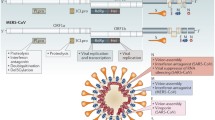
Abstract
Coronaviruses (CoVs) infect humans and can cause lung, kidney, heart, brain, and intestinal infections that can range from mild to lethal type. Especially, β-coronaviruses causing severe-acute respiratory syndrome (SARS)-CoV-1, the Middle East respiratory syndrome (MERS)-CoV, and SARS-CoV-2 (2019-nCoV) are dangerous and can easily be transmitted from human to human. The outbreak of coronavirus disease 2019 (COVID-19), which is caused by SARS-CoV-2 infection, has become a global crisis. In this situation, the development of anticoronaviral therapies is highly warranted. However, to date, no approved vaccines or drugs against CoV infections are available. In this chapter, we have made an effort to discuss molecular level targets for the development of therapies against coronavirus diseases, SARS-CoV-1, MERS-CoV, and SARS-CoV-2, targeting CoV replication and its life cycle. These targets include the spike glycoprotein and its host-receptors for viral entry, proteases that are essential for cleaving polyproteins to produce functional proteins, and RNA-dependent RNA polymerase for viral RNA replication.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Pillaiyar T, Meenakshisundaram S, Manickam M (2020) Recent discovery and development of inhibitors targeting coronaviruses. Drug Discov Today 25(4):668–688
Pillaiyar T, Manickam M, Namasivayam V et al (2016) An overview of severe acute respiratory syndrome-coronavirus (SARS-CoV) 3CL protease inhibitors: peptidomimetics and small molecule chemotherapy. J Med Chem 59:6595–6628
Pillaiyar T, Manickam M, Jung SH (2015) Middle East respiratory syndrome-coronavirus (MERS-CoV): an updated overview and pharmacotherapeutics. Med Chem 5(8):361–372
de Groot RJ et al (2011) Family coronaviridae. In: King AMQ (ed) Ninth Report of the International Committee on Taxonomy of Viruses. Elsevier, Oxford, pp 806–828
Geller C, Varbanov M, Duval RE (2012) Human coronaviruses: insights into environmental resistance and its influence on the development of new antiseptic strategies. Viruses 4(11):3044–3068
Hamre D, Procknow JJ (1966) A new virus isolated from the human respiratory tract. Proc Soc Exp Biol Med 121(1):190–193
McIntosh K, Dees JH, Becker WB et al (1967) Recovery in tracheal organ cultures of novel viruses from patients with respiratory disease. Proc Natl Acad Sci U S A 57(4):933–940
WHO (2003) Communicable disease surveillance and response. World Health Organization, Geneva. http://www.who.int/csr/sars/archive/2003_05_07a/en, http://www.who.int/csr/sars/country/en/country2003_08_15.pdf
WHO (2014) Middle East respiratory syndrome coronavirus (MERS-CoV) summary and literature update-as of 20 January. WHO, Geneva
Pillaiyar T, Wendt LL, Manickam M et al (2020) The recent outbreaks of human coronaviruses: a medicinal chemistry perspective. Med Res Rev. https://doi.org/10.1002/med.21724
Su S, Wong G, Shi W et al (2016) Epidemiology, genetic recombination and pathogenesis of coronaviruses. Trends Microbiol 24(6):490–502
Forni D, Cagliani R, Clerici M, Sironi M (2017) Molecular evolution of human coronavirus genomes. Trends Microbiol 25(1):35–48
Cui J, Li F, Shi ZL (2019) Origin and evolution of pathogenic coronaviruses. Nat Rev Microbiol 17(3):181–192
Zhu N, Zhang D, Wang W et al (2020) A novel coronavirus from patients with pneumonia in China 2019. N Engl J Med 382:727–733
Fehr AR, Perlman S (2015) Coronaviruses: an overview of their replication and pathogenesis. In: Coronaviruses. Methods in molecular biology, vol 1282. Humana Press, New York, NY, pp 1–23
Liu C, Zhou Q, Li Y et al (2020) Research and development on therapeutic agents and vaccines for COVID-19 and related human coronavirus diseases. ACS Cent Sci 6:315–331
Kai H, Kai M (2020) Interactions of coronaviruses with ACE2, angiotensin-II, and RAS inhibitors-lessons from available evidence and insights into COVID-19. Hypertens Res 43:648. https://doi.org/10.1038/s41440-020-0455-8
Van Doremalen N, Miazgowicz KL, Milne-Price S et al (2014) Host species restriction of Middle East respiratory syndrome coronavirus through its receptor, dipeptidyl peptidase 4. J Virol 88(16):9220–9232
Yeager CL, Ashmun RA, Williams RK et al (1992) Human aminopeptidase N is a receptor for human coronavirus 229E. Nature 357(6377):420–422
Huang X, Dong W, Milewska A et al (2015) Human coronavirus HKU1 spike protein uses O-acetylated sialic acid as an attachment receptor determinant and employs hemagglutinin-esterase protein as a receptor-destroying enzyme. J Virol 89(14):7202–7213
Li G, Fan Y, Lai Y et al (2020) Coronavirus infections and immune responses. J Med Virol 92(4):424–432
Ben-Zvi I, Kivity S, Langevitz P, Shoenfeld Y (2012) Hydroxychloroquine: from malaria to autoimmunity. Clin Rev Allergy Immunol 42(2):145–153
Savarino A, Di L, Trani DI, Cauda R, Cassone A (2006) New insights into the antiviral effects of chloroquine. Lancet Infect Dis 6(2):67–69
Yan Y, Zou Z, Sun Y, Li X, Xu KF, Wei Y et al (2013) Anti-malaria drug chloroquine is highly effective in treating avian influenza A H5N1 virus infection in an animal model. Cell Res 32(2):300–302
Vincent MJ, Bergeron E, Benjannet S, Erickson BR, Rollin BE, Ksiazek TG et al (2005) Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J 2:69
Al-Bari MAA (2017) Targeting endosomal acidification by chloroquine analogs as a promising strategy for the treatment of emerging viral diseases. Pharmacol Res Perspect 5(1):e00293
Keyaerts E, Vijge L, Maes P, Neyts J, Van Ranst M (2004) In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine. Biochem Biophys Res Commun 323(1):264–268
Yao X, Ye F, Zhang M, Cui C, Huang B, Nui P et al (2020) In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis 71:732. https://doi.org/10.1093/cid/ciaa237
Gao J, Tian Z, Yang X (2020) Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends 14(1):72
Stahlmann R, Lode H (2020) Medication for COVID-19 – an overview of approaches currently under study. Dtsch Arztebl Int 117(13):213–219
Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M et al (2020) Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents 56:105949. https://doi.org/10.1016/j.ijantimicag.2020.105949
Chen Z, Hu J, Zhang Z, Jiang S, Han S, Yan D et al (2020) Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. MedRXiv. https://doi.org/10.1101/2020.03.22.20040758
Zhang Q, Wang Y, Qi C, Shen L, Li J (2020) Clinical trial analysis of 2019-nCoVtherapy registered in China. J Med Virol 92:540. https://doi.org/10.1002/jmv.25733
Cortegiani A, Ingoglia G, Ippolito M, Giarratano A, Einav S (2020) A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care 57:279. https://doi.org/10.1016/j.jcrc.2020.03.005
Chan KW, Wong VT, Tang SCW (2020) COVID-19: an update on the epidemiological, clinical preventive and therapeutic evidence and guidelines of integrative Chinese-Western medicine for the management of 2019 novel coronavirus disease. Am J Chin Med 48:737–762. https://doi.org/10.1142/S0192415X20500378
ClinicalTrials gov (2020) NCT04303507
ClinicalTrials gov (2020) NCT04303299
Blaising J, Polyak SJ, Pécheur EI (2014) Arbidol as a broad spectrum antiviral: an update. Antiviral Res 107:84–94
Leneva IA, Russell RJ, Boriskin YS, Hay AJ Characteristics of arbidol-resistant mutants of influenza virus: implications for the mechanism of anti-influenza action of arbidol. Antiviral Res 81(2):132–140
Boriskin YS, Pécheur EI, Polyak SJ Arbidol: a broad-spectrum antiviral that inhibits acute and chronic HCV infection. Virol J 3:56
Blaising J, Lévy PL, Polyak SJ, Stanifer M, Boulant S, Pécheur EI (2013) Arbidol inhibits viral entry by interfering with clathrin-dependent trafficking. Antiviral Res 100(1):215–219
Pécheur EI, Borisevich V, Halfmann P, Morrey JD, Smee DF, Prichard M et al (2016) The synthetic antiviral drug arbidol inhibits globally prevalent pathogenic viruses. J Virol 90(6):3086–3092
Hulseberg CE, Fénéant L, Szymanska de Wijs KM, Kessler NP, Nelson EA, Shoemaker CJ et al (2019) Arbidol and other low-molecular-weight drugs that inhibit Lassa and Ebola viruses. J Virol 93(8):e02185–e02118
Wang Z, Yang B, Li Q, Wen L, Zhang R (2020) Clinical features of 69 cases with coronavirus disease 2019 in Wuhan China. Clin Infect Dis 79:763. https://doi.org/10.1093/cid/ciaa272
Deng L, Li C, Zeng Q, Liu X, Li X, Zhang H et al (2020) Arbidol combined with LPV/r versus LPV/r alone against corona virus disease 2019: a retrospective cohort study. J Infect 81:e1. https://doi.org/10.1016/j.jinf.2020.03.002
ClinicalTrials.gov (2020) NCT04255017
ClinicalTrials.gov (2020) NCT04260594
ClinicalTrials.gov (2020) NCT04273763
ClinicalTrials.gov (2020) NCT04286503
Ohkoshi M, Fujji S (1983) Effect of the synthetic protease inhibitor [N,N-dimethylcarbamoyl-methyl 4-(4-guanidinobenzoyloxy)-phenylacetate] methanesulfate on carcinogenesis by 3 methylcholanthrene in mouse skin. J Natl Cancer Inst 71(5):1053–1057
Ohkoshi M, Oka T (1984) Clinical experience with a protease inhibitor [N,N dimethylcarbamoylmethyl4-(4-guanidinobenzoyloxy)-phenylacetate] methanesulfate for prevention of recurrence of carcinoma of the mouth and in treatment of terminal carcinoma. J Maxillofac Surg 12(4):148–152
Ikeda S, Manabe M, Muramatsu T, Takamori K, Ogawa H (1988) Protease inhibitor therapy for recessive dystrophic epidermolysis bullosa. In vitro effect and clinical trial with camostat mesylate. J Am Acad Dermatol 18(16):1246–1252
Göke B, Stöckmann F, Müller R, Lankisch PG, Creutzfeld W (1984) Effect of a specific serine protease inhibitor on the rat pancreas: systemic administration of camostat and exocrine pancreatic secretion. Digestion 30(3):171–178
Adler G, Müllenhoff A, Koop I, Bozkurt T, Göke B, Beglinger C et al (2020) Stimulation of pancreatic secretion in man by a protease inhibitor (camostate). Eur J Clin Invest 18(1)
Sai JK, Suyama M, Kubokawa Y, Matsumura Y, Inami K, Watanabe S (2010) Efficacy of camostat mesilate against dyspepsia associated with non-alcoholic mild pancreatic disease. J Gastroenterol 45(3):335–341
Yamawaki H, Futagami S, Kaneto K, Agawa S, Higuchi K, Murakami M et al (2019) Camostat mesilate, pancrelipase, and rabeprazole combination therapy improves epigastric pain in early chronic pancreatitis and functional dyspepsia with pancreatic enzyme abnormalities. Digestion 99(4):283–292
Ramsey ML, Nuttall J, Hart PA (2019) A phase 1/2 trial to evaluate the pharmacokinetics, safety, and efficacy of NI-03 in patients with Chronic pancreatitis: study protocol for a randomized controlled trial on the assessment of camostat treatment in chronic pancreatitis (TACTIC). Trials 20(1):501
ClinicalTrials.gov (2016) NCT02693093
Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S (2020) SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181:271–280
Iwako M, Ino Y, Motoyoshi A, Ozeki M, Sato T, Kumuri M et al (1986) Pharmacological studies of FUT-175, nafamostat mesilate. V. Effects on the pancreatic enzymes and experimental acute pancreatitis in rats. Jpn J Pharmacol 41(2):155–162
Hiraishi M, Yamazaki Z, Ichikawa K, Kanai F, Idezuki Y, Onishi K et al (1988) Plasma collection using nafamostat mesilate and dipyridamole as an anticoagulant. Int J Artif Organ 11(3):212–216
Hirota M, Shimosegawa T, Kitamura K, Takeda K, Takeyama Y, Mayumi T et al (2020) Continuous regional arterial infusion versus intravenous administration of the protease inhibitor nafamostat mesilate for predicted severe acute pancreatitis: a multicenter, randomized, open-label phase 2 trial. J Gastroenterol 55(3):342–352
Yamamoto M, Matsuyama S, Li X, Takeda M, Kawaguchi Y, Inoue JI et al (2016) Identification of nafamostat as a potent inhibitor of middle east respiratory syndrome coronavirus S protein-mediated membrane fusion using the split-protein based cell-cell fusion assay. Antimicrob Agents Chemother 60(11):6532–6539
Lu R, Zhao X, Li J, Niu P, Wang B, Wu H et al (2020) Genomic characterization and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet 395(10224):565–574
Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D (2020) Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell 181:281. https://doi.org/10.1016/j.cell.2020.02.058
Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M et al (2020) Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res 30(3):269–271
Hirota M, Shimosegawa T, Kitamura K, Takeda K, Takeyama Y, Mayumi T et al (2020) Continuous regional arterial infusion versus intravenous administration of the protease inhibitor nafamostat mesilate for predicted severe acute pancreatitis: a multicenter, randomized, open-label, phase 2 trial. J Gastroenterol 55(3):342–352
Adedeji AO, Severson W, Jonsson C et al (2013) Novel inhibitors of severe acute respiratory syndrome coronavirus entry that act by three distinct mechanisms. J Virol 87(14):8017–8028
Van Dongen MJP, Kadam RU, Juraszek J et al (2019) A small-molecule fusion inhibitor of influenza virus is orally active in mice. Science 363(6431):eaar6221
Yi L, Li Z, Yuan K et al (2004) Small molecules blocking the entry of severe acute respiratory syndrome coronavirus into host cells. J Virol 78(20):11334–11339
Coughlin M, Lou G, Martinez O et al (2007) Generation and characterization of human monoclonal neutralizing antibodies with distinct binding and sequence features against SARS coronavirus using XenoMouse. Virology 361(1):93–102
Sisk JM, Frieman MB, Machamer CE (2018) Coronavirus S protein-induced fusion is blocked prior to hemifusion by Abl kinase inhibitors. J Gen Virol 99(5):619–630
Coleman CM, Sisk JM, Mingo RM et al (2016) Abelson kinase inhibitors are potent inhibitors of severe acute respiratory syndrome coronavirus and Middle East respiratory syndrome coronavirus fusion. J Virol 90:8924–8933.aa
Xu X, Chen P, Wang J et al (2020) Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci China Life Sci 63(3):457–460
Jin Z, Du X, Xu Y et al (2020) Structure of M pro from SARS-CoV-2 and discovery of its inhibitors. Nature 582(7811):289–293
Singh N, Halliday AC, Thomas JM et al (2013) A safe lithium mimetic for bipolar disorder. Nat Commun 4(1332):1–7
Lynch E, Kil J, Tran U et al (2007) Development of ebselen, a glutathione peroxidase mimic, for the prevention and treatment of noise-induced hearing loss. Hear Res 226(1–2):44–51
Baig MH, Sharma T, Ahmad I, Abohashrh M, Alam MM, Dong J-J (2021) Is PF-00835231 a Pan-SARS-CoV-2 Mpro Inhibitor? A comparative study. Molecules 26, 1678. https://doi.org/10.3390/molecules26061678
de Vries M, Mohamed AS, Prescott RA, Valero-Jimenez AM, Desvignes L, O’Connor R, Steppan C, Anderson AS, Binder J, Dittmann M (2020) Comparative study of a 3CLpro inhibitor and remdesivir against both major SARS-CoV-2 clades in human airway models. BioRxiv. https://doi.org/10.1101/2020.08.28.272880
Dai W, Zhang B, Su H et al (2020) Structure-based design of antiviral drug candidates targeting the SARS-CoV-2 main protease. Science 368(6497):1331–1335
Zhang L, Lin D, Kusov Y et al (2020) α-Ketoamides as broad-spectrum inhibitors of coronavirus and enterovirus replication: structure-based design, synthesis, and activity assessment. J Med Chem 63(9):4562–4578
Shin JS, Jung E, Kim M, Baric RS, Go YY (2018) Saracatinib inhibits Middle East respiratory syndrome-coronavirus replication in vitro. Viruses 10:283
Ho TY, Wu SL, Chen JC, Li CC, Hsiang CY (2007) Emodin blocks the SARS coronavirus spike protein and angiotensin converting enzyme 2 interaction. Antiviral Res 74:92–101
Kao RY, Tsui WH, Lee TS, Tanner JA, Watt RM, Huang JD, Hu L, Chen G, Chen Z, Zhang L, He T, Chan KH, Tse H, To AP, Ng LW, Wong BC, Tsoi HW, Yang D, Ho DD, Yuen KY (2004) Identification of novel small-molecule inhibitors of severe acute respiratory syndrome-associated coronavirus by chemical genetics. Chem Biol 11:1293–1299
Pillaiyar T, Meenakshisundaram S, Manickam M, Sankaranarayanan M (2020) A medicinal chemistry perspective of drug repositioning: recent advances and challenges in drug discovery. Eur J Med Chem 195:112275
Coleman CM, Sisk JM, Mingo RM, Nelson EA, White JM, Frieman MB (2016) Abelson kinase inhibitors are potent inhibitors of severe acute respiratory syndrome coronavirus and Middle East respiratory syndrome coronavirus fusion. J Virol 90:8924–8933
Sham HL, Kempf DJ, Molla A, Marsh KC, Kumar GN, Chen CM et al (1998) ABT-378, a highly potent inhibitor of the human immunodeficiency virus protease. Antimicrob Agents Chemother 42(12):3218–3224
Benson CA, Deeks SG, Brun SC, Gulick RM, Eron JJ, Kessler HA et al (2002) Safety and antiviral activity at 48 weeks of lopinavir/ritonavir plus nevirapine and 2 nucleoside reverse-transcriptase inhibitors in human immunodeficiency virus type 1-infected protease inhibitor-experienced patients. J Infect Dis 185(5):599–607
Corbett AH, Lim ML, Kashuba AD (2002) Kaletra (lopinavir/ritonavir). Ann Pharmacother 36(7–8):1193–1203
Chu CM, Cheng VCC, Hung IFN, Wong MML, Chan KH, Chan KS (2004) Role of lopinavir/ritonavir in the treatment of SARS: initial virological and clinical findings. Thorax 59(3):252–256
Chan JF, Yao Y, Yeung ML, Deng W, Bao L, Jia L (2015) Treatment with lopinavir/ritonavir or interferon-β1b improves outcome of MERS-CoV infection in a nonhuman primate model of common marmoset. J Infect Dis 212(12):1904–1913
Arabi YM, Asiri AY, Assiri AM, Aziz Jokhdar HA, Alothman A, Balkhy HH et al (2020) Treatment of Middle East respiratory syndrome with a combination of lopinavir/ritonavir and interferon-β1b (MIRACLE trial): statistical analysis plan for a recursive two-stage group sequential randomized controlled trial. Trials 21(1):8
Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G et al (2020) A trial of lopinavir/ritonavirin adults hospitalized with severe Covid-19. N Engl J Med 382:1787. https://doi.org/10.1056/NEJMoa2001282
ClinicalTrials.gov (2020) NCT04261907
ClinicalTrials.gov (2020) NCT04295551
ClinicalTrials.gov (2020) NCT04276688
ClinicalTrials.gov (2020) NCT04307693
ClinicalTrials.gov (2020) NCT04315948
NIH (2020) NIH clinical trial of hydroxychloroquine, a potential therapy for COVID-19, begins. National Institutes of Health (NIH) (Press release). Accessed 11 Apr 2020
Mehra MR, Desai SS, Ruschitzka F, Patel AN (2020) Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. Lancet 22:2020. https://doi.org/10.1016/S0140-6736(20)31180-6
Lambert DW, Yarski M, Warner FJ et al (2005) Tumor necrosis factor-alpha convertase (ADAM17) mediates regulated ectodomain shedding of the severe-acute respiratory syndrome-coronavirus (SARS-CoV) receptor, angiotensin-converting enzyme-2 (ACE2). J Biolumin Chemilumin 280:30113–30119
Haga S, Nagata N, Okamura T et al (2010) TACE antagonists blocking ACE2 shedding caused by the spike protein of SARS-CoV are candidate antiviral compounds. Antiviral Res 85:551–555
Towler P, Staker B, Prasad SG et al (2004) ACE2 X-ray structures reveal a large hinge-bending motion important for inhibitor binding and catalysis. J Biolumin Chemilumin 279:17996–18007
Kadam RU, Wilson IA (2017) Structural basis of influenza virus inhibition by the antiviral drug Arbidol. Proc Natl Acad Sci U S A 114(2):206–214
Li G, De Clercq E (2020) Therapeutic options for the 2019 novel coronavirus (2019-nCoV). Nat Rev Drug Discov 19:149–150
The Efficacy of lopinavir plus ritonavir and arbidol against novel coronavirus infection (ELACOI). https://clinicaltrials.gov/ct2/show/NCT04252885
Zheng YY, Ma YT, Zhang JY, Xie X (2020) COVID-19 and the cardiovascular system. Nat Rev Cardiol 17:259–260
Fang L, Karakiulakis G, Roth M (2020) Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med 8(4):e21.91
Warren TK, Jordan R, Lo MK, Ray AS, Mackman RL, Soloveva V et al (2016) Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys. Nature 531(7594):381–385
Lo MK, Jordan R, Arvey A, Sudhamsu J, Shrivastava-Ranjan P, Hotard AL et al (2017) GS-5734 and its parent nucleoside analog inhibit filo-, pneumo-, and paramyxoviruses. Sci Rep 7(1):43395
Sheahan TP, Sims AC, Graham RL, Menachery VD, Gralinski LE, Case JB et al (2017) Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses. Sci Transl Med 9(396):eaal3653
Agostini ML, Andres EL, Sims AC, Graham RL, Sheahan TP, Lu X et al (2018) Coronavirus susceptibility to the antiviral remdesivir (GS-5734) is mediated by theviral polymerase and the proofreading exoribonuclease. MBio 9(2):e00221–e00218
Tchesnokov EP, Feng JY, Porter DP, Götte M (2019) Mechanisms of inhibition ofEbola virus RNA-dependent RNA polymerase by remdesivir. Viruses 11(4):326
Gordon CJ, Tchesnokov EP, Feng JY, Porter DP, Götte M (2020) The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from middle east respiratory syndrome coronavirus. J Biol Chem 295:4773. https://doi.org/10.1074/jbc.AC120.013056
Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M et al (2020) Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) invitro. Cell Res 30(3):269–271
de Wit E, Feldmann F, Cronin J, Jordan R, Okumura A, Thomas T et al (2020) Prophylactic and therapeutic remdesivir (GS-5734) treatment in the 345 rhesus macaque model of MERS-CoV infection. Proc Natl Acad Sci U S A 117:6771. https://doi.org/10.1073/pnas.1922083117
Mulangu S, Dodd LE, Davey RT, Mbaya OT, Proschan M, Mukadi D et al (2019) A randomized, controlled trial of Ebola virus disease therapeutics. N Engl J Med 381(24):2293–2303
Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A et al (2020) Compassionate use of remdesivir for patients with severe COVID-19. N Engl J Med 382:2327. https://doi.org/10.1056/NEJMoa2007016
ClinicalTrials.gov (2020) NCT04252664
ClinicalTrials.gov (2020) NCT04257656
ClinicalTrials.gov (2020) NCT04280705
ClinicalTrials.gov (2020) NCT04292730
ClinicalTrials.gov (2020) NCT04292899
ClinicalTrials.gov (2020) NCT04302766
ClinicalTrials.gov (2020) NCT04314817
Graci JD, Cameron CE (2006) Mechanisms of action of ribavirin against distinct viruses. Rev Med Virol 16:37–48
Chu CM, Cheng VCC, Hung IFN et al (2004) Role of lopinavir/ritonavir in the treatment of SARS: initial virological and clinical findings. Thorax 59:252–256
Furuta Y, Takahashi K, Fukuda Y, Kuno M, Kamiyama T, Kozaki K et al (2002) In vitro and in vivo activities of anti-influenza virus compound T-705. Antimicrob Agents Chemother 46(4):977–981
Furuta Y, Komeno T, Nakamura T (2017) Favipiravir (T-705), A broad spectrum inhibitor of viral RNA polymerase. Proc Jpn Acad Ser B Phys Biol Sci 93(7):449–463
Jin Z, Smith LK, Rajwanshi VK, Kim B, Deval J (2014) The ambiguous base-pairing and high substrate efficiency of T-705 (favipiravir) ribofuranosyl 5′-triphosphate towards influenza A virus polymerase. PLoS One 8(7):e68347
Delang L, Abdelnabi R, Neyts J (2018) Favipiravir as a potential countermeasure against neglected and emerging RNA viruses. Antiviral Res 153:85–94
Furuta Y, Takahashi K, Shiraki K, Sakamoto K, Smee DF, Barnard DL et al (2009) T-705 (favipiravir) and related compounds: novel broad-spectrum inhibitors of RNAviral infections. Antiviral Res 82(3):95–102
Furuta Y, Takahashi K, Shiraki K et al (2009) T-705 (favipiravir) and related compounds: novel broad-spectrum inhibitors of RNA viral infections. Antiviral Res 82:95–102
Sissoko D, Laouenan C, Folkesson E, M’Lebing AB, Beavogui AH, Baize S et al (2016) Experimental treatment with favipiravir for Ebola virus disease (the JIKI Trial): a historically controlled, single-arm proof-of-concept trial in Guinea. PLoS Med 13(3):e1001967
ClinicalTrials.gov (2020) NCT04310228
Sheahan TP, Sims AC, Zhou S et al (2020) An orally bioavailable broad-spectrum antiviral inhibits SARS-CoV-2 in human airway epithelial cell cultures and multiple coronaviruses in mice. Sci Transl Med 12(541):eabb5883
COVID-19 first in human study to evaluate safety, tolerability, and pharmacokinetics of EIDD-2801 in healthy volunteers. https://clinicaltrials.gov/ct2/show/NCT04392219
Yin W, Mao C, Luan X et al (2020) Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir. Science 368(6498):1499–1504
Acknowledgment
Disclosure: Some parts of this article have been previously published in ref. 10.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Pillaiyar, T., Manickam, M., Meenakshisundaram, S., Benjamine, A.J. (2021). Candidate Drugs for the Potential Treatment of Coronavirus Diseases. In: Roy, K. (eds) In Silico Modeling of Drugs Against Coronaviruses. Methods in Pharmacology and Toxicology. Humana, New York, NY. https://doi.org/10.1007/7653_2020_67
Download citation
DOI: https://doi.org/10.1007/7653_2020_67
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1365-8
Online ISBN: 978-1-0716-1366-5
eBook Packages: Springer Protocols