Daher JP, Lin BM, Stafa K, Kim J, Banerjee R, Westerlund M, Pletnikova O, Glauser L, Yang L, Liu Y, Swing DA, Beal MF, Troncoso JC, McCaffery JM, Jenkins NA, Copeland NG, Galter D, Thomas B, Lee MK, Dawson TM, Dawson VL, Moore DJ (2011) Dopaminergic neuronal loss, reduced neurite complexity and autophagic abnormalities in transgenic mice expressing G2019S mutant LRRK2. PLoS ONE 6(4):e18568
Richfield EK, Thiruchelvam MJ, Cory-Slechta DA, Wuertzer C, Gainetdinov RR, Caron MG, Di Monte DA, Federoff HJ (2002) Behavioral and neurochemical effects of wild-type and mutated human alpha-synuclein in transgenic mice. Exp Neurol 175(1):35–48
Rutledge EA, Halbert CL, Russell DW (1998) Infectious clones and vectors derived from adeno-associated virus (AAV) serotypes other than AAV type 2. J Virol 72(1):309–319
Samulski RJ, Berns KI, Tan M, Muzyczka N (1982) Cloning of adeno-associated virus into pBR322: rescue of intact virus from the recombinant plasmid in human cells. Proc Natl Acad Sci U S A 79(6):2077–2081
Samulski RJ, Chang LS, Shenk T (1987) A recombinant plasmid from which an infectious adeno-associated virus genome can be excised in vitro and its use to study viral replication. J Virol 61(10):3096–3101
Sanchez-Guajardo V, Febbraro F, Kirik D, Romero-Ramos M (2010) Microglia acquire distinct activation profiles depending on the degree of alpha-synuclein neuropathology in a rAAV based model of Parkinson’s disease. PLoS ONE 5(1):e8784
Satake W, Nakabayashi Y, Mizuta I, Hirota Y, Ito C, Kubo M, Kawaguchi T, Tsunoda T, Watanabe M, Takeda A, Tomiyama H, Nakashima K, Hasegawa K, Obata F, Yoshikawa T, Kawakami H, Sakoda S, Yamamoto M, Hattori N, Murata M, Nakamura Y, Toda T (2009) Genome-wide association study identifies common variants at four loci as genetic risk factors for Parkinson’s disease. Nat Genet 41(12):1303–1307
Schmidt M, Grot E, Cervenka P, Wainer S, Buck C, Chiorini JA (2006) Identification and characterization of novel adeno-associated virus isolates in ATCC virus stocks. J Virol 80(10):5082–5085
Shelkovnikova TA, Ustyugov AA, Millership S, Peters O, Anichtchik O, Spillantini MG, Buchman VL, Bachurin SO, Ninkina NN (2011) Dimebon does not ameliorate pathological changes caused by expression of truncated (1-120) human alpha-synuclein in dopaminergic neurons of transgenic mice. Neurodegener Dis 8(6):430–437
Shin JH, Ko HS, Kang H, Lee Y, Lee YI, Pletinkova O, Troconso JC, Dawson VL, Dawson TM (2011) PARIS (ZNF746) repression of PGC-1alpha contributes to neurodegeneration in Parkinson’s disease. Cell 144(5):689–702
Shults CW (2006) Lewy bodies. Proc Natl Acad Sci U S A 103(6):1661–1668
Simon-Sanchez J, Schulte C, Bras JM, Sharma M, Gibbs JR, Berg D, Paisan-Ruiz C, Lichtner P, Scholz SW, Hernandez DG, Kruger R, Federoff M, Klein C, Goate A, Perlmutter J, Bonin M, Nalls MA, Illig T, Gieger C, Houlden H, Steffens M, Okun MS, Racette BA, Cookson MR, Foote KD, Fernandez HH, Traynor BJ, Schreiber S, Arepalli S, Zonozi R, Gwinn K, van der Brug M, Lopez G, Chanock SJ, Schatzkin A, Park Y, Hollenbeck A, Gao J, Huang X, Wood NW, Lorenz D, Deuschl G, Chen H, Riess O, Hardy JA, Singleton AB, Gasser T (2009) Genome-wide association study reveals genetic risk underlying Parkinson’s disease. Nat Genet 41(12):1308–1312
Singleton AB, Farrer M, Johnson J, Singleton A, Hague S, Kachergus J, Hulihan M, Peuralinna T, Dutra A, Nussbaum R, Lincoln S, Crawley A, Hanson M, Maraganore D, Adler C, Cookson MR, Muenter M, Baptista M, Miller D, Blancato J, Hardy J, Gwinn-Hardy K (2003) alpha-Synuclein locus triplication causes Parkinson’s disease. Science 302(5646):841
Spillantini MG, Schmidt ML, Lee VM, Trojanowski JQ, Jakes R, Goedert M (1997) Alpha-synuclein in Lewy bodies. Nature 388(6645):839–840
St Martin JL, Klucken J, Outeiro TF, Nguyen P, Keller-McGandy C, Cantuti-Castelvetri I, Grammatopoulos TN, Standaert DG, Hyman BT, McLean PJ (2007) Dopaminergic neuron loss and up-regulation of chaperone protein mRNA induced by targeted over-expression of alpha-synuclein in mouse substantia nigra. J Neurochem 100(6):1449–1457
Taymans JM, Vandenberghe LH, Haute CV, Thiry I, Deroose CM, Mortelmans L, Wilson JM, Debyser Z, Baekelandt V (2007) Comparative analysis of adeno-associated viral vector serotypes 1, 2, 5, 7, and 8 in mouse brain. Hum Gene Ther 18(3):195–206
Tenenbaum L, Chtarto A, Lehtonen E, Velu T, Brotchi J, Levivier M (2004) Recombinant AAV-mediated gene delivery to the central nervous system. J Gene Med 6(Suppl 1):S212–S222
Theodore S, Cao S, McLean PJ, Standaert DG (2008) Targeted overexpression of human alpha-synuclein triggers microglial activation and an adaptive immune response in a mouse model of Parkinson disease. J Neuropathol Exp Neurol 67(12):1149–1158
Thiruchelvam M, Brockel BJ, Richfield EK, Baggs RB, Cory-Slechta DA (2000) Potentiated and preferential effects of combined paraquat and maneb on nigrostriatal dopamine systems: environmental risk factors for Parkinson’s disease? Brain Res 873(2):225–234
Thiruchelvam MJ, Powers JM, Cory-Slechta DA, Richfield EK (2004) Risk factors for dopaminergic neuron loss in human alpha-synuclein transgenic mice. Eur J Neurosci 19(4):845–854
Toelen J, Van der Perren A, Carlon M, Michiels M, Lock M, Vandenberghe L, Bannert N, Wilson JM, Gijsbers R, Debyser Z (2014) Novel approach to research grade AAV vector manufacturing and separation of distinct AAV serotypes for in vivo applications (in revision)
Tofaris GK, Garcia Reitbock P, Humby T, Lambourne SL, O’Connell M, Ghetti B, Gossage H, Emson PC, Wilkinson LS, Goedert M, Spillantini MG (2006) Pathological changes in dopaminergic nerve cells of the substantia nigra and olfactory bulb in mice transgenic for truncated human alpha-synuclein(1-120): implications for Lewy body disorders. J Neurosci 26 (15):3942–3950
Tong Y, Pisani A, Martella G, Karouani M, Yamaguchi H, Pothos EN, Shen J (2009) R1441C mutation in LRRK2 impairs dopaminergic neurotransmission in mice. Proc Natl Acad Sci U S A 106(34):14622–14627
Ulusoy A, Sahin G, Bjorklund T, Aebischer P, Kirik D (2009) Dose optimization for long-term rAAV-mediated RNA interference in the nigrostriatal projection neurons. Mol Ther 17(9):1574–1584
Ungerstedt U, Arbuthnott GW (1970) Quantitative recording of rotational behavior in rats after 6-hydroxy-dopamine lesions of the nigrostriatal dopamine system. Brain Res 24(3):485–493
Van der Perren A, Toelen J, Carlon M, Van den Haute C, Coun F, Heeman B, Reumers V, Vandenberghe LH, Wilson JM, Debyser Z, Baekelandt V (2011) Efficient and stable transduction of dopaminergic neurons in rat substantia nigra by rAAV 2/1, 2/2, 2/5, 2/6.2, 2/7, 2/8 and 2/9. Gene Ther 18(5):517–527
Van der Perren A, Macchi F, Toelen J, Van Rompuy AS, Casteels C, Sarre S, Gerard G, Casadei N, Nuber S, Himmelreich U, Osorio Garcia MI, Michotte Y, Bormans G, Van Laere K, Gijsbers R, Van den Haute C, Debyser Z, Baekelandt V (2014) Longitudinal characterization of a robust rat model for Parkinson's disease based on overexpression of alpha-synuclein with rAAV2/7 viral vectors (in revision)
Vercammen L, Van der Perren A, Vaudano E, Gijsbers R, Debyser Z, Van den Haute C, Baekelandt V (2006) Parkin protects against neurotoxicity in the 6-hydroxydopamine rat model for Parkinson’s disease. Mol Ther 14(5):716–723
Von Coelln R, Thomas B, Savitt JM, Lim KL, Sasaki M, Hess EJ, Dawson VL, Dawson TM (2004) Loss of locus coeruleus neurons and reduced startle in parkin null mice. Proc Natl Acad Sci U S A 101(29):10744–10749
Wakamatsu M, Ishii A, Iwata S, Sakagami J, Ukai Y, Ono M, Kanbe D, Muramatsu S, Kobayashi K, Iwatsubo T, Yoshimoto M (2008) Selective loss of nigral dopamine neurons induced by overexpression of truncated human alpha-synuclein in mice. Neurobiol Aging 29(4):574–585
Wang L, Qi X, Shen R, Sun Y, Tuveson DA (2009) An shRNA silencing a non-toxic transgene reduces nutrient consumption and increases production of adenoviral vectors in a novel packaging cell. J Cell Physiol 219(2):365–371
Winklhofer KF (2007) The parkin protein as a therapeutic target in Parkinson’s disease. Expert Opin Ther Targets 11(12):1543–1552
Winner B, Jappelli R, Maji SK, Desplats PA, Boyer L, Aigner S, Hetzer C, Loher T, Vilar M, Campioni S, Tzitzilonis C, Soragni A, Jessberger S, Mira H, Consiglio A, Pham E, Masliah E, Gage FH, Riek R (2011) In vivo demonstration that alpha-synuclein oligomers are toxic. Proc Natl Acad Sci U S A 108(10):4194–4199
Wirdefeldt K, Adami HO, Cole P, Trichopoulos D, Mandel J (2011) Epidemiology and etiology of Parkinson’s disease: a review of the evidence. Eur J Epidemiol 26(Suppl 1):S1–S58
Wright JF (2009) Transient transfection methods for clinical adeno-associated viral vector production. Hum Gene Ther 20(7):698–706
Wu Z, Asokan A, Samulski RJ (2006) Adeno-associated virus serotypes: vector toolkit for human gene therapy. Mol Ther 14(3):316–327
Xiao X, Li J, Samulski RJ (1996) Efficient long-term gene transfer into muscle tissue of immunocompetent mice by adeno-associated virus vector. J Virol 70(11):8098–8108
Yamada M, Iwatsubo T, Mizuno Y, Mochizuki H (2004) Overexpression of alpha-synuclein in rat substantia nigra results in loss of dopaminergic neurons, phosphorylation of alpha-synuclein and activation of caspase-9: resemblance to pathogenetic changes in Parkinson’s disease. J Neurochem 91(2):451–461
Zarranz JJ, Alegre J, Gomez-Esteban JC, Lezcano E, Ros R, Ampuero I, Vidal L, Hoenicka J, Rodriguez O, Atares B, Llorens V, Gomez Tortosa E, del Ser T, Munoz DG, de Yebenes JG (2004) The new mutation, E46 K, of alpha-synuclein causes Parkinson and Lewy body dementia. Ann Neurol 55(2):164–173
Zennou V, Petit C, Guetard D, Nerhbass U, Montagnier L, Charneau P (2000) HIV-1 genome nuclear import is mediated by a central DNA flap. Cell 101(2):173–185
Zhu XR, Maskri L, Herold C, Bader V, Stichel CC, Gunturkun O, Lubbert H (2007) Non-motor behavioural impairments in parkin-deficient mice. Eur J Neurosci 26(7):1902–1911
Zufferey R, Nagy D, Mandel RJ, Naldini L, Trono D (1997) Multiply attenuated lentiviral vector achieves efficient gene delivery in vivo. Nat Biotechnol 15(9):871–875
Zufferey R, Donello JE, Trono D, Hope TJ (1999) Woodchuck hepatitis virus posttranscriptional regulatory element enhances expression of transgenes delivered by retroviral vectors. J Virol 73(4):2886–2892
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Van der Perren, A., Van den Haute, C., Baekelandt, V. (2014). Viral Vector-Based Models of Parkinson’s Disease. In: Nguyen, H., Cenci, M. (eds) Behavioral Neurobiology of Huntington's Disease and Parkinson's Disease. Current Topics in Behavioral Neurosciences, vol 22. Springer, Berlin, Heidelberg. https://doi.org/10.1007/7854_2014_310
Download citation
DOI: https://doi.org/10.1007/7854_2014_310
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-46343-7
Online ISBN: 978-3-662-46344-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)