Abstract
Embryonic stem cells (ESCs) differentiation via embryoid body (EB) formation is an established method that generates the three germ layers. However, EB differentiation poses several problems including formation of heterogeneous cell populations. Herein, we described a differentiation protocol on enhancing mesoderm derivation from murine ESCs (mESCs) using conditioned medium (CM) from HepG2 cells. We used this technique to direct hematopoiesis by generating “embryoid-like” colonies (ELCs) from murine (m) ESCs without following standard formation of EBs. Our CM-mESCs group yielded an almost fivefold increase in ELC formation (p ≤ 0.05) and higher expression of mesoderm genes;-Brachyury-T, Goosecoid, and Flk-1 compared with control mESCs group. Hematopoietic colony formation from CM-mESCs was also enhanced by twofold at days 7 and 14 with earlier colony commitment compared to control mESCs (p ≤ 0.05). This early clonogenic capacity was confirmed morphologically by the presence of nucleated erythrocytes and macrophages as early as day 7 in culture using standard 14-day colony-forming assay. Early expression of hematopoietic primitive (ζ-globin) and definitive (β-globin) erythroid genes and proteins was also observed by day 7 in the CM-treated culture. These data indicate that hematopoietic cells more quickly differentiate from CM-treated, compared with those using standard EB approaches, and provide an efficient bioprocess platform for erythroid-specific differentiation of ESCs.
Access provided by CONRICYT – Journals CONACYT. Download protocol PDF
Similar content being viewed by others
Keywords:
1 Introduction
Embryonic stem cells (ESCs) spontaneously differentiate in vitro through formation of embryoid bodies (EBs), tissue-like spheroids cultivated in suspension culture (1–3). EB formation recapitulates some aspects of early embryogenesis, including the formation of a complex three-dimensional arrangement wherein cell–cell and cell–matrix interactions support the development of the three embryonic germ layers—mesoderm, ectoderm, and endoderm. Following EB formation, cells are returned to adherent culture conditions in order for directed differentiation to occur (4–6). However, EB-based methodologies are imprecise since spontaneous differentiation into the three germ layers is not controlled (4, 5). Blood is derived from mesoderm in direct competition with spontaneous angiogenic and cardiomyogenic differentiation (6–9). To recapitulate the process of blood formation from mesoderm in vitro, ESCs are allowed to form EBs for *Footnote 15–8 days followed by terminal hematopoietic differentiation through the use of various supplements and cytokines (3, 6, 10). Recent efforts in in vitro red cell production starting from ESCs with a view to clinical applications have resulted in successful serum-free cultures for hematopoietic differentiation and the production of functional nucleated red blood cells (11–13). However, these methods have all used EB formation and coculture with feeder cells or conditioned media (CM). Non-EB cultures of hematopoietic cells derived from ESCs can also be achieved with coculture using stromal cells (11, 14–16). Although coculture provides microenvironmental cues for differentiation of ESCs, the method is labor-intense and problematic in the separation of feeders from differentiated cells and the possible formation of heterokaryons within the culture (17). Recombinant cytokines are used for more defined culture conditions for the controlled differentiation of mesoderm from ESCs. However, the method is costly and can, paradoxically, lead to multilineage development due to the redundant action of cytokines and morphogens, such as BMP-4 and Activin-A (5, 18). Alternatively, CM from the culture of differentiated somatic cells contain soluble factors, although largely undefined, that direct ESC differentiation into layers such as mesoderm in vitro and are less resource-intensive, making it a practical alternative for the study of mesoderm derivatives in vitro (5, 17, 19–21). We have previously demonstrated that CM, obtained from cultures of the human hepatocarcinoma cell line HepG2, enhanced mesoderm formation from murine ESCs (mESCs) without EB formation (5). HepG2 cells have characteristics similar to those of visceral endoderm, an early organizer during embryogenesis that secret signaling molecules specifying cell fate (19, 22, 23). The similarities between liver carcinoma cell lines and visceral endoderm have led to the suggestion that HepG2 CM reiterate visceral endoderm-like signaling in this system (5, 19). Differentiation of mESCs cultured in HepG2 CM resulted in a restricted repertoire of cell types comprised of mesoderm and parietal endoderm providing an efficient method for enriched mesoderm formation in vitro (5, 24). Herein, we extend our studies of directed mesoderm differentiation from mESCs using HepG2 CM and present an efficient method for the in vitro development of the hematopoietic lineage, as an alternative process for potential study and application of in vitro erythropoiesis from ESCs.
2 Materials
2.1 Tissue Culture
-
1.
Undifferentiated E14 Tg2α murine ES and Hepatocarcinoma (HepG2) cell lines (ATCC).
-
2.
Culture flask with tissue culture-treated low-toxin (endotoxin <0.5 endotoxin units per milliliter [EU/mL]) high-grade polystyrene culture surfaces (Nunc; T-75 flasks)
-
3.
35 × 10 mm culture petri dishes (Corning nontreated petri dishes [available through Sigma-Aldrich, UK]).
-
4.
Blunt-end needles (Stem Cell Technologies, UK).
-
5.
Leukemia inhibitory factor (LIF; ESGRO) (Chemicon [available through Milipore, UK]).
-
6.
Gelatine solution type B from bovine skin 2 % (Sigma-Aldrich, UK). Diluted the concentrated solution with tissue culture-grade phosphate-buffered saline (PBS) to 0.1 %. For example, increase volume of 1 mL concentrated solution to 20 mL by tissue culture-grade PBS.
-
7.
Dimethyl sulfoxide Hybri-Max (DMSO) (Sigma Aldrich, UK).
-
8.
1× tissue culture-grade PBS without calcium or magnesium (Gibco Invitrogen Life Technologies, UK).
-
9.
High-glucose Dulbecco’s modified Eagle’s medium (DMEM) (1×) contains 4,500 mg/L glucose, pyridoxine hydrochloride and l-glutamine, but no sodium pyruvate (Gibco Invitrogen Life Technologies, UK).
-
10.
Heat-inactivated fetal bovine serum (FBS) characterized and screened for ESCs growth (batch tested) (Gibco Invitrogen Life Technologies, UK).
-
11.
200 mM l-glutamine (Gibco Invitrogen Life Technologies, UK).
-
12.
Tripsin-ethylenediaminetetraacetic acid (EDTA) solution: 0.05 and 0.25 % trypsin/0.53 mM EDTA in 0.1 M PBS without calcium or magnesium (Gibco Invitorgen Life Technologies, UK).
-
13.
100 units/mL penicillin and 100 μg/mL streptomycin (Gibco Invitrogen Life Technologies, UK).
-
14.
Water for embryo transfer, sterile-filtered, BioXtra (Sigma-Aldrich, UK).
-
15.
14.3 M mercaptoethanol 2-(ME) (Sigma-Aldrich); dilute in the solution into 100 mM by adding 40 mL concentrated solution into 4,960 mL water for embryo transfer.
-
16.
Iscove’s Modified Dulbecco’s Medium (IMDM) with high glucose and sodium pyruvate (Gibco Invitrogen Life Technologies, UK).
-
17.
0.1 mM Nonessential Amino Acids (Sigma-Aldrich, UK).
-
18.
100 mM monothioglycerol (MTG) (Sigma-Aldrich, UK).
-
19.
1% BIT (1 % BSA, 10 mg/mL Insulin, and 200 mg/mL Transferrin; Stem Cell Technologies, UK).
-
20.
Growth factors: mouse stem cell factor (mSCF), mouse interleukin (mIL)-3, human (h) IL-6, and human erythropoietin (hEPO; all supplied by R&D Systems, UK).
-
21.
Es-Cult M3120 medium (Stem Cell Technologies, UK).
-
22.
15- and 50-mL polypropylene conical centrifuge tubes (VWR International, UK).
-
23.
Cryogenic vials (VWR International, UK).
-
24.
Ethanol 96–99 % (absolute) GPR (VWR International, UK).
-
25.
Virkon (VWR International, UK).
-
26.
100 % isopropyl alcohol bath (Nalgene Mr. Frosty, UK).
2.1.1 Media
-
1.
mESCs maintenance medium : High-glucose DMEM supplemented with 10 % v/v FBS, 2 mM l-glutamine, 100 units/mL penicillin and 100 μg/mL streptomycin, 0.1 mM 2-ME and 0.1 mM LIF immediately before use. For example, add 50 mL FBS, 5 mL l-glutamine, 5 mL penicillin and streptomycin to 440 mL high-glucose DMEM. Add 1 μL 100 mM 2-ME per milliliter and 1 μL 1,000 U LIF per milliliter of the culture medium immediately before use (see Note 1).
-
2.
HepG2 maintenance medium: High glucose DMEM without sodium pyruvate supplemented with 10 % FBS, 2 mM l-glutamine and 100 units/mL penicillin and 100 μg/mL streptomycin.
-
3.
Primary differentiation medium: High glucose IMDM with sodium pyruvate supplemented with 1 % Es-Cult, 15 % FBS, 2 mM l-glutamine, 150 μM MTG, and 40 ng/mL mSCF.
-
4.
Feeding medium: Primary differentiation medium supplemented with 160 ng/mL mSCF, 30 ng/mL mIL-3, 20 ng/mL h IL-6, and 3 U/mL hEPO.
-
5.
Hematopoietic differentiation medium: High glucose IMDM with sodium pyruvate supplemented with 15 % FBS, 2 mM l-Glutamine, 150 mM MTG, 1 % BIT, 150 ng/mL mSCF, 30 ng/mL mIL-3, 30 ng/mL hIL-6, 3 U/mL hEPO, and 1 % Es-Cult medium.
2.1.2 General Cell Maintenance Procedure
The following maintenance is applicable to many cell types and cell culture media.
-
1.
Maintain the cells in an incubator at 37 °C in a humidified atmosphere of 5 % CO2/95 % air.
-
2.
Control the water level in the incubator’s tank daily.
-
3.
Check cells daily under inverted microscope. If any contamination is observed in the cultured cells, then take the culture plate out the incubator and apply 10 % virkon for approximately 1–2 h and then discard the plate.
-
4.
After using a tissue culture hood, wipe it with 1 % virkon and aspirate some 10 % virkon for decontamination of tube and contents of canister.
-
5.
Discard content of canister after 1–2 h treatment with virkon.
2.2 Reverse Transcriptase Polymerase Chain Reaction
-
1.
Reverse transcriptase polymerase chain reaction (RT-PCR)-grade water (Ambion, UK).
-
2.
Total RNA Isolation kit, RNeasy mini kit (Qiagen, UK). This kit contains the following ready-made buffers and columns:
-
(a)
Lysis buffer: Buffer RLT, add 10 μL of β-ME for working solution.
-
(b)
Buffer RW1.
-
(c)
Washing buffer: Buffer RPE, add 4 volumes of absolute ethanol (96–99 %) for working solution.
-
(d)
RNase-free water.
-
(e)
Qiashredder spin column.
-
(f)
RNeasy spin column.
-
(a)
-
3.
Tissue culture-grade 1× PBS without calcium or magnesium (Gibco Invitrogen Life Technologies, UK).
-
4.
RNase-, DNA-, adenosine triphosphate (ATP)-, and pyrogen-free 1.5 mL Eppendorf tube (VWR International, UK).
-
5.
Cuvette (Eppendorf, UK).
-
6.
Spectrophotometer (Eppendorf Biophotometer, Germany).
-
7.
TE Buffer at pH 7.0 (Ambion, UK).
-
8.
Molecular biology-grade ethanol (VWR International, UK).
-
9.
Reverse Transcription (RT) system kit (Promega, UK). This kit contains the following reagents:
-
(a)
MgCl2, 25 mM.
-
(b)
Reverse Transcription 10× Buffer.
-
(c)
dNTP Mixture, 10 mM.
-
(d)
Recombinant RNasin® Ribonuclease Inhibitor.
-
(e)
AMV Reverse Transcriptase (High Concentration).
-
(f)
Random Primers.
-
(g)
Nuclease-Free Water.
-
(a)
-
10.
Mastermix PCR reagents contains (all by Invitrogen, UK):
-
(a)
1× PCR Buffer.
-
(b)
0.2 mM dNTP mixture.
-
(c)
1.5 mM MgCl2.
-
(d)
0.2 μM each primer (forward and reverse).
-
(e)
1 unit of Platinum Taq DNA Polymerase.
-
(f)
DNase free water.
-
(a)
-
11.
Agarose powder (Invitrogen, UK).
-
12.
10 μg/mL Ethidium bromide (Sigma-Aldrich, UK).
-
13.
Gel-documentation system with ultra-violet light (Gene Flash, Syngene Bio Imaging).
-
14.
-
(a)
Primers for endoderm:
Gata-4 [glutamyl-tRNA(Gln) amidotransferase subunit A-1] (571-bp amplicon size):
-
Forward: CTCCTACTCCAG CCCCTACC
-
Reverse: GTGGCATTGCTGGAGTTACC
α-feto protein [alpha-1-fetoprotein] (299-bp amplicon size):
-
Forward: CCTTGGCTGCTCAGTACGACAAGG
-
Reverse: CCTGCAGACACTCCAGCGAGTTTC
-
-
(b)
Primers for mesoderm:
Brac-T [brachyury transcription factor T-gene] (388-bp amplicon size):
-
Forward: AAGGAACCACCGGTCATCG
-
Reverse: CGTGTGCGTCAGTGGTGTGTAATG
Flk-1 [fetal liver kinase-1] (599-bp amplicon size):
-
Forward: CCTGGTCAAACAGCTCATCA
-
Reverse: AAGCGTCTGCCTCAATCACT
GSC [goosecoid] (516-bp amplicon size):
-
Forward: ATGCTGCCCTACATGAACGT
-
Reverse: CAGTCCTGGGCCTGTACATT
-
-
(c)
Primers for ectoderm:
Sox-1 [sex determining region Y-box1] (388-bp amplicon size):
-
Forward: GCACACAGCGTTTTCTCGG
-
Reverse: ACATCCGACTCCTCTTCCC
Nestin [neuroepithelial stem cell specific protein] (259-bp amplicon size):
-
Forward: CGGCCCACGCATCCCCCATCC
-
Reverse: AGCGGCCTTCCAATCTCTGTTCC
-
-
(d)
Primers for hematopoietic progenitors:
Gata-2 [glutamyl-tRNA(Gln) amidotransferase subunit A-1] (336-bp amplicon size):
-
Forward: TGCAACACACCACCCGATACC
-
Reverse: CAATTTGGACAACAGGTGCCC
c-kit [cellular-homolog of protein kinase transmembrane receptor] (458-bp amplicon size):
-
Forward: GCTCATAAATGGCATGCTCCAGTGT
-
Reverse: GAAGTTGCGTCGGGTCTATGTAAAC
-
-
(e)
Primers for myeloid-erythroid:
c-myb [cellular-myeloblastosis oncogene] (522-bp amplicon size):
-
Forward: GAGCTTGTCCAGAAATATGGTCCTAAG
-
Reverse: GGCTGCCGCAGCCGGCTGAGGGAC
SCL [stem cell leukemia/T-cell acute lymphocytic protein 1] (277-bp amplicon size):
-
Forward: TAGCCTTAGCCAGCCGCTCG
-
Reverse: GCGGAGGATCTCATTCTTGC
-
-
(f)
Primers for erythroid:
Gata-1 [glutamyl-tRNA(Gln) amidotransferase subunit A-1] (581-bp amplicon size):
-
Forward: ATGCCTGTAATCCCAGCACT
-
Reverse: TCATGGTGGTAGCTGGTAGC
βH1-globin [hemoglobin beta-H1 chain] (610-bp amplicon size):
-
Forward: TTTGCACACTTGAGATCATCTC
-
Reverse: GGTCTCCTTGAGGTTGTTCCATG
β-major globin [beta-major globin] (610-bp amplicon size):
-
Forward: ATGGTGCACCTGACTGATGCTG
-
Reverse: GGTTTAGTGGTACTTGTGAGCC
-
-
(g)
Housekeeping:
GAPDH [gene glyceraldehyde-3-phosphate dehydrogenase] (500-bp amplicon size):
-
Forward primer: CATCACCATCTTCCAGGAGC
-
Reverse primer: ATGCCAGTGAGCTTCCCGTC
-
-
(a)
-
15.
Thermal cycler system (G-Storm, UK).
2.3 Western Blot Analysis
-
1.
RIPA lysis buffer (Santa Cruz Biotechnology, Germany).
-
2.
Protein Quantification: BCA™ Assay Kit (Pierce Biotechnology, UK).
-
3.
Bovine serum albumin (BSA; Sigma-Aldrich, UK).
-
4.
Tissue culture-grade 1× PBS without calcium or magnesium (Gibco Invitrogen Life Technologies, UK).
-
5.
96 well plate (VWR International, UK).
-
6.
Enzyme-linked immunosorbent assay (ELISA) reader (ELx808, BIO-TEK, USA).
-
7.
Laemmli sample buffer (contain 0.125 M Tris–HCL at pH 6.8, 4 % SDS, 20 % glycerol, 10 % βME, and 0.005 % bromophenol blue; BioRad, UK).
-
8.
Precision Plus Protein™ All Blue Standards (BioRad, UK).
-
9.
10× Tris/Glycine premixed electrophoresis buffer (BioRad, UK).
-
10.
NuPAGE® Novex® 4–12 % Bis-Tris Protein Gels, 1.0 mm, 10 well (Invitrogen, UK).
-
11.
0.2 μm nitrocellulose membrane (BioRad, UK).
-
12.
C140 Mini Blot Module (Thermo Electron).
-
13.
EC120 mini vertical gel electrophoresis unit (Thermo Electron).
-
14.
SuperSignal West Pico chemiluminescent substrate (Pierce Biotechnology, UK).
-
15.
Dyversity gel-documentation system (SynGene) using the Genesnap program (SynGene).
-
16.
Rabbit polyclonal antibodies, GAPDH (1:20,000; Abcam, UK) and Gata-1 (1:200; Abcam, UK).
-
17.
Goat anti-rabbit polyclonal IgG Horseradish peroxidase (HRP, 1:10,000; Abcam, UK).
-
18.
5 % nonfat skim milk solution.
2.4 Wright-Giemsa Analysis
-
1.
Cytospin cyto-centrifudge set (Hettich Rotina 46R Centrifuge, Germany).
-
2.
Wright-Giemsa stain (Sigma-Aldrich, UK).
-
3.
Coplin staining jars (Sigma-Aldrich, UK).
-
4.
Olympus BX51 microscope and DP50 camera (Olympus UK Ltd.).
-
5.
Microscope slides (Thermo Scientific, UK).
3 Methods
3.1 Tissue Culture
3.1.1 Gelatin Coating of Culture Surface
-
1.
Add adequate amount of 0.1 % gelatin solution to the culture surface to cover the entire surface of T75 flask and leave in a 37 °C incubator for at least 1 h.
-
2.
Aspirate the gelatin solution before use and add the cell suspension.
3.1.2 Thawing Cells
-
1.
Prepare the culture flask with gelatin-coated; prepare culture medium (supplemented high-glucose DMEM) and prewarmed it in 37 °C water bath.
-
2.
Remove the frozen cell vial from the liquid nitrogen, submerge the vial in the 37 °C water bath, and check for thawing.
-
3.
Remove the vial just before the final ice crystal disappears and wipe the outside of the vial with 70 % ethanol.
-
4.
Transfer cells to a centrifuge tube and add 9 mL medium drop-wise, swirling constantly.
-
5.
Centrifuge the cells for 5 min at 200 × g (see Note 2).
-
6.
Aspirate medium from the pellet, resuspend cells in the medium, and apply to the gelatin-coated plate.
3.1.3 Passaging Cells (Trypsinization)
-
1.
Feed cells about 1 h before passaging.
-
2.
Warm an aliquoted amount of Trypsin-EDTA 0.05 % (for mESCs culture) and 0.25 % (for HepG2 culture), and culture medium.
-
3.
Aspirate medium from cultured cells and wash three times with 1× PBS.
-
4.
Add Trypsin-EDTA solution and put cells in a 37 °C incubator for 5–10 min.
-
5.
Gently pipet the cells up and down to separate them into single cells.
-
6.
Transfer the cells into a centrifuge tube and add culture medium to the amount of passaging medium (see Note 3).
-
7.
Centrifuge the cells for 5 min at 200 × g.
-
8.
Remove the medium. Resuspend and plate out the cells required.
3.1.4 Freezing Cells
This freezing protocol is applicable to both mESCs and HepG2 cell lines.
-
1.
Put freezing medium on ice.
-
2.
Trypsinize the cells as described in Section 3.1.3.
-
3.
Add culture medium to the cell-trypsin medium and centrifuge the cells at 200 × g for 5 min.
-
4.
Remove the medium and resuspend cells in an appropriate volume of medium.
-
5.
Count the cells and centrifuge as before.
-
6.
Remove the medium and add an appropriate volume of the freezing medium so that the cell concentration is between 2 × 106 and 5 × 106 per milliliter of suspension.
-
7.
Aliquot suspension into 1 mL cryovials, transfer the vials into a 100 % isopropyl alcohol bath, and leave the bath in the −80 °C freezer overnight. Alternatively, put them in a −20 °C freezer for 1 h, then transfer to a −80 °C freezer overnight.
-
8.
Transfer to liquid nitrogen the next day.
3.1.5 Culture of HepG2 Cells and Preparation of HepG2 Conditioned Medium (CM)
-
1.
Thaw a frozen cell vial (see Section 3.1.4) and resuspend cell in HepG2 cell medium (see Section 2.1.1).
-
2.
Seed onto a non-gelatin-coated tissue culture flask.
-
3.
Change the medium every 2 days and passage the cells every 3 or 4 days with 0.25 % Trypsin–EDTA (see Section 3.1.3).
-
4.
For CM preparation, seed a density of 5.0 × 104 cell/cm2 HepG2 cells on tissue culture flasks and culture the cells at 370 C in a 5 % CO2 humidified water-based incubator for the following 4 days without medium changing.
-
5.
Collect the culture medium after 4 days, filter-sterilized the medium using 0.22 μm filter unit (VWR International, UK).
-
6.
Supplement the filtered medium with 0.1 mM β-ME and store the medium at −20 °C.
-
7.
Prior to culture use, mix the medium collected from HepG2 cells as explained with fresh mESCs maintenance medium with a ratio of 1:1, and add 1,000 U/mL of LIF in the mixture (19) (see Note 4).
3.1.6 Culture of mESCs
3.1.6.1 Culture of Undifferentiated Cells
-
1.
Seed undifferentiated mESCs on gelatin-coated flask and change the medium every other day.
-
2.
When a subconfluent flask of undifferentiated mESCs is obtained, passage the cells by trypsinization (see Section 3.1.3) and plate the cells on gelatin-coated flask (Fig. 1).
Fig. 1 Experimental design: control murine embryonic stem cell (mESC) and conditioned medium (CM)-mESC groups. The control group consisted of normal mESC culture, exposed to standard Iscove’s modified Dulbecco’s medium (IMDM) prior to differentiation that had undergone an 8-day embryoid-like colony (ELC) formation period using primary differentiation medium followed by a further 14 days in the secondary hematopoietic differentiation medium, as previously described (25). The experimental CM-mESC group consisted of mESCs previously exposed to HepG2 CM prior to differentiation, followed by the same 8-day ELC formation period using primary differentiation medium and a further 14 days of culture in the secondary hematopoietic differentiation medium, as explained previously. Both culture groups were analyzed simultaneously
3.1.6.2 Culture of Predifferentiated Cells with HepG2-CM (“CM-mESCs”)
-
1.
Passage the subconfluent flask of undifferentiated mESC, count and plate 3.0 × 106 cells per T-75 gelatin-coated flask.
-
2.
Culture the cells in HepG2-CM (see Section 3.1.5) and change the medium every other day by adding 1,000 U/mL LIF (ESGRO) for 3 days.
3.1.6.3 Formation of “Embryoid-Like Colonies (ELCs)” in Primary Differentiation
-
1.
Trypsinize the “CM-mESCs” and seed 3 × 103 cells/mL on low adherence 35-mm Petri-dishes supplemented with primary differentiation medium (see Section 2.1.1) (see Note 5).
-
2.
On day 7 of culture, feed the cells with feeding medium (see Section 2.1.1) and culture for an additional day (day 8 of culture).
3.1.6.4 Hematopoietic Colony Forming Unit (CFU) Assay
-
1.
Disrupt 8-day-old ELCs and replate 2.0 × 105 cells/mL on low adherence 35-mm Petri-dishes supplemented with hematopoietic differentiation medium (see Section 2.1.1) (see Note 6).
-
2.
Incubate the cells for an additional 14 days. Score the resultant colonies using standard criteria based on the manufacturer’s instruction (Stem Cell Technologies Hematopoietic Manual).
Figure 1 shows a schematic diagram of experimental design for conditioned medium mesodermal enhancement.
3.2 Reverse Transcriptase Polymerase Chain Reaction
3.2.1 Total RNA Isolation and Quantification
Total RNA isolations are adopted from Qiagen’s instruction for Rneasy Kit product.
-
1.
Harvest the cells by trypsinization as described in Section 3.1.3, neutralize the trypsin with serum-containing medium, and count the cells.
-
2.
Centrifuge and discard the supernatant.
-
3.
Wash the pellet with the ice-cold PBS and aliquot the cells into <5 × 106.
-
4.
Centrifuge and discard the supernatant.
-
5.
Add 350 mL lysis buffer RLT for each 5 × 106 aliquot.
-
6.
Mix by pipetting or vortexing (see Note 7).
-
7.
Homogenize the cell lysate in a Qiashredder spin column and centrifuge at 14,000–16,000 × g for 2 min.
-
8.
Add 350 mL 70 % ethanol into the cell lysate through the column.
-
9.
Wash the total RNA that bound on the membrane column twice with buffer RW1 before ethanol removed using buffer RPE.
-
10.
Elute the RNA in a 1.5 mL collection tube using RNase-free water.
-
11.
Quantitate the RNA by spectrophotometry (see Note 8).
-
12.
Store the RNA at −80 °C after quantification.
3.2.2 Complimentary DNA (cDNA) Formation by Reverse Transcriptase (RT)
Total mRNA is reversed transcribed into CDNA following Promega’s instruction using Reverse Transcription System Kit.
-
1.
Add 1 mg RNA sample in a PCR tube and adjust the volume to 10 mL by RNase-free water.
-
2.
Heat the RNA sample at 70 °C for 10 min, briefly centrifuge and cool on ice for 5 min.
-
3.
Prepare the master-mix by adding 5 mm MgCl2, 1 mM of 2′-deoxynucleoside 5′-triphosphate (dNTP) mixture, 1 unit/μL recombinant RNasin ribonuclease inhibitor, 5 units/μL AMV reverse transcriptase and 0.5 mg/μL random primers.
-
4.
Add the master mix (10 μL) to the RNA sample.
-
5.
Incubate at 25 °C for 10 min, and then reverse transcriptase is performed at 42 °C for 40 min followed by denaturation at 90 °C for 5 min and a cooling step at 4 °C for 5 min.
3.2.3 Polymerase Chain Reaction (PCR) Formation
The data presented here are adopted from Invitrogen’s protocol for the use of Taq polymerase and RT-PCR reagents.
-
1.
Prepare an amount of 50 mL containing cDNA samples with mastermix (1× PCR Buffer, 0.2 mM dNTP mixture, 1.5 mM MgCl2, 0.2 μM each primer (forward and reverse), 1 unit of Platinum Taq DNA Polymerase and DNase free water.
-
2.
Adjust the temperature profile as follows: Taq polymerase activation, 95 °C for 2 min; denaturation, 94 °C for 30 s; annealing temperature (AT)for 30 s, 72 °C for 60 s; followed by final elongation step at 72 °C for 5 min.
3.2.4 Gel Electrophoresis
-
1.
Mix 1.5 g of agarose agar powder with 100 mL of 1× TBE solution, dissolve in a microwave.
-
2.
Add 10 μg/mL of EtBr into the mix solution before being casted to solidify.
-
3.
Transfer the casted solidified gel to electrophoresis system filled with 1× TBE buffer.
-
4.
Mix 5 μL of 100 bp DNA ladder and 1 μL of Blue/Orange loading dye used as DNA reference.
-
5.
Load the DNA reference mixture into one well of the casted solidified gel.
-
6.
Mix 10 μL of PCR sample and 2 μL of Blue/Orange loading dye. Load each sample mixture into each well of the gel.
-
7.
Run electrophoresis at 95 V for 50 min.
-
8.
Visualize the gel in a gel documentation system with ultra-violet light (Gene Flash, Syngene Bio Imaging).
The result of this experiment showed a high expression of mesodermal marker gene, Flk-1 in CM-mESCs than observed in control mESCs at day 8 of culture. Figure 2 shows the DNA gel electrophoresis of the PCR products.
Formation of ELCs in both culture groups at day 8. (a) Shown are examples of (i) control and (ii) CM-mESC ELCs. Images were captured at 20 · magnification under light microscopy (scales for each image: 200 mm). (b) A higher number of ELCs formed was observed in the CM-mESC group on day 8. Data shown are mean number of colonies—SD (n = 5; **p < 0.01). (c) RT-PCR analysis of genes indicative of the three germ layers at day 8 of culture in control mESCs and CM-ESCs. Expression of genes of all three germ layers was observed in ELCs from control mESCs, whereas a higher expression of Flk-1 and loss of endoderm gene expression (GATA-4 and AFP) indicated more pronounced differentiation toward mesoderm with some early differentiation toward the hematopoietic lineage (Gata-2 and C-Kit) in those of CM-mESCs. Negative controls consisted of samples without cDNA. AFP a-fetoprotein, Flk-1 fetal-liver kinase-1, Bra-T brachyury-T, GS goosecoid, Sox-1 sry-related HMG box-1, GAPDH glyceraldehyde 3-phosphate dehydrogenase
3.3 Western Blot Analysis
3.3.1 Protein Extraction and Quantification
-
1.
Harvest cell by scraping.
-
2.
Lyse cell in 150 μL of RIPA lysis buffer, incubate at 4 °C for 30 min and centrifuge at 10,000 × g for 5 min.
-
3.
Collect the protein-containing supernatant for quantification using BCA™ Assay Kit.
-
4.
Dilute BSA in PBS from 0 to 500 μg/mL to create a protein standard curve.
-
5.
Load 25 μL of sample into 96 well plate followed by mixing with 200 μL of BCA working reagent.
-
6.
Incubate the plate in orbital shaking incubator at 37 °C for 30 min.
-
7.
Take the reading at a wavelength of 560 nm on an ELISA microplate reader.
-
8.
Quantify each protein sample against the BSA protein standard curve.
3.3.2 Sample Preparation and SDS-PAGE
-
1.
Mix each protein sample with Laemli sample buffer, heat at 95 °C for 10 min and place on ice prior for gel loading.
-
2.
Load 40 mg of protein mixed on NuPAGE® Novex® 4–12 % Bis-Tris Protein Gel electrophoresis.
-
3.
Load protein ladder Precision Plus Protein™ All Blue Standards in one well.
-
4.
Run one-dimensional SDS-PAGE to separate protein in EC120 mini vertical gel electrophoresis unit filled with 1× diluted Tris/Glycine premixed electrophoresis buffer.
-
5.
Apply a constant voltage of 110 V at initial stage for 15 min or till the sample passes the stacking gel before a constant voltage of 150 V is apply for another 45 min or until the dye reached the bottom of the gel.
-
6.
Transfer the protein blot onto 0.2 μm nitrocellulose membranes with 1× transfer buffer for 30 min followed by protein blotting on C140 Mini Blot Module at a constant current of 30 mA for 50 min (see Note 9).
3.3.3 Western Blot Analysis
-
1.
Block nonspecific antibody binding by incubation with 5 % milk in 0.1 M PBS for 1 h.
-
2.
Incubate blots at room temperature with primary and secondary antibodies for 1 h respectively.
-
3.
Detect primary antibody binding with secondary antibody goat anti-rabbit polyclonal IgG Horseradish peroxidase by the SuperSignal West Pico chemiluminescent substrate.
-
4.
Capture image using the Dyversity gel documentation system (SynGene) using the Gene snap program (SynGene).
3.4 Wright-Giemsa Cytospin Staining
-
1.
Prepare 1 × 106 cells/mL cell suspension in 100 μL PBS and load it into a well of cytospin centrifuge set containing slide.
-
2.
Spin the cells for 3 min at 100 × g.
-
3.
Air-dry the slide for 5–10 min before stained with Wright-Giemsa stain in Coplin jar for 5–10 s.
-
4.
Wash slide by dipping in deionised water for 2 min.
-
5.
View image on the Olympus BX51 Microscope and capture using the DP50 camera.
The result of this experiment showed an early erythroid maturation identified by nucleated erythrocytes and macrophages found in the CFUs from CM-mESCs at day 7. This early erythroid maturation of adult type at day 7 of CM-mESC culture was confirmed by detection of Gata1 and β-major globin proteins with a lower expression of the primitive erythroid marker ζ-globin. Figure 3 shows Wright-Giemsa staining and the protein blotting of the western blot products.
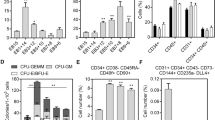
Clonogenic capacity and gene and protein expression of terminally differentiated mESCs at days 7 and 14. (a) Standard methylcellulose assay was used to determine hematopoietic clonogenic capacity for burst-forming unit erythroid (BFU-E), colony forming unit (CFU)-E, CFU-GM, and CFU-GEMM at days 7 and 14 from ELCs of CM-mESCs and control mESCs. Morphological changes observed in the CM-ESC group at day 14 of culture were consistent with loss of hemoglobin within the colonies as indicated by the reduction of reddish hue. Images were captured at 20× magnification under light microscopy (scales for each image: 200 mm). (b) Summary bar graph of CFU numbers from both mESC groups at days 7 and 14 of the terminal differentiation culture. A higher number of all hematopoietic CFUs was observed in the CM-mESC group. (c) Wright-Giemsa staining of hand-picked BFU-E and CFU-GM colonies at day 7 of cultivated CM-mESCs confirmed the early maturation of hematopoietic colonies. Images were captured at 20× magnification under light microscopy (scales for each image: 200 mm). Data shown are mean number of colonies—SD (n = 5; **p < 0.01). nE nucleated erythroid cells, Mac macrophage. (d) RT-PCR analysis of myeloid-erythroid genes in cells from day 7 CFU cultures in control mESCs and CM-mESCs indicates early expression of hematopoietic genes only in the experimental condition. Incubation for the standard 14 days resulted in expression of all hematopoietic and myeloid-erythroid markers in both control and experimental groups with loss of expression of the primitive erythroid marker bH1 globin. The negative control consisted of samples without cDNA. (e) Western blot analysis of CFUs from days 7 and 14 confirms early erythroid maturation at day 7 in the CM-mESC experimental group with primitive erythroid expression of z-globin protein. The negative control consisted of samples without protein extract
4 Notes
-
1.
2-ME loses it thiol (-SH) group in the presence of metallic salts within 1–2 days Therefore, 2-ME must be added fresh to the culture medium immediately before use.
-
2.
The duration and centrifugal force may differ for different cell lines, but generally 5 min at 200 × g is suitable.
-
3.
The serum in the culture medium inhibits trypsin.
-
4.
HepG2 conditioned medium (CM) was formulated by mixing 50 % of fresh mESCs growth medium with 50 % of the medium collected from HepG2 cells and subsequently with addition of 1,000 units/mL of LIF in the mixture.
-
5.
The cell density of the suspension will be cell line dependent and will vary on their ability to differentiate in methylcellulose. Optimally, there will be 50–100 colonies per dish in 1 mL of methylcellulose. As a first step, it may be necessary to perform a dose curve to determine the number of cells required to yield the optimal number of EBs. The number of EBs obtained should be linear with ESCs input.
-
6.
The actual number plated will vary depending on the cell line and conditions used, as well the age of the colonies, but the density of 1–5 × 104 cells per dish should provide a useful starting range. When first establishing optimal plating densities, it is advisable to try two different cell concentrations which differ by two- to threefold.
-
7.
In the total RNA isolation protocol, the samples may be stored at −20 or −80 °C after homogenization in lysis buffer.
-
8.
Find the OD at A 260 and A 280. The A 260:A 280 should between 1.6 and 2.
-
9.
The blotted membrane was air-dried for at least 1 h at room temperature prior to immunostaining.
Notes
- 1.
*Keller et al. 1993 has reported that ESCs efficiently undergo differentiation in vitro to mesoderm and hematopoietic cells that this in vitro system recapitulates day 6.5 to 7.5 of mouse hematopoietic development. Embryonic stem cells differentiated as embryoid bodies (EBs) develop erythroid precursors by day 4 of differentiations, and by day 6, more than 85% of EBs contain such cells. The number of the EPO responsive precursors increased by day 8 of differentiation, them remained constant, and finally began to decline by day by day 12. Therefore, we hypothesized that day 5-8 are the period of mesoderm differentation in EB to recapitulate embryonic development based on the previous study reported.
Reference ammended: 3(a). Irion S, Clarke RL, Luche H, Kim I, Morrison SJ, Fehling HJ, Keller GM (2010) Temporal specification of blood progenitors from mouse embryonic stem cells and induced pluripotent stem cells. Development 137(17):2829–2839. doi:10.1242/dev.0421193 (b). Keller GM, Kennedy M, Papayannopoulou T, Wiles MV (1993) Hemataopoietic commitment during embryonic stem cell differentiation in culture. Mol and Cell Biol: 473–486.
References
Evans MJ, Kaufman MH (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292(5819):154–156
Kennedy M, Firpo M, Choi K, Wall C, Robertson S, Kabrun N, Keller G (1997) A common precursor for primitive erythropoiesis and definitive haematopoiesis. Nature 386(6624):488–493. doi:10.1038/386488a0
Irion S, Clarke RL, Luche H, Kim I, Morrison SJ, Fehling HJ, Keller GM (2010) Temporal specification of blood progenitors from mouse embryonic stem cells and induced pluripotent stem cells. Development 137(17):2829–2839. doi:10.1242/dev.042119
Placzek MR, Chung IM, Macedo HM, Ismail S, Mortera Blanco T, Lim M, Cha JM, Fauzi I, Kang Y, Yeo DC, Ma CY, Polak JM, Panoskaltsis N, Mantalaris A (2009) Stem cell bioprocessing: fundamentals and principles. J R Soc Interface 6(32):209–232. doi:10.1098/rsif.2008.0442
Hwang YS, Randle WL, Bielby RC, Polak JM, Mantalaris A (2006) Enhanced derivation of osteogenic cells from murine embryonic stem cells after treatment with HepG2-conditioned medium and modulation of the embryoid body formation period: application to skeletal tissue engineering. Tissue Eng 12(6):1381–1392. doi:10.1089/ten.2006.12.1381
Zhang WJ, Park C, Arentson E, Choi K (2005) Modulation of hematopoietic and endothelial cell differentiation from mouse embryonic stem cells by different culture conditions. Blood 105(1):111–114. doi:10.1182/blood-2004-04-1306
Bielinska M, Narita N, Heikinheimo M, Porter SB, Wilson DB (1996) Erythropoiesis and vasculogenesis in embryoid bodies lacking visceral yolk sac endoderm. Blood 88(10):3720–3730
Eilken HM, Nishikawa S, Schroeder T (2009) Continuous single-cell imaging of blood generation from haemogenic endothelium. Nature 457(7231):896–900. doi:10.1038/nature07760
Kouskoff V, Lacaud G, Schwantz S, Fehling HJ, Keller G (2005) Sequential development of hematopoietic and cardiac mesoderm during embryonic stem cell differentiation. Proc Natl Acad Sci U S A 102(37):13170–13175. doi:10.1073/pnas.0501672102
Pick M, Azzola L, Mossman A, Stanley EG, Elefanty AG (2007) Differentiation of human embryonic stem cells in serum-free medium reveals distinct roles for bone morphogenetic protein 4, vascular endothelial growth factor, stem cell factor, and fibroblast growth factor 2 in hematopoiesis. Stem Cells 25(9):2206–2214. doi:10.1634/stemcells.2006-0713
Lu SJ, Feng Q, Park JS, Vida L, Lee BS, Strausbauch M, Wettstein PJ, Honig GR, Lanza R (2008) Biologic properties and enucleation of red blood cells from human embryonic stem cells. Blood 112(12):4475–4484. doi:10.1182/blood-2008-05-157198
Kennedy M, D'Souza SL, Lynch-Kattman M, Schwantz S, Keller G (2007) Development of the hemangioblast defines the onset of hematopoiesis in human ES cell differentiation cultures. Blood 109(7):2679–2687. doi:10.1182/blood-2006-09-047704
Lapillonne H, Kobari L, Mazurier C, Tropel P, Giarratana MC, Zanella-Cleon I, Kiger L, Wattenhofer-Donze M, Puccio H, Hebert N, Francina A, Andreu G, Viville S, Douay L (2010) Red blood cell generation from human induced pluripotent stem cells: perspectives for transfusion medicine. Haematologica 95(10):1651–1659. doi:10.3324/haematol.2010.023556
Ma F, Ebihara Y, Umeda K, Sakai H, Hanada S, Zhang H, Zaike Y, Tsuchida E, Nakahata T, Nakauchi H, Tsuji K (2008) Generation of functional erythrocytes from human embryonic stem cell-derived definitive hematopoiesis. Proc Natl Acad Sci U S A 105(35):13087–13092. doi:10.1073/pnas.0802220105
Hiroyama T, Miharada K, Sudo K, Danjo I, Aoki N, Nakamura Y (2008) Establishment of mouse embryonic stem cell-derived erythroid progenitor cell lines able to produce functional red blood cells. PLoS One 3(2):e1544. doi:10.1371/journal.pone.0001544
Ji J, Vijayaragavan K, Bosse M, Menendez P, Weisel K, Bhatia M (2008) OP9 stroma augments survival of hematopoietic precursors and progenitors during hematopoietic differentiation from human embryonic stem cells. Stem Cells 26(10):2485–2495. doi:10.1634/stemcells.2008-0642
Heng BC, Cao T, Haider HK, Wang DZ, Sim EK, Ng SC (2004) An overview and synopsis of techniques for directing stem cell differentiation in vitro. Cell Tissue Res 315(3):291–303. doi:10.1007/s00441-003-0847-5
Willems E, Leyns L (2008) Patterning of mouse embryonic stem cell-derived pan-mesoderm by Activin A/Nodal and Bmp4 signaling requires Fibroblast Growth Factor activity. Differentiation 76(7):745–759. doi:10.1111/j.1432-0436.2007.00257.x
Rathjen J, Lake JA, Bettess MD, Washington JM, Chapman G, Rathjen PD (1999) Formation of a primitive ectoderm like cell population, EPL cells, from ES cells in response to biologically derived factors. J Cell Sci 112(Pt 5):601–612
Rathjen J, Haines BP, Hudson KM, Nesci A, Dunn S, Rathjen PD (2002) Directed differentiation of pluripotent cells to neural lineages: homogeneous formation and differentiation of a neurectoderm population. Development 129(11):2649–2661
Mummery C, Ward-van Oostwaard D, Doevendans P, Spijker R, van den Brink S, Hassink R, van der Heyden M, Opthof T, Pera M, de la Riviere AB, Passier R, Tertoolen L (2003) Differentiation of human embryonic stem cells to cardiomyocytes: role of coculture with visceral endoderm-like cells. Circulation 107(21):2733–2740. doi:10.1161/01.CIR.0000068356.38592.68
Rodda SJ, Kavanagh SJ, Rathjen J, Rathjen PD (2002) Embryonic stem cell differentiation and the analysis of mammalian development. Int J Dev Biol 46(4):449–458
Rathjen J, Rathjen PD (2001) Mouse ES cells: experimental exploitation of pluripotent differentiation potential. Curr Opin Genet Dev 11(5):587–594
Kang Y, Nagy JM, Polak JM, Mantalaris A (2009) Proteomic characterization of the conditioned media produced by the visceral endoderm-like cell lines HepG2 and END2: toward a defined medium for the osteogenic/chondrogenic differentiation of embryonic stem cells. Stem Cells Dev 18(1):77–91. doi:10.1089/scd.2008.0026
Keller GM, Webb S, Kennedy M (2002) Hematopoietic development of ES cells in culture. Methods Mol Med 63:209–230
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Fauzi, I., Panoskaltsis, N., Mantalaris, A. (2015). In Vitro Differentiation of Embryonic Stem Cells into Hematopoietic Lineage: Towards Erythroid Progenitor’s Production. In: Turksen, K. (eds) Embryonic Stem Cell Protocols. Methods in Molecular Biology, vol 1341. Humana Press, New York, NY. https://doi.org/10.1007/7651_2015_218
Download citation
DOI: https://doi.org/10.1007/7651_2015_218
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-2953-5
Online ISBN: 978-1-4939-2954-2
eBook Packages: Springer Protocols