Abstract
In the current chapter, digital morphometric analysis (DMA) was used to quantify two markers of biocompatibility around commonly used biomaterials. In the field of biomaterial evaluation for biocompatibility, more sophisticated methods are now being used to precisely characterize the elicited response from the surrounding tissue towards the implanted material. One reason for this is due to the fact that many newer biomaterial innovations are incorporating pharmaceutical agents (e.g., drug eluting stents and drug eluting balloons). Therefore, as described in many of the other chapters in this book, components of toxicology and pharmacology are being evaluated along with biocompatibility.
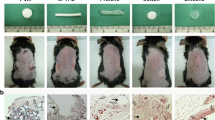
In this chapter, expanded polytetrafluoroethylene (ePTFE) was compared to polypropylene (PP) for inflammatory and foreign body response. Each material was implanted into dorsal subcutaneous spaces and evaluated after 2, 4, and 12 weeks. Each sample was reacted with an antibody to cluster of differentiation-68 (CD-68). The resulting slides were scanned and evaluated using DMA in order to obtain accurate, reproducible, and consistent results. Expanded PTFE demonstrated a lower overall weighted inflammatory score when compared to PP across all timepoints. This chapter describes the use of DMA as a novel approach to measure the inflammatory score that is associated with a specific biomaterial. Current and future medical devices will need to use various analytical tools to comprehensively assess device, biomaterial, or a combination therapy’s biocompatibility. The next chapter further describes how quantitative data from histology and immunohistochemistry assessments can be coupled with quantitative polymerase chain reactions (PCR) as assessment tools for product development.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Williams DF (1987) Definitions in biomaterials: proceedings of a consensus conference of the European society for biomaterials, Chester, England, 3–5 March 1986
Anderson JM (2001) Biological responses to materials. Annu Rev Mater Res 31(1):81–110
Anderson JM (1988) Inflammatory response to implants. Am Soc Artif Implant Organs J 34(2):101–107
Galante JO, Lemons J, Spector M, Wilson PD, Wright TM (1991) The biologic effects of implant materials. J Orthop Res 9(5):760–775. doi:10.1002/jor.1100090516
Anderson JM, McNally AK (2011) Biocompatibility of implants: lymphocyte/macrophage interactions. In: Seminars in immunopathology, vol 33, no. 3, Springer, pp 221–233. doi:10.1007/s00281-011-0244-1
Kidd KR, Dal Ponte DB, Kellar RS, Williams SK (2001) A comparative evaluation of the tissue responses associated with polymeric implants in the rat and mouse. J Biomed Mater Res 59(4):682–689
Kellar RS, Landeen LK, Shepherd BR, Naughton GK, Ratcliffe A, Williams SK (2001) Scaffold-based three-dimensional human fibroblast culture provides a structural matrix that supports angiogenesis in infarcted heart tissue. Circulation 104(17):2063–2068
Doussis IA, Gatter KC, Mason DY (1993) CD68 reactivity of non-macrophage derived tumours in cytological specimens. J Clin Pathol 46(4):334–336
Kellar RS, Lancaster JJ, Thai HM, Juneman E, Johnson NM, Byrne HG, Stansifer M, Arsanjani R, Baer M, Bebbington C, Flashner M, Yarranton G, Goldman S (2011) Antibody to granulocyte macrophage colony-stimulating factor reduces the number of activated tissue macrophages and improves left ventricular function after myocardial infarction in a rat coronary artery ligation model. J Cardiovasc Pharmacol 57(5):568–574
Nakopoulou L, Giannopoulou I, Gakiopoulou H, Liapis H, Tzonou A, Davaris PS (1999) Matrix metalloproteinase-1 and -3 in breast cancer: correlation with progesterone receptors and other clinicopathologic features. Hum Pathol 30(4):436–442. doi:10.1016/S0046-8177(99)90120-X
Słodkowska J, Filas V, Buszkiewicz E, Trzeciak P, Wojciechowski M, Koktysz R, Garcia Rojo M (2010) Study on breast carcinoma Her2/neu and hormonal receptors status assessed by automated images analysis systems: ACIS III (dako) and ScanScope (aperio). Folia Histochem Cytobiol 48(1):19–25. doi:10.2478/v10042-010-0015-1
Cole B, Gomoll A, Yanke A, Pylawka T, Lewis P, MacGillivray J, Williams J (2007) Biocompatibility of a polymer patch for rotator cuff repair. Knee Surg Sports Traumatol Arthrosc 15(5):632–637. doi:10.1007/s00167-006-0187-6
Diller RB, Kellar RS (2014) Validating whole slide digital morphometric analysis as a microscopy tool. Microsc Microanal 1–7. doi:10.1017/S1431927614013567
Zhao S, Pinholt EM, Madsen JE, Donath K (2000) Histological evaluation of different biodegradable and non-biodegradable membranes implanted subcutaneously in rats. J Craniomaxillofac Surg 28(2):116–122
Voskerician G, Jin J, White MF, Williams CP, Rosen MJ (2010) Effect of biomaterial design criteria on the performance of surgical meshes for abdominal hernia repair: a pre-clinical evaluation in a chronic rat model. J Mater Sci Mater Med 21(6):1989–1995
Voskerician G, Gingras PH, Anderson JM (2006) Macroporous condensed poly (tetrafluoroethylene). I. In vivo inflammatory response and healing characteristics. J Biomed Mater Res A 76(2):234–242
Rosch R, Junge K, Schachtrupp A, Klinge U, Klosterhalfen B, Schumpelick V (2003) Mesh implants in hernia repair. Eur Surg Res 35(3):161–166
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Diller, R.B., Audet, R.G., Kellar, R.S. (2014). Quantitative Histopathology for Evaluation of In Vivo Biocompatibility Associated with Biomedical Implants. In: Potts, S., Eberhard, D., Wharton, Jr., K. (eds) Molecular Histopathology and Tissue Biomarkers in Drug and Diagnostic Development. Methods in Pharmacology and Toxicology. Humana Press, New York, NY. https://doi.org/10.1007/7653_2014_37
Download citation
DOI: https://doi.org/10.1007/7653_2014_37
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-2680-0
Online ISBN: 978-1-4939-2681-7
eBook Packages: Springer Protocols