Abstract
Introduction
Heat shock proteins (HSP) are pivotal players in the normal cellular physiological processes and possess regulatory functions in pathogenesis of age-related disorders. HSP as chaperons are participating in protein folding, proper protein conformation, and prevention of undesired protein aggregation. In here, we provide the essential roles of HSP in inflammation with special focus on the ageing-related inflammatory diseases such as Alzheimer’s disease, Parkinson’s disease, diabetes, rheumatoid arthritis, and atherosclerosis.
Methods
A literature based collection of articles in the available search engines (PubMed and Google Scholar).
Results
We show the interrelation of HSP and inflammation-related ageing disorders such as Alzheimer’s disease, Parkinson’s disease, diabetes, rheumatoid arthritis, and atherosclerosis.
Conclusions
Understanding the critical roles of HPS would help in designing and manufacturing therapeutics for ameliorating the symptoms associated with age-related diseases.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
In 1962, Ritossa has been discovered Heat chock proteins (HSP). They are a family of highly conserved ubiquitous proteins. This family composed of group of different molecular weight proteins including HSP10, Hsp27, HSP40, Hsp60, Hsp70, Hsp90 and Hsp110 [25]. However, Hsp70 and 90 are the two major types that have the potential to bind to the unfolded protein helping them to folded and synthesized properly [25, 120]. Some types of this protein could express substantially, while the most of them expressed under stress conditions [166]. A variety of environmental or physiologic stress could lead to production and activation of HSP such as inflammation, hypoxia, chemotherapy, infections, as well as thermal injury [1, 86, 157,158,159,160,161]. In living systems, HSP have essential activities including polypeptides folding, proteins transportation, and formation of multiprotein complexes [62]. Moreover, they can prevent apoptosis, cleared aggregated proteins, and ameliorates the cytotoxic impact of toxic proteins.
During aging, declined HSP expression was reported in several tissues particularly muscle, liver, neurons, and vascular system [100, 133], with resultant protein aggregation, a commonly noted feature in neurodegenerative disorders [57, 64]. On the other hand, it is now well documented that, aging is related to presence of high concentration of pro-inflammatory cytokines such as interleukins (IL), IFNα, IFNβ, C-reactive protein (CRP), tumor necrosis factor (TNF), and transforming growth factor-β (TGFβ) [49, 55]. This inflammatory response becomes evolutionarily benefit during adulthood. However, during aging, it becomes detrimental due to inactive natural selection [53]. Inflamm-ageing is a chronic inflammatory response associated to the physiologic aging. It is the primary risk factor for the common age-related pathologies including malignancy, dementia, and cardiovascular disorders. Moreover, it may be considered as generalized health indicator for mobility disability, impairment of daily activities, and premature death [105, 156, 162].
Recently, HSP proteins were reported for their anti-inflammatory and antiapoptotic effect. Therefore, they have the potential to modulate and reduce the responses against various inflammatory cytokines. Consequently, understanding for the essential role of HSP in pathogenesis of age-related chronic inflammatory diseases may be a promising target to block the establishment of those diseases [44]. The aim of the present work is to update knowledge concerning the key role of HSP in pathogenesis of age-related inflammatory diseases including neurodegenerative disorders (such as Alzheimer’s disease and Parkinson’s disease), vascular disorders (such as Atherosclerosis), diabetes, rheumatoid arthritis, and neoplastic changes.
1.1 History and Types of HSP
HSP are a collection of common and highly preserved proteins. According to their size, HSP have been categorized into two groups: small molecular weight HSP and high molecular weight HSP. The first group contains four families: Hsp60, Hsp70, Hsp90, and Hsp110. Some of these proteins have continuous expression whereas stressful conditions induce the expression of the others [166]. High molecular weight HSP are ATP-dependent chaperones and need cochaperones to modify their conformation and ATP binding. On the other hand, small molecular weight HSP are ATP independent chaperones. HSP are stimulated by many environmental and physiological factors, such as inflammation, hypoxia, temperature stress, anticancer chemotherapy, or infections [86].
1.2 Role of HSP in the Inflammatory Mechanism
The best biological stimulant to induce the innate immunity response is the invasion of a foreign molecule. Innate immune recognition receptors decide to respond or ignore that stimulus by activation of PAMP (pathogen-associated molecular patterns) molecules, i.e. factors linked with groups of pathogens (for example bacterial CpG DNA, lipopolysaccharides, etc.). Other pathway for the induction of innate immunity is ‘danger theory’ [119]. By this hypothesis, the innate immunity can be induced by endogenous substances produced by the stressed or damaged tissue. Based on this theory, stressed cells can transfer stress to other cells. The stress signals produced by cells can be the HSP stimulated in response to the damage, so they are possible candidates for signaling cellular stress or tissue damage. Some of HSP such as Hsp70 and Hsp60 have been detected to be capable of signaling by TLR-4, TLR-2, and CD14 [8, 187]. Now, it is well established that Hsp70 is in charge of the stimulation of monocytes, macrophages, natural killer cells, dendritic cells, hepatocytes, etc. [26, 42, 58, 174]. Furthermore, extracellular HSP have been detected to work as powerful immunosuppressive or immunostimulatory molecules according to the various circumstances [124].
In addition to Hsp70, other HSP have been detected in the extracellular matrix, such as Hsp27 [108], Grp78/BIP [45, 99], Hsp90 [184], and Hsp60 [124]. The biological relation of these factors has been enhanced by the presence of Hsp70 in the serum of patients having myocardial infarction [47], chronic inflammation [135], coronary artery disease [212, 216], lung injury [59], infections [135], ischemia/reperfusion events [75], cancer [12], diabetes [137], hypertension during pregnancy [128], etc. HSP can also be detected at lower levels in the serum of healthy individuals [145]. Interestingly, the existence of Hsp70 in plasma is linked to enhanced survival of sever ill patients [217]. Other extracellular HSP such as Hsp90 [170], Hsp60 [211], and Hsp27 [108] have also been correlated with many diseases like coronary heart disease, pancreatic carcinoma, systemic lupus erythematosus or cancer metastasis.
Cytoprotection is the main role of HSP. Some investigations have revealed the number of cells surviving elevated when the temperature increased till 43 °C. This thermotolerance was attributed to the increased formation of HSP [104]. Some reports suggested the role of HSP in the suppression of stress-activated kinases. [56] revealed that preheating the human leukemic cells resulted in decreased cell death after heat shock, which was correlated with p38 activation and suppression of JNK. This influence might be made by HSP. This overexpression suppressed the stress kinase-activating influences of ultraviolet irradiation, H2O2 and heat. Park et al. indicated that Hsp72 inhibits the JNK signaling pathway by prohibition of JNK phosphorylation by its upstream kinase SEK1 [140]. Moreover, Hsp70 has been involved in the suppression of IKKγ and following synthesis of IKK complexes [163]. As for the impact of NfkB in inflammation, the suppression of its kinase, IKK, has specific therapeutic importance for inflammation. Furthermore, overexpression of Hsp70 prevents sepsis-induced lung injury in rats through suppression of the IKK complex [193].
1.3 Role of HSP in Aging-Related Diseases
The aggregation of oxidized proteins is an essential feature of the major neurological diseases, and many investigations have revealed that elevating HSP levels can have useful influences [101]. HSP have been detected to decrease the symptoms of Alzheimer’s disease, a disease caused by the aggregation of β-amyloid in neurons [148]. The aggregation of β-amyloid was decreased after the over-expression of HSP in primary neurons and neural blastoma cells [189]. In an in vitro study, Hsp70 was revealed to help in the digestion of amyloid plaques by enhancing microglia [90].
In Parkinson’s disease, cytoplasmic protein aggregates (Lewy bodies) containing numerous proteins such as HSP, α-synuclein, ubiquitin and parkin are present in neurons [51]. Cells have increased levels of HSPB5 (αB crystalline) were detected to have a significantly decreased number of Lewy bodies [23]. Although, Amyotrophic lateral sclerosis (ALS) has no known cause, there is a powerful proof connecting this neurodegenerative disease with dysfunctional superoxide dismutase (SOD). It has been detected that Hsp70 interacts with ubiquitin to aim the degradation of dysfunctional SOD by proteasome [186]. Treatment of mice with HSP inducer, arimoclomol, lowered the progression of ALS [93]. ROS participate in the development of multiple sclerosis lesions, and an elevation in HSP expression with the beginning of multiple sclerosis has been detected [132]. Antioxidant therapies have a beneficial effect for multiple sclerosis [167], and an elevation in HSP is accompanied by the decrease of plaques in multiple sclerosis [152].
In addition to having a pivotal role in the treatment of age-related diseases, modification of HSP levels reveals a healthy lifespan and elevating longevity of humans. The function of HSR and HSP across species means that several influences observed in lower organisms could be utilized in humans. Many investigations on C. elegans have revealed marked elevations in life-span with the increased expression of HSP [191]. Moreover, enhanced longevity because of caloric restriction demanded a functional HSR pathway in C. elegans [175]. In Drosophila, HSP revealed the ability to elevate the lifespan of Drosophila, where overexpression of HSP22 was detected to enhance protection against stress and expand longevity [129]. Moreover, Murine models have assisted to reveal the mechanisms behind elevated life-span. Mouse embryonic fibroblasts were detected to reach replicative senescence far sooner at elevated levels of oxidative stress [142], and mice lacking the ubiquitin ligase/co-chaperone CHIP reveal a lowered lifespan with a quickly ageing phenotype [126]. A comparison between short-lived Mus musculus and long-lived Peromyscus leucopus, two closely related species of mice, revealed that the difference in life-span correlated with a variance in oxidative stress tolerance [37]. Moreover, caloric restriction, which is known to increase maximal life-span in murine models, was detected to keep levels of Hsp70 and Hsp60, which normally both decrease during ageing [35].
Exposure of human fibroblasts to frequent heat stress was detected to elevate the levels of Hsp70, Hsp27, HspA8 and Hsp90 and elevate tolerance to oxidative stress; however, no elevation in their Hayflick limit was detected [52]. Extracellular (secreted) HSP, in contrast to intracellular HSP, could have detrimental influences and be pro-inflammatory. Importantly, plasma levels of Hsp70, when compared to controls, were inversely correlated with longevity and significantly decreased in centenarian offspring [178].
1.4 Role of HSP in Alzheimer’s Disease
Recently, Alzheimer’s disease (AD) has a major effect on the international public health. AD characterized by the abnormal synthesis of Tau and amyloid-peptides (Aβ), resulting in the pathological creation of intracellular neurofibrillary tangles (NFTs) and extracellular senile plaques. Insoluble Aβ with a sequence between 38 and 42 amino acids created senile plaques in brains of AD patients [178]. According to the amyloid hypothesis, β-amyloid precursor protein (APP) which is a trans-membrane protein created Aβ peptides. Aβ42 is the main motif in amyloid plaques and creates the most toxic oligomers. Therefore, the increased synthesis of Aβ stimulates cell death, finally resulting in dementia [141]. Moreover, the pathological hyperphosphorylation of protein Tau and its misfolding and accumulation within the cytoplasm resulted in the intra-cellular NFT lesion [63].
The major cause of neuron’s injury in AD is because of stress stimulated by the misfolding of Tau and Aβ peptides, inducing the synthesis of toxic oligomers and finally NFTs, the significant of the chaperones in AD has been proofed in the last two decades [113]. Among molecular chaperones, Heat Shock Proteins (HSP) are major constituent of the chaperone and Hsp90, Hsp70 and Hsp60 are deemed target of special superiority in AD [116] and as cancer [32].
Molecular chaperones modify protein activity, organize protein folding and target misfolded or accumulated proteins for degradation or for refolding. HSP are pivotal to ease the protein folding process [39]. They share in various mechanisms to guard the cells against stress-related mechanisms hurtful to the cell [39]. So, as detected in different neurodegenerative diseases, failure of these cellular mechanisms can lead to pathogenic lesions. Several data revealed that HSP organize protein misfolding in many neurodegenerative diseases, such as AD, showing preventative roles and/or working as pathogenic factors. Stress-induced proteins like chaperones have been reported to work as preventive molecules for cells of the nervous system [116]. Several proofs revealed that oxidative stress is a characteristic of PD and AD [208]. Abnormal aggregation of Tau and Aβ proteins and mitochondrial dysfunction can share in making the imbalance between antioxidant and oxidant mechanisms defining oxidative damage in AD patients [208]. In the brain, oxidative stress can make destruction that share in neuronal loss [2]. Reactive oxygen species (ROS) can aggregate inside cells and have negative influences on all biological molecules, determining, for instance, enzyme inactivation, nucleic acid breakage, lipid peroxidation and polysaccharide depolymerization. Under these stress circumstances, the expression of the genes encoding HSP was stimulated [2]. Furthermore, increased levels of ROS and mitochondrial dysfunction might synthesis a vicious circle sharing in AD progression and instauration [116].
Recently, several results were acquired from research on anti-cancer agents. Some compounds of therapeutic impacts were detected but clinical trials are not granted till now. So, acquisition further information is essential and several questions should be lighted, such as: (i) mode of action of HSP inhibitors; (ii) AD biochemical pathways related to HSP; (iii) sensing of client/HSP protein-protein interactions at the molecular level; (iv) selection of stress-induced versus constitutive HSP [138, 151]. In conclusion, HSP targeting might be the fundamental for potential drugs in the polypharmacological approach and multitargeted drug discovery and toward a complex disease such as AD. The role of HSP in the pathogenesis of Alzheimer’s disease is illustrated in Fig. 1.
Down-regulation of Hsp90 in Alzheimer’s disease induce decline in aggregation and hyperphosphorylation of Tau protein. In cellular stress and Hsp90 inhibitors, Heat Shock Factor 1 (HSF-1) dissociates from the chaperone and induces the activation of heat shock genes within nucleus and stress response through production of Hsp90, Hsp70 and Hsp40, restoring protein homeostasis
1.5 Role of HSP in Parkinson’s Disease
Parkinson’s disease (PD) is the second most popular neurodegenerative disorder influencing 1% of the population over 60 [41]. People having PD suffer from cardinal motor symptoms comprise muscular rigidity, bradykinesia, gait decay or rest tremor but often cause nonmotor symptoms, like psychiatric symptoms and cognitive sickness. Loss of the dopaminergic neurons of the substantia nigra (SN) pars compacta lead to most of symptoms accompanied by PD result from [91]. Recently, PD is handled surgically, by deep brain stimulation (DBS) and, pharmacologically, by supporting dopamine tone (e.g., dopamine replacement with L-dopa) [91]. As the disease advances L-dopa remediation is accompanied by complications comprising dyskinesia and motor fluctuation. DBS is limited to a group of patients suffering from L-dopa-induced complications and L-dopa responsive motor symptoms, but without marked psychiatric disturbance or cognitive sickness. Interestingly, both interventions result in symptomatic cure and do not slow the progression of PD.
Thus, there is a requirement for a remedy targeting the main sources of the disease. By the pathological view, PD cause the existence of proteinaceous intracellular aggregates composed primarily of 𝛼-synuclein, called Lewy pathology (Lewy neurites and Lewy bodies). Multiplications and missense mutations of the SNCA gene, which encodes for 𝛼-synuclein, induce the tendency of 𝛼-synuclein to self-accumulate and cause heritable forms of PD and therefore involving 𝛼-synuclein accumulation in the pathogenesis of the disease [147, 172]. While there is suspicion concerning the specific form of accumulates (“species”) that are neurotoxic, novel proof supposes that 𝛼- synuclein toxicity is granted by soluble oligomeric species [36, 92, 179]. Due to the pivotal role of 𝛼-synuclein accumulation in PD, researchers study about the nature and modification of the molecular pathways in charge of directing protein misfolding and folding, decreasing abnormal protein aggregation and keeping proper protein confirmation, gives a potential path for distinguishing a disease altering strategy.
Early proof involving molecular chaperones in the pathobiology of PD concluded from the detection by Auluck et al. [11] that Hsp70 overexpression alleviate 𝛼-synuclein-mediated dopaminergic neurodegeneration in a Drosophila model. This indicates that Hsp70 can have a neuroprotective role in PD. Posteriorly, McLean et al. [122] reported that the overexpression of Hsp70 and Hsp40 family members decreases the synthesis of 𝛼-synuclein accumulates in vitro and that Lewy bodies colocalize with multiple chaperone proteins. Molecular chaperones were involved in the pathobiology of PD by the detection of mutations within the promoter region upstream of both inducible and expressed Hsp70 family members elevate the danger of PD [201]. Moreover, mutations in the HspA9 (mortalin), Hsp70, were indicated to enhance the progress of PD [43]; on the other hand, other groups indicate mutations in HspA9 are not a common reason of early-onset PD as they are also detected in patient controls [54].
The ability of Hsp70 overexpression to improve 𝛼-synuclein toxicity has been well studied in yeast by autonomous groups which have revealed that Hsp70 overexpression can reduce 𝛼-synuclein in mediated cell death [50] and decrease high molecular weight aggregates in rodent models of PD [102, 127]. Hsp70 overexpression was detected to be preventive against cell death caused by the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), mitochondrial complex I inhibitor, both in vivo [46] and in vitro [149]. 𝛼-synuclein aggregation is not characteristic to the toxin model, but 𝛼-synuclein is necessary for MPTP-induced cell death as revealed by the opposition of 𝛼-synuclein null mice to MPTP [40]. On the other hand, mitochondrial HspA9 may play a pivotal role in the mitochondrial flaws inspired by the pathological A53T mutant 𝛼-synuclein as HspA9 knockdown prevents against the mitochondrial fragmentation and elevated tendency to the complex I inhibitor, rotenone, stimulated by A53T overexpression [111].
The mechanism, Hsp70 decreases 𝛼-synuclein toxicity, appears to be dependent mechanism on both its function in protein degradation and its refolding activity by the ALP and UPS. Mutations which change the ATPase function of Hsp70 (K71S) cancel its preventive influence on 𝛼-synuclein toxicity, suggesting that Hsp70 folding activity is essential for its preventive function [102]. Importantly, this mutation has no influence on the ability of Hsp70 to inhibit 𝛼-synuclein accumulation [102], indicating that Hsp70 utilizes clear mechanisms to decrease the aggregation and toxicity and of 𝛼-synuclein. Moreover, Hsp70 can ease disassemble of preformed 𝛼-synuclein aggregates [134]. Gao et al. [60] revealed that an Hsp70 machine composed of HspH2, DNAJB1, and HspA8 could efficiently disaggregate created 𝛼-synuclein fibrils in vitro.
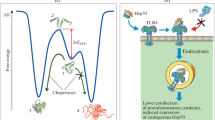
Many studies have indicated that CMA may play a pivotal role in alleviating 𝛼-synuclein toxicity [204]. Promoted 𝛼-synuclein expression in both paraquat and transgenic models of PD leads to the enhancement of HspA8 and LAMP2A expression and a larger movement of 𝛼-synuclein into the lysosomes [115]. Furthermore, both HspA8 and LAMP2A have decreased expression in the SN of PD patients [3], and a novel investigation revealed a link between the 𝛼-synuclein aggregation and loss of LAMP2A in postmortem PD brains [131]. Importantly, the detected reduction in HspA8 and LAMP2A expression anatomically overlaps with an elevation in miRNAs able to translationally suppress both HspA8 and LAMP2A [4], and implicate miRNAs in PD-associated chaperone dysregulation. In conclusion, the ability of Hsp70 and its cochaperones to disaggregate, refold, and aim for destruction of toxic 𝛼-synuclein species indicates that molecular chaperones can have a pivotal role in the pathobiology of PD. The role of HSP in the pathogenesis of Parkinson disease is illustrated in Fig. 2.
Proposed role of molecular and small molecule chaperones in proteostasis in Parkinson disease. At normal state, Hsp90 bind to HSF-1 and block its activity. However, in presence of Hsp90 inhibitors (such as SNX compounds, geldanamycin, 17-AAG) or proteotoxic stress, active HSF-1 separated from Hsp90 and translocates to the nucleus where it stimulates the expression of Hsp70. Members of inducible Hsp70 family induce proteasomal degradation via a pathway activated by E3 ligases, CHIP, and Parkin. This degradation is prevented by members of BAG family and enhanced by celastrol and carbenoxolone (small molecule HSF-1 activators). In Proteotoxic stress, the misfolded proteins directed for degradation via the interactions of autophagy-lysosome system with various chaperone-mediated autophagy. Chaperone/cochaperone complexes can play a role in disaggregation of already formed protein aggregates. Also, the pharmacologic chaperones such as isofagomine and ambroxol can activate glucocerebrosidase (GCase) in the lysosome to further stimulation for chaperone-mediated autophagy. In the endoplasmic reticulum and mitochondria, chaperone are regulated by specific members of the Hsp70 family, HspA5 and HspA9, respectively
1.6 Role of HSP in Diabetes
Diabetes is a condition implicated a chronic elevation of blood glucose levels (hyperglycemia). This disease is classified into 2 types: type 1, which is accompanied by the demolition of pancreatic beta cells leading to scanty insulin production; and type 2, which embraces a range of disorders that finally result in hyperglycemia [196]. Both types of diabetes elevate the potential for the development of microvascular disorders, such as neuropathy, retinopathy and nephropathy, and for macrovascular disorders [10].
It has been mentioned that these HSP chaperones are accompanied by several clinical disorders, containing diabetes. HSP have been involved in the sources of type 1 diabetes and in the cure of the obesity and insulin resistance implicated in type 2 diabetes [34]. Reduced expression of Hsp70 and suppression of heat shock factor-1 (HSF-1) have been reported in different tissues of rats with type 1 diabetes. Inhibition of HSP 70 levels by diabetes is accompanied by elevation in tissue inflammation. Moreover, the same authors mentioned that normal Hsp70 and HSF-1 stimulation by endurance exercise has been prevented by diabetes [185].
Many investigations have revealed reduced expression of HSP in patients with type 1 and 2 diabetes. It appears that the decrease in chaperon activity in diabetic patients is one of the major causes for beginnings of diabetic problems. So, researchers are seeking to use various techniques, inclusive pharmaceutical and chemical compounds, thermotherapy and exercise, to stimulate the expression of HSP [81]. Former investigations suggest that an elevation in protein stability and a decrease in protein glycation can markedly reduce the complications of diabetes [85].
Diabetes is a disorder including elevated glycation, oxidation and inflammation; therefore, it would have been foretold that levels of HSP could be highly preventive in persons suffering from diabetes. However, results of investigations in humans and animals with diabetes detected reduced HSP expression. Therefore, the paradoxically reduced levels of HSP confirm the destruction caused by diabetes lesions. Intracellular HSP, through blocking nuclear factor-kB (NF-kB) activation, have anti-inflammatory influences on cells. Protein kinase C activation by NF-kB is a primary pathway resulting in diabetes-induced cytokine gene expression. Therefore, reduced levels of HSP in cases of diabetes will elevate the activity of NF-kB and confirm inflammation [80].
Type 2 diabetes mellitus is age related; it can lower longevity and fast several traits accompanied by aging. HSP are factors which have a pivotal role in aging and longevity [81]. Patients with type 2 diabetes have elevated incidence of neurodegenerative diseases, such as Parkinson and Alzheimer diseases. In Parkinson and Alzheimer diseases, amyloid precursor aggregation may result in a common loss of insulin signaling in the pancreatic beta cells and in the brain. Moreover, loss of insulin signaling results in reduced HSP in beta cells or neurons, which leads to abnormal protein aggregation and function. It has been mentioned that administration of Hsp70 and insulin can lower amyloid aggregation in the brain [81].
Bimoclomol is a drug that can elevate the fluidity of membrane and expands the activity of HSF-1, therefore can elevate the levels of Hsp70. It has been mentioned that bimoclomol lowers tissue damage, enhances wound healing, ameliorates insulin sensitivity in animal models of diabetes and decreases diabetes complications [74]. Lipoic acid administration in patients with neuropathy and type-1 diabetes was accompanied by normalization of the low level of Hsp72. This Influence was attributed to clinical amelioration in the neuropathy in these patients. It has been mentioned that thiazolidinediones, carvedilol and exercise elevate HSP. The anti-inflammatory action of on the pancreatic beta cells could be linked to the elevation of Hsp70 levels by this drug. Nitric oxide is a powerful inducer of HSP expression. Drugs that retrieve the secretion of nitric oxide from blood vessels, such as angiotensin-converting enzyme inhibitors, beta-adrenergic blockers, thiazolidinediones and HMG-CoA reductase inhibitors, are correlated to outstanding results in clinical trials of diabetes. Near-infrared light therapy releases nitric oxide from endothelium and, thus, treat diabetic neuropathy.
Finally, the oral or intravenous administration of HSP is impractical due to the intracellular position of HSP. On the other hand, it has been mentioned that liposomal delivery of Hsp72 into renal tubular cells blocks induction of NF-kB tumor necrosis factor and, so, blocks ischemia-induced apoptosis. It is an important finding that several drugs or conditions that may elevate HSP levels and also block NF-kB (i.e. statins [76], exercise [150], pentoxifyllin and carvedilol [79].
1.7 Role of HSP in Atherosclerosis
Atherosclerosis is an old disease slowly progressing disease that becomes manifested in the middle age or later, even if it begins in childhood [155]. In past, atherothrombosis is considered as the first killer of the aging people in the developed countries; however, dramatic increase in its incidence in the developing countries was recently reported. Currently, around 39% of death cases reported in the U.K. is concerning to atherosclerosis, however about 12 million of American citizens suffered atherosclerosis-related diseases [13]. This disease is characterized by lipids deposition, especially of low-density lipoproteins (LDLs), on the endothelial layer of medium- sized and large arteries, together with remodeling of arterial walls and severe infiltration of immune cells, forming the characteristic plaques called atheroma. Although the signs of disease have been discovered in Egyptian mummies more than 4000 years old [180], the lipid composition of atheroma and the combined mononuclear infiltration were first described about two centuries ago [121]. However, the scientists have reached to the inflammatory hallmarks in progress and pathogenesis of atherosclerosis over the past 30–40 years [73]. Recently, several researches proved that inflammation is the first steps of atherosclerosis [109]. They concluded that the expression of adhesion molecule on the endothelial cells, such as vascular adhesion molecule-1, intercellular adhesion molecule-1, and E-selectin, beside the activation of macrophages, T lymphocytes, mast cells, and several cytokines suggesting involvement of inflammatory and immune processes in the pathogenic progress of atherosclerosis [67, 72].
From another sight of view, the key role played by the immune reactivity in the pathogenesis of atherosclerosis confirmed the essential contribution of inflammatory process. Many investigators induced experimental atherosclerosis in rabbits by high fat diet, and examined the therapeutic activities of immunosuppressive drugs, authors concluded marked prevention for plaque formation and inflammatory infiltration in the aorta; they outlined the relationship between the declined production of local inflammatory and immune cells and the reduction in cholesterol content in the arterial walls [68, 190].
Normally, cells subjected to stress stimuli, such as oxidized LDL, heat shock, infectious, surgical, mechanical stress, or cytokine activation, will respond by production of increased levels of HSP to protect themselves from stress stimuli [16]. Accordingly, it was described that HSP could expressed in high levels in cardiovascular tissues to initiate the inflammatory process, and that they may be expressed during the progress of atherosclerosis as an autoantigen [143].
In 1990, Berberian et al. reported for the first time the increased expression of Hsp70 in arteries of human and rabbits. Authors concluded that the distribution of HSP in arteries was correlated to necrosis, lipid accumulation, and macrophages infiltration in human atheroma. Interestingly, Hsp70 was found to be concentrated mainly in the central thickened portions of atheroma around the accumulated lipid and sites of tissue necrosis [88]. On contrary, some of the most complex plaques contained foci of smooth muscle cells without obvious relation to necrosis or increased expression of HSP [87]; where Hsp70 was produced in arterial wall cells even in dendritic cells [20]. Authors concluded that HSP production was increased within the depth of plate, particularly in macrophages and associated to necrotic tissue.
Consistently, Xu et al. [207] indicated that Hsp70 is overexpressed in the advanced atherosclerotic lesions. Authors found that Hsp70 ameliorates the NFκB activation, suggesting its anti-inflammatory potential. In another studies, authors declared that the levels of Hsp70 in plasma have a direct [203] and inverse [119] relation to atherosclerosis severity. Additional investigations concluded that administration of Hsp70 promoted the production of pro-inflammatory (such as IL-6) [9] and anti-inflammatory (such as Treg) cytokines [194]. Interestingly, Hsp70 could be considered as a favor factor for progression of atherosclerosis as well as mononuclear inflammatory infiltration. This theory confirmed in study designed by Xie et al. [203]; authors concluded that feeding diet with high-cholesterol level led to increased levels of Hsp70 in plasma. Additionally, the exogenous supplementation of Hsp70 promotes production of adhesion molecules within mononuclear cells in peripheral blood. In contrast, Madrigal-Matute et al. [114] observed that overexpression of Hsp70 was associated with declined oxidative stress and inflammatory response in the walls of arteries; suggesting its protective potential. Therefore, the promoting and inhibition effect Hsp70 against atherosclerosis are still a debate matter [18].
On the other hand, several investigations focused on the role of Hsp90 in atherogenesis. It was observed that the overexpression of Hsp90 is related to instability of atheroma. As well, the inhibition of Hsp90 led to declined production of inflammatory cells and oxidative stress due to reduced activation of transcription factors (such as the activators of transcription and NFκB signal transducers). Interestingly, the suppression of Hsp90 activity could be benefit in promoting the overexpression of Hsp70, with subsequent inhibition of the proinflammatory response and atherogenesis [114].
Recently, a growing body of evidence suggested the direct atherogenic potential for Hsp60; where increased expression of Hsp60 usually precedes the growth of atherosclerotic plaque [95]. In humans, the increased level of circulating Hsp60 and anti-Hsp60 are correlated to thickness of carotid artery wall [202], atherosclerotic lesions [146], and atherosclerosis-associated morbidity and mortality [205]. Furthermore, early atherosclerotic lesion was induced by transfer of Hsp60 reactive T cells [197]; where specific immunity of T-cell to Hsp60 is induced (Knoflach et al. 2007). In addition, administration of Hsp60 might induce or suppress atherogenesis, based on administration route, and the involved co-stimulatory molecules. Administration of Hsp60 parenterally activate infiltration of Hsp60-specific T cells, with subsequent secretion for anti-Hsp60 antibodies, pro-inflammatory cytokines, accumulations of macrophages and lipid, and atheroma formation. However, administration of Hsp60 via oral or nasal route reduced the atherosclerotic lesions, due to induction of Tregs and anti-inflammatory mediators including interleukin-10 (IL-10) and transforming growth factor beta (TGF-β) [197]. In human atherosclerosis, Kleindienst and colleagues indicated that Hsp60 was identified on smooth muscle cells, mononuclear inflammatory cells, and endothelial cells of aorta and carotid artery compared to the small blood vessels that had no sclerotic lesions. The positive correlation between the severity of atherosclerosis and the produced Hsp60 was also confirmed by Hammerer-Lercher et al. [70]. In another investigation, the expressions of Hsp60 and Hsp70 in the aortic tree showed positive correlation with the progress of atherosclerosis in apoE-deficient mice [95]. The main expression sites for both HSP were within macrophages, smooth muscle cells, endothelium, and CD3 T lymphocytes [95].
Interestingly, results from another study revealed that Hsp47 might be also involved in atherogenesis [198]. Strong expression of Hsp47 was proved locally in atherosclerotic arteries (particularly in the collagenous areas) but not in normal artery. HSP47 was expressed mainly in cells produce type I procollagen [154]. Results from this study suggested the role of Hsp47 in atheroma formation in human coronary. In addition, authors concluded the upregulation of Hsp47 as a response to stress; this conclusion might indicate the possible role of Hsp47 in plaque stability.
Hsp27 is an intracellular chaperone that possesses an important role in stabilization of RNA, beside its role in the antioxidant and antiapoptotic responses [14]. In atherosclerosis, extracellular production of Hsp27 from atheroma was evident; may be due to cellular damage or as a co-secretion with exosomes or lysosomes. After secretion, in the extracellular space, Hsp27 able to binds with several receptors on cell membrane of inflammatory immune cells and endothelial cells, such as CD14, CD36, CD40, CD91, scavenger receptor A (SR-A), and toll like receptors (TLRs) as TLR2, TLR3, and TLR4 [19]. Interestingly, data from available research concluded the ameliorative role played by Hsp27 during atherogenesis. On the same line, the definition of Hsp27 as an estrogen receptor-associated protein could explain the ameliorative role played by estrogens during atherogenesis [153]. Consistent with that, several studies demonstrated that atheroma has low content of Hsp27 [117], and therefore, low circulating levels of Hsp27 indicates more severe atherosclerotic lesions [169]. On contrary, overexpression of Hsp27 may protect against atherogenesis [38]. The protective potential of Hsp27 against atherosclerotic disease may attribute to its suppressive activity for NFκB activation [14], involvement in lipid homeostasis via competing with LDL in binding to SR-A, with subsequent formation of foam cell [14], and declined the cholesterol content in atheroma the serum [38]. The role of HSP in the pathogenesis of atherosclerosis is illustrated in Fig. 3.
1.8 Role of HSP in Rheumatoid Arthritis
Rheumatoid arthritis (RA) is a widely known chronic inflammatory disease that particularly affects the aging population. It occurs due to damage of the synovial membranes of joints via infiltration of mononuclear and/or polynuclear inflammatory cells including macrophages, lymphocytes, and neutrophils [69, 123]. Usually, during the course of RA, patient developed severe pain due to progressive injury or even complete loss of bone and cartilage around the inflamed joint. Generally, the pathogenesis of this disease is complicated; however, the pathologic events associated to RA suggesting an autoimmune cause in form of T-cell-mediated chronic inflammatory response [209].
On the other hand, it has been observed that overexpression of HSP family might be involved in RA pathogenesis; where increased expression can regulate the progress of disease [83]. The inflammatory events and other stress factors occur in synovial membrane are able to increase HSP expression. In RA, the hypoxia and reperfusion injury of the rheumatoid joint, lead to production of high levels of reactive oxygen species (ROS) [77]. Subsequently, this increased production of ROS and the high synovial content of inflammatory mediators (such as tumor necrosis factor (TNF)- a and interleukin (IL-l) will act as stress factors [199]. Consistently, Schett et al. [165] concluded that, in RA but not osteoarthritic, heat shock transcription factor 1 (HSFl) was activated and undergo hyper-phosphorylation and nuclear translocation that could lead to regulation of Hsp70 transcription. In another investigation, authors used cultured RA SM synovial fibroblasts to study the expression of Hsp70 in AR, they observed that the proinflammatory mediators including IL-l0 and TNF-α were able to upregulate expression of Hsp70 in cultured fibroblasts (Luo et al. 2008). In another research groups, authors declared that several types of HSP and chaperones were overexpressed in RA such as human Hsp27, Hsp90α, Hsp60 [168], and Hsp65 [83]. On contrary, Worthington et al. [200] concluded that human Hsp65 was expressed equally in RA and control non-inflamed synovia. Moreover, Hsp60 was expressed equally in mitochondria of RA and osteoarthritis, as assessed by immunohistology [22].
Karlsson-Parra et al. [96] observed that human Hsp65 (huHsp65) was overexpressed at the cartilage-pannus junction; they concluded that the eroding front possessed the maximum expression compared to SM itself. Interestingly, the same authors identified the expression of huHSP65 in rheumatoid nodules; where rheumatoid nodules are the pathognomonic histologic and clinical feature of RA. This expression could be attributed to the presence of non-caseated hypoxic center, where hypoxia act as stress factor lead to increased expression of HSP.
Lewthwaite et al. [106] observed the correlation between huHsp60 circulating in plasma and the psychosocial and physiologic stress. Consistent with this finding, the increased levels of circulating huHsp60 was correlated to carotid atherosclerotic plaque [202]. Generally, in osteoarthritis, HSP are produced mainly from the chondrocytes [171]. However, in RA patient, they synthesized mainly in synovial intimal cells [112]. Therefore, it could be concluded that the inflammatory signaling in this tissue are able to initiate the production of HSP; with subsequent protection for the host cells.
In another theory, RA may define as autoimmune-inflammatory disease; where the immune system attacks the synovial fluid-membranes exist in different joints. Neglect treatment of RA could result in severe inflammatory response [173]. This inflammation has the potential to attract several immune components such as immune chemokine, cytokines, and lymphocytes to the infection area [84]. As a response to RA infection, the synthesis of HSP particularly Hsp70 is increased. It is now well documented that Hsp70 possess an anti-apoptotic property through inhibition of proinflammatory and proapoptotic factors such as Caspases and JNK (Jun N-terminal) signaling, cytochrome c release, and apoptosome formation [66]. Therefore, the overexpression of Hsp70 in synovial membrane during the infection with rheumatoid arthritis fibroblast-like synoviocyte (RA-FLSs) is not surprisingly. Herein, Hsp70 acts to control the inflammatory process through blocking of pro-inflammatory signaling, and to regulate the effect of T-cells [165].
In another study, Kang et al. [94] observed repression of Hsp70 produced in RA fibroblast-like synoviocytes (FLSs) after treatment with sodium nitroprusside (SNP) in an in vitro experiment. Authors reported that Hsp70 downregulated cells showed better survive compared to control cells. It was concluded that downregulation of Hsp70 protects RA FLSs against apoptosis induced via nitric oxide production through activation of the Akt signaling pathway. However, the real in vivo function of Hsp70 in the RA is still not fully clear. By considering these findings, we can conclude that inhibition of Hsp70 in RA may be used as a therapeutic approach to control the severe inflammatory response occurs in RA. Additionally, van Roon et al. [188] noted that T-cells collected from patients suffered RA have the potential to react with huHsp60 to suppress the activation of the pro-inflammatory mediator (TNF-α) via induction of Th2 cytokine regulator. However, this regulation is not reported for Hsp65 isolated from Mycobacterium tuberculosis [144]. Consistently, several investigators attributed this response of T-cell to self-Hsp70 and Hsp60 to production of the regulatory mediators (interleukin-4 and interleukin-10), with subsequent prevention of arthritic diseases [6, 97, 144]. Taken together, it is may conclude that huHsp60 and mycobacterial Hsp60 might be considered as promising potential vaccines against autoimmune inflammatory diseases.
1.9 Role of HSP in Cancer
The progressive loss of physiologic and immunologic potency is a characteristic feature for the elderly [33]. Growing body of evidences has proved cancer augmentation by aging, which may be due to age- associated immune dysregulation [182], with subsequent poor prognosis [98]. Interestingly, around 50% of malignancies are diagnosed in aging patient over than 65 years old [78]. Several investigations concluded the anti-apoptotic activities of HSP. Therefore, it is not surprising that the high levels of HSP may have the potential to protect malignant cells against therapy-induced apoptosis [89]. The apoptotic process may occur in either intrinsic or extrinsic pathways. Whatever pathway, the final event is induction of caspases proteases, which is cleaved enzymatically leading to activation of the apoptotic stimulus [28]. In the intrinsic pathway, apoptosome is formed by mitochondria; where cytochrome c released from mitochondria to the cytosol, and then interact with pro-caspase-9 and cytosolic apoptosis protease activating factor-1 (APAF-1) forming apoptosome. Apoptosome is responsible for initiation of apoptotic cascade via activation of pro-caspase-3 [177].
Currently, it has been well known that Hsp27 and Hsp70 have the potential to inhibit the formation of apoptosome, with in turn inhibition of apoptosis. Another theory for inhibition of apoptosis by Hsp27 is direct binding to APAF-1, which subsequently led to inhibition of apoptosome formation [61]. Additionally, Hsp90 suppress pro-caspase-9 activation by cytochrome c [139]. On the other hand, the extrinsic apoptotic pathway work via binding to the respective ligands of death receptors (TNF receptor 1, TNF receptor superfamily, apoptosis antigen-1), leading to their activation and formation of death inducing complex at the plasma membrane. This complex activates the pro-caspase-8, which in turn induce direct or indirect activation of caspases [21].
In another research group, authors cleared that phosphorylated dimers of Hsp27 can bind to Daxx protein competitively with FAS; lead to subsequent interference with FAS-mediated apoptotic pathway [31]. Additionally, Bruey et al. [24] investigate the interaction between Hsp27 and cytochrome c. Authors concluded that Hsp27 can block Caspase activation via its binding to cytochrome c and inhibition for interaction with procaspase-9 and apoptotic protease activating factor-1 (APAF-1). In CD133 + colorectal cancer stem cells, activation of Hsp27 inhibits the cleavage of caspase-3 and -9 in the apoptosis pathway. However, its inhibition promotes apoptotic cascade in CD133+ cells [110]. Additionally, inhibition of Hsp27 activation up-regulated the activity of caspase-3 in glioblastoma cells [107].
Moreover, the anti-apoptotic activity of Hsp90 could be discussed by its ability to bind to the anti-apoptotic protein (such as AKT1) and suppress its activation, which in turn enhanced cell surviving [62]. In contrast, several investigators concluded the pro- apoptotic activities of Hsp60 in in vitro experiment [164]. In addition to the antiapoptotic and proapoptotic activities of HSP members, some members such as Hsp27 are essential also in regulation, progression, and metastasis of tumor cells. Interestingly, blocking of Hsp27 led to decline in matrix metalloproteinase (MMP), epithelial-to-mesenchymal transition, migration, and metastasis of neoplastic cells [65]. In addition, in human prostatic malignancy, Hsp27 has the potential to up-regulates MMP2 activity stimulated by transforming growth factor b (TGF-b), lead to promoting cell invasion [206]. Additionally, Hsp27 reported to enhance the neoplastic migration in bladder malignancy [210], and promote metastasis of epithelial ovarian cancer to peritoneum [215].
Thuringer et al. [181] studied effect of Hsp27 on progression and metastasis of breast cancer. They concluded that Hsp27 has a direct role in enhancement of angiogenic activity and neoplastic migration via upregulating gene transcription of vascular endothelial growth factor (VEGF) and activated VEGF receptor type 2. In another study, [136] found that Hsp27 inhibit p53-induced activation of p21 in neoplastic cells, with in turn regulation of p53 signaling. Moreover, proliferation of lung cancer cells could be enhanced by Hsp27-induced activation of activator protein-1 [214]. However, in gastric adenocarcinoma, cancer progression could be enhanced by the C-X-C chemokine receptor type 1 (CXCR1); CXCR1 has the potential to decrease Hsp27 expression, indicating the relationship between cancer progress, Hsp27, and CXCR1 [82].
It has been reported that Hsp90AA1 is involved in enhancement of invasiveness and mobility of cancer cells [195], where it is required for the invasion of fibrosarcoma cells [48]. On these bases, Hsp90AA1 found to enhance the in vitro invasion of breast cancer and melanoma, with in turn increment of the metastatic activities. Also, serum Hsp90AA1 increased in breast, liver, pancreas, and lung cancer in correlation to degree of malignancy [192]. However, inhibition of Hsp90AA1 suppresses the metastatic invasion in mouse melanoma [176]. This enhancement of HSP against tumor invasion potential may attribute to their binding to the extracellular receptors activating ERK1/2 and PI3K-Akt pathways [71]. Additionally, Tsuneki et al. [183] reported that HspA9 is released from oral squamous carcinoma cells, and then interact with podoplanin; that is an adhesion molecule responsible for the invasion potential of tumor. Moreover, HspB6 has a role in angiogenesis, progression, and migration. For example, overexpression of HspB6 led to increase density of heart capillaries in mice [213].
In recent years, many researchers studied the extracellular and intracellular localization of HSP in tumor cells. In normal cells, it is uncommon to localized Hsp60 on the cell membrane. However, this localization is frequent in malignant cells [27, 29]. In addition, it was reported that Hsp60 is exist in exosomes released from malignant cells in human [125]. The extracellular HSP have several functions; one of them is immune modulation. For instance, TNFα and IL-6 were produced in mast cells under stimulation of HspA1A via activation of toll-like receptors 2 and 4 (TLR4, TLR2) [130] and interleukin 12 (IL-12) [15]. Additionally, treatment with HspA1A led to activation of macrophage and production of TNFα [5]. Recently, in vitro studies were performed on murine leukemia monocytes and hepatocellular carcinoma cell line, author concluded release of HspA1A, Hsp90AA1, and HSPD1 from exosomes, which enhancing the activity of macrophages, natural killer, and mononuclear cells [103]. In contrast, Chalmin et al. [30] described the immunosuppressive role of HspA1A released from exosome; HspA1A is reported to suppress tumor immune surveillance via activation of myeloid-derived suppressor cells. In addition, in colorectal carcinoma, secretion of HspH1 led to differentiation of macrophage, with in turn anti-inflammatory profile and pro-tumor effect [17]. However, in primary breast tumor cells, released HspB1 led to monocytes differentiate into proangiogenic macrophages [7]. The role of HSP in the pathogenesis of cancer is summarized in Fig. 4.
Schematic representation for the role of Hsp27 in carcinogenesis. Hsp27 suppressed apoptosis with subsequent enhancement for tumorigenesis. Unphosphorylated Hsp27 present as large oligomers and possess chaperonage activities. Phosphorylated Hsp27 switches to smaller oligomers and lose their chaperonage activities and initiate their pro-oncogenic activities. Hyperactivation of Hsp27 induces inhibition of apoptosis
2 Conclusions
HSP are the cornerstone for repairing damaged proteins resulted exposure of cells to different stresses including the age-related disorders. Hence, enhancing and modulation of HSP functions would help the human welfare through promoting healthy lifespan and elevating the longevity of humans.
Abbreviations
- AD:
-
Alzheimer’s disease
- ALS:
-
amyotrophic lateral sclerosis
- Aβ:
-
amyloid β peptides
- CRP:
-
C-reactive protein
- HSF:
-
heat shock factor
- HSP:
-
heat shock proteins
- IL:
-
interleukins
- PD:
-
Parkinson’s disease
- RA:
-
Rheumatoid arthritis
- ROS:
-
reactive oxygen species
- SOD:
-
superoxide dismutase
- TGFβ:
-
transforming growth factor-β
- TNF:
-
tumor necrosis factor
References
Abdelnour SA, Abd El-Hack ME, Khafaga AF, Arif M, Taha AE, Noreldin AE (2019) Stress biomarkers and proteomics alteration to thermal stress in ruminants: a review. J Therm Biol 79:120–134
Abdul HM, Butterfield DA (2007) Involvement of PI3K/PKG/ERK1/2 signaling pathways in cortical neurons to trigger protection by cotreatment of acetyl-L-carnitine and alpha-lipoic acid against HNE-mediated oxidative stress and neurotoxicity: implications for Alzheimer’s disease. Free Radic Biol Med 42:371–384
Alvarez-Erviti L, Rodriguez-Oroz MC, Cooper JM, Caballero C, Ferrer I, Obeso JA, Schapira AH (2010) Chaperone-mediated autophagy markers in Parkinson disease brains. Arch Neurol 67:1464–1472
Alvarez-Erviti L, Seow Y, Schapira AH, Rodriguez-Oroz MC, Obeso JA, Cooper JM (2013) Influence of microRNA deregulation on chaperone-mediated autophagy and α-synuclein pathology in Parkinson’s disease. Cell Death Dis 4:e545
Anand PK, Anand E, Bleck CK, Anes E, Griffiths G (2010) Exosomal Hsp70 induces a pro-inflammatory response to foreign particles including mycobacteria. PLoS One 5:e10136
Anderton SM, Van Der Zee R, Prakken B, Noordzij A, Van Eden W (1995) Activation of T cells recognizing self 60-kD heat shock protein can protect against experimental arthritis. J Exp Med 181:943–952
Arispe N, Doh M, De Maio A (2002) Lipid interaction differentiates the constitutive and stress-induced heat shock proteins Hsc70 and Hsp70. Cell Stress Chaperones 7:330–338
Asea A (2003) Chaperokine-induced signal transduction pathways. Exerc Immunol Rev 9:25–33
Asea A, Kraeft S-K, Kurt-Jones EA, Stevenson MA, Chen LB, Finberg RW, Koo GC, Calderwood SK (2000) Hsp70 stimulates cytokine production through a CD14-dependant pathway, demonstrating its dual role as a chaperone and cytokine. Nat Med 6:435
Atalay M, Oksala N, Lappalainen J, Laaksonen DE, Sen CK, Roy S (2009) Heat shock proteins in diabetes and wound healing. Curr Protein Pept Sci 10:85–95
Auluck PK, Chan HY, Trojanowski JQ, Lee VM, Bonini NM (2002) Chaperone suppression of α-synuclein toxicity in a Drosophila model for Parkinson’s disease. Science 295:865–868
Azuma K, Shichijo S, Takedatsu H, Komatsu N, Sawamizu H, Itoh K (2003) Heat shock cognate protein 70 encodes antigenic epitopes recognised by HLA-B4601-restricted cytotoxic T lymphocytes from cancer patients. Br J Cancer 89:1079–1085
Barylski M, Małyszko J, Rysz J, Myśliwiec M, Banach M (2011) Lipids, blood pressure, kidney–what was new in 2011? Arch Med Sci 7:1055
Batulan Z, Pulakazhi Venu VK, Li Y, Koumbadinga G, Alvarez-Olmedo DG, Shi C, O’Brien ER (2016) Extracellular release and signaling by heat shock protein 27: role in modifying vascular inflammation. Front Immunol 7:285
Bausero MA, Gastpar R, Multhoff G, Asea A (2005) Alternative mechanism by which IFN-γ enhances tumor recognition: active release of heat shock protein 72. J Immunol 175:2900–2912
Benjamin IJ, McMillan DR (1998) Stress (heat shock) proteins: molecular chaperones in cardiovascular biology and disease. Circ Res 83:117–132
Berthenet K, Boudesco C, Collura A, Svrcek M, Richaud S, Hammann A, Causse S, Yousfi N, Wanherdrick K, Duplomb L, Duval A, Garrido C, Jego G (2016) Extracellular Hsp110 skews macrophage polarization in colorectal cancer. Onco Targets Ther 5:e1170264
Bielecka-Dabrowa A, Barylski M, Mikhailidis DP, Rysz J, Banach M (2009) HSP 70 and atherosclerosis–protector or activator? Expert Opin Ther Targets 13:307–317
Binder CJ, Chang M-K, Shaw PX, Miller YI, Hartvigsen K, Dewan A, Witztum JL (2002) Innate and acquired immunity in atherogenesis. Nat Med 8:1218
Bobryshev YV, Lord RS (2002) Expression of heat shock protein–70 by dendritic cells in the arterial intima and its potential significance in atherogenesis. J Vasc Surg 35:368–375
Boldin MP, Goncharov TM, Goltseve YV, Wallach D (1996) Involvement of MACH, a novel MORT1/FADD-interacting protease, in Fas/APO-1-and TNF receptor–induced cell death. Cell 85:803–815
Boog CJ, de Graeff-Meeder ER, Lucassen MA, van der Zee R, Voorhorst-Ogink MM, van Kooten PJ, Geuze HJ, van Eden W (1992) Two monoclonal antibodies generated against human hsp60 show reactivity with synovial membranes of patients with juvenile chronic arthritis. J Exp Med 175:1805–1810
Braak H, Del Tredici K, Sandmann-Kiel D, Rub U, Schultz C (2001) Nerve cells expressing heat-shock proteins in Parkinson’s disease. Acta Neuropathol 102:449–454
Bruey J-M, Ducasse C, Bonniaud P, Ravagnan L, Susin SA, Diaz-Latoud C, Gurbuxani S, Arrigo A-P, Kroemer G, Solary E (2000) Hsp27 negatively regulates cell death by interacting with cytochrome c. Nat Cell Biol 2:645
Calderwood S (2007) Molecular chaperones and the ubiquitin proteasome system in aging. In: The ubiquitin proteasome system in the central nervous system. Nova, New York, pp 537–552
Calderwood SK, Gong J (2016) Heat shock proteins promote cancer: it’s a protection racket. Trends Biochem Sci 41:311–323
Campanella C, Bucchieri F, Merendino AM, Fucarino A, Burgio G, Corona DF, Barbieri G, David S, Farina F, Zummo G (2012) The odyssey of Hsp60 from tumor cells to other destinations includes plasma membrane-associated stages and Golgi and exosomal protein-trafficking modalities. PLoS One 7:e42008
Cappello F, Bellafiore M, Palma A, Bucchieri F (2002) Defective apoptosis and tumorigenesis: role of p53 mutation and Fas/FasL system dysregulation. Eur J Histochem 46:199–208
Cappello F, de Macario EC, Di Felice V, Zummo G, Macario AJ (2009) Chlamydia trachomatis infection and anti-Hsp60 immunity: the two sides of the coin. PLoS Pathog 5:e1000552
Chalmin F, Ladoire S, Mignot G, Vincent J, Bruchard M, Remy-Martin JP, Boireau W, Rouleau A, Simon B, Lanneau D, De Thonel A, Multhoff G, Hamman A, Martin F, Chauffert B, Solary E, Zitvogel L, Garrido C, Ryffel B, Borg C, Apetoh L, Rebe C, Ghiringhelli F (2010) Membrane-associated Hsp72 from tumor-derived exosomes mediates STAT3-dependent immunosuppressive function of mouse and human myeloid-derived suppressor cells. J Clin Invest 120:457–471
Charette SJ, Landry J (2000) The interaction of Hsp27 with Daxx identifies a potential regulatory role of Hsp27 in Fas-induced apoptosis. Ann N Y Acad Sci 926:126–131
Chatterjee S, Burns TF (2017) Targeting heat shock proteins in cancer: a promising therapeutic approach. Int J Mol Sci 18:1978
Chen W, Zheng R, Zhang S, Zeng H, Xia C, Zuo T, Yang Z, Zou X, He J (2017) Cancer incidence and mortality in China, 2013. Cancer Lett 401:63–71
Chung J, Nguyen A-K, Henstridge DC, Holmes AG, Chan MHS, Mesa JL, Lancaster GI, Southgate RJ, Bruce CR, Duffy SJ, Horvath I, Mestril R, Watt MJ, Hooper PL, Kingwell BA, Vigh L, Hevener A, Febbraio MA (2008) HSP72 protects against obesity-induced insulin resistance. Proc Natl Acad Sci 105:1739–1744
Colotti C, Cavallini G, Vitale RL, Donati A, Maltinti M, Del Ry S, Bergamini E, Giannessi D (2005) Effects of aging and anti-aging caloric restrictions on carbonyl and heat shock protein levels and expression. Biogerontology 6:397–406
Conway KA, Harper JD, Lansbury PT (1998) Accelerated in vitro fibril formation by a mutant α-synuclein linked to early-onset Parkinson disease. Nat Med 4:1318–1320
Csiszar A, Labinskyy N, Zhao X, Hu F, Serpillon S, Huang Z, Ballabh P, Levy RJ, Hintze TH, Wolin MS, Austad SN, Podlutsky A, Ungvari Z (2007) Vascular superoxide and hydrogen peroxide production and oxidative stress resistance in two closely related rodent species with disparate longevity. Aging Cell 6:783–797
Cuerrier CM, Chen Y-X, Tremblay D, Rayner K, McNulty M, Zhao X, Kennedy CR, de BelleRoche J, Pelling AE, O’Brien ER (2013) Chronic over-expression of heat shock protein 27 attenuates atherogenesis and enhances plaque remodeling: a combined histological and mechanical assessment of aortic lesions. PLoS One 8:e55867
Czarnecka AM, Campanella C, Zummo G, Cappello F (2006) Mitochondrial chaperones in cancer: from molecular biology to clinical diagnostics. Cancer Biol Ther 5:714–720
Dauer W, Kholodilov N, Vila M, Trillat AC, Goodchild R, Larsen KE, Staal R, Tieu K, Schmitz Y, Yuan CA, Rocha M, Jackson-Lewis V, Hersch S, Sulzer D, Przedborski S, Burke R, Hen R (2002) Resistance of α-synuclein null mice to the parkinsonian neurotoxin MPTP. Proc Natl Acad Sci U S A 99:14524–14529
de Lau LM, Breteler MM (2006) Epidemiology of Parkinson’s disease. Lancet Neurol 5:525–535
De Maio A (2011) Extracellular heat shock proteins, cellular export vesicles, and the Stress Observation System: a form of communication during injury, infection, and cell damage. It is never known how far a controversial finding will go! Dedicated to Ferruccio Ritossa. Cell Stress Chaperones 16:235–249
De Mena L, Coto E, Sanchez-Ferrero E, Ribacoba R, Guisasola LM, Salvador C, Blazquez M, Alvarez V (2009) Mutational screening of the mortalin gene (HSPA9) in Parkinson’s disease. J Neural Transm (Vienna) 116:1289–1293
de Toda IM, De la Fuente M (2015) The role of Hsp70 in oxi-inflamm-aging and its use as a potential biomarker of lifespan. Biogerontology 16:709–721
Delpino A, Castelli M (2002) The 78 kDa glucose-regulated protein (GRP78/BIP) is expressed on the cell membrane, is released into cell culture medium and is also present in human peripheral circulation. Biosci Rep 22:407–420
Dong Z, Wolfer DP, Lipp HP, Bueler H (2005) Hsp70 gene transfer by adeno-associated virus inhibits MPTP-induced nigrostriatal degeneration in the mouse model of Parkinson disease. Mol Ther 11:80–88
Dybdahl B, Slordahl SA, Waage A, Kierulf P, Espevik T, Sundan A (2005) Myocardial ischaemia and the inflammatory response: release of heat shock protein 70 after myocardial infarction. Heart 91:299–304
Eustace BK, Sakurai T, Stewart JK, Yimlamai D, Unger C, Zehetmeier C, Lain B, Torella C, Henning SW, Beste G (2004) Functional proteomic screens reveal an essential extracellular role for hsp90α in cancer cell invasiveness. Nat Cell Biol 6:507
Ferrucci L, Semba RD, Guralnik JM, Ershler WB, Bandinelli S, Patel KV, Sun K, Woodman RC, Andrews NC, Cotter RJ (2010) Proinflammatory state, hepcidin, and anemia in older persons. Blood 115:3810–3816
Flower TR, Chesnokova LS, Froelich CA, Dixon C, Witt SN (2005) Heat shock prevents α-synuclein-induced apoptosis in a yeast model of Parkinson’s disease. J Mol Biol 351:1081–1100
Foley P, Riederer P (2000) Influence of neurotoxins and oxidative stress on the onset and progression of Parkinson’s disease. J Neurol 247(Suppl 2):Ii82–Ii94
Fonager J, Beedholm R, Clark BF, Rattan SI (2002) Mild stress-induced stimulation of heat-shock protein synthesis and improved functional ability of human fibroblasts undergoing aging in vitro. Exp Gerontol 37:1223–1228
Franceschi C, Garagnani P, Vitale G, Capri M, Salvioli S (2017) Inflammaging and ‘Garb-aging’. Trends Endocrinol Metab 28:199–212
Freimann K, Zschiedrich K, Bruggemann N, Grunewald A, Pawlack H, Hagenah J, Lohmann K, Klein C, Westenberger A (2013) Mortalin mutations are not a frequent cause of early-onset Parkinson disease. Neurobiol Aging 34:2694.e2619-e2620
Fulop T, Larbi A, Dupuis G, Le Page A, Frost EH, Cohen AA, Witkowski JM, Franceschi C (2018) Immunosenescence and inflamm-aging as two sides of the same coin: friends or foes? Front Immunol 8:1960
Gabai VL, Meriin AB, Mosser DD, Caron AW, Rits S, Shifrin VI, Sherman MY (1997) Hsp70 prevents activation of stress kinases. A novel pathway of cellular thermotolerance. J Biol Chem 272:18033–18037
Gagliano N, Grizzi F, Annoni G (2007) Mechanisms of aging and liver functions. Dig Dis 25:118–123
Gaipl US, Multhoff G, Scheithauer H, Lauber K, Hehlgans S, Frey B, Rodel F (2014) Kill and spread the word: stimulation of antitumor immune responses in the context of radiotherapy. Immunotherapy 6:597–610
Ganter MT, Ware LB, Howard M, Roux J, Gartland B, Matthay MA, Fleshner M, Pittet JF (2006) Extracellular heat shock protein 72 is a marker of the stress protein response in acute lung injury. Am J Physiol Lung Cell Mol Physiol 291:L354–L361
Gao X, Carroni M, Nussbaum-Krammer C, Mogk A, Nillegoda NB, Szlachcic A, Guilbride DL, Saibil HR, Mayer MP, Bukau B (2015) Human Hsp70 disaggregase reverses Parkinson’s-linked α-synuclein amyloid fibrils. Mol Cell 59:781–793
Garrido C, Bruey J-M, Fromentin A, Hammann A, Arrigo AP, Solary E (1999) Hsp27 inhibits cytochrome c-dependent activation of procaspase-9. FASEB J 13:2061–2070
Garrido C, Gurbuxani S, Ravagnan L, Kroemer G (2001) Heat shock proteins: endogenous modulators of apoptotic cell death. Biochem Biophys Res Commun 286:433–442
Gerson JE, Castillo-Carranza DL, Kayed R (2014) Advances in therapeutics for neurodegenerative tauopathies: moving toward the specific targeting of the most toxic tau species. ACS Chem Neurosci 5:752–769
Gestwicki JE, Garza D (2012) Protein quality control in neurodegenerative disease. In: Progress in molecular biology and translational science. Prog Mol Biol Transl Sci 107:327–353. https://doi.org/10.1016/B978-0-12-385883-2.00003-5
Gibert B, Simon S, Dimitrova V, Diaz-Latoud C, Arrigo A-P (2013) Peptide aptamers: tools to negatively or positively modulate HSPB1 (27) function. Philos Trans R Soc B Biol Sci 368:20120075
Gill C, Meyer M, FitzGerald U, Samali A (2006) The role of heat shock proteins in the regulation of apoptosis. In: Stress response: a molecular biology approach. Research Signpost, Trivandrum, pp 1–25
Gimbrone MA Jr, Topper JN, Nagel T, Anderson KR, Garcia-Cardeña G (2000) Endothelial dysfunction, hemodynamic forces, and Atherogenesisa. Ann N Y Acad Sci 902:230–240
Greenstein SM, Sun S, Calderon TM, Kim DY, Schreiber TC, Schechner RS, Tellis VA, Berman JW (2000) Mycophenolate mofetil treatment reduces atherosclerosis in the cholesterol-fed rabbit. J Surg Res 91:123–129
Guo Q, Wang Y, Xu D, Nossent J, Pavlos NJ, Xu J (2018) Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res 6:1–14
Hammerer-Lercher A, Mair J, Bonatti J, Watzka SB, Puschendorf B, Dirnhofer S (2001) Hypoxia induces heat shock protein expression in human coronary artery bypass grafts. Cardiovasc Res 50:115–124
Hance MW, Dole K, Gopal U, Bohonowych JE, Jezierska-Drutel A, Neumann CA, Liu H, Garraway IP, Isaacs JS (2012) Secreted Hsp90 is a novel regulator of the epithelial to mesenchymal transition (EMT) in prostate cancer. J Biol Chem 287:37732–37744
Hansson GK (2001) Immune mechanisms in atherosclerosis. Arterioscler Thromb Vasc Biol 21:1876–1890
Hansson GK, Libby P (2006) The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol 6:508
Hargitai J, Lewis H, Boros I, Rácz TM, Fiser A, Kurucz I, Benjamin I, Vı́gh L, Pénzes Z, Csermely P, Latchman DS (2003) Bimoclomol, a heat shock protein co-inducer, acts by the prolonged activation of heat shock factor-1. Biochem Biophys Res Commun 307:689–695
Hecker JG, McGarvey M (2011) Heat shock proteins as biomarkers for the rapid detection of brain and spinal cord ischemia: a review and comparison to other methods of detection in thoracic aneurysm repair. Cell Stress Chaperones 16:119–131
Heneka MT, Gavrilyuk V, Landreth GE, O’Banion MK, Weinberg G, Feinstein DL (2003) Noradrenergic depletion increases inflammatory responses in brain: effects on IκB and Hsp70 expression. J Neurochem 85:387–398
Hitchon CA, El-Gabalawy HS (2004) Oxidation in rheumatoid arthritis. Arthritis Res Ther 6:265
Hong H, Wang Q, Li J, Liu H, Meng X, Zhang H (2019) Aging, cancer and immunity. J Cancer 10:3021
Hooper PL, Hooper JJ (2005) Loss of defense against stress: diabetes and heat shock proteins. Diabetes Technol Ther 7:204–208
Hooper PL, Hooper PL (2009) Inflammation, heat shock proteins, and type 2 diabetes. Cell Stress Chaperones 14:113–115
Hooper PL, Balogh G, Rivas E, Kavanagh K, Vigh L (2014) The importance of the cellular stress response in the pathogenesis and treatment of type 2 diabetes. Cell Stress Chaperones 19:447–464
Hu W, Wang J, Luo G, Luo B, Wu C, Wang W, Xiao Y, Li J (2013) Proteomics-based analysis of differentially expressed proteins in the CXCR1-knockdown gastric carcinoma MKN45 cell line and its parental cell. Acta Biochim Biophys Sin 45:857–866
Huang M-N, Yu H, Moudgil KD (2010) The involvement of heat-shock proteins in the pathogenesis of autoimmune arthritis: a critical appraisal. Semin Arthritis Rheum 40:164–175
Ikwegbue PC, Masamba P, Oyinloye BE, Kappo AP (2018) Roles of heat shock proteins in apoptosis, oxidative stress, human inflammatory diseases, and cancer. Pharmaceuticals 11:2
Jafarnejad A, Bathaie SZ, Nakhjavani M, Hassan MZ, Banasadegh S (2008) The improvement effect of L-Lys as a chemical chaperone on STZ-induced diabetic rats, protein structure and function. Diabetes Metab Res Rev 24:64–73
Jindal S (1996a) Heat shock proteins: applications in health and disease. Trends Biotechnol 14:17–20
Johnson AD, Berberian PA, Tytell M, Bond MG (1993) Atherosclerosis alters the localization of Hsp70 in human and macaque aortas. Exp Mol Pathol 58:155–168
Johnson AD, Berberian PA, Tytell M, Bond MG (1995) Differential distribution of 70-kD heat shock protein in atherosclerosis: its potential role in arterial SMC survival. Arterioscler Thromb Vasc Biol 15:27–36
Joly A-L, Wettstein G, Mignot G, Ghiringhelli F, Garrido C (2010) Dual role of heat shock proteins as regulators of apoptosis and innate immunity. J Innate Immun 2:238–247
Kakimura J, Kitamura Y, Takata K, Umeki M, Suzuki S, Shibagaki K, Taniguchi T, Nomura Y, Gebicke-Haerter PJ, Smith MA, Perry G, Shimohama S (2002) Microglial activation and amyloid-beta clearance induced by exogenous heat-shock proteins. FASEB J 16:601–603
Kalia LV, Lang AE (2015) Parkinson’s disease. Lancet 386:896–912
Kalia LV, Kalia SK, McLean PJ, Lozano AM, Lang AE (2013) α-Synuclein oligomers and clinical implications for Parkinson disease. Ann Neurol 73:155–169
Kalmar B, Novoselov S, Gray A, Cheetham ME, Margulis B, Greensmith L (2008) Late stage treatment with arimoclomol delays disease progression and prevents protein aggregation in the SOD1 mouse model of ALS. J Neurochem 107:339–350
Kang EH, Kim DJ, Lee EY, Lee YJ, Lee EB, Song YW (2009) Downregulation of heat shock protein 70 protects rheumatoid arthritis fibroblast-like synoviocytes from nitric oxide-induced apoptosis. Arthritis Res Ther 11:R130
Kanwar RK, Kanwar JR, Wang D, Ormrod DJ, Krissansen GW (2001) Temporal expression of heat shock proteins 60 and 70 at lesion-prone sites during atherogenesis in ApoE-deficient mice. Arterioscler Thromb Vasc Biol 21:1991–1997
Karlsson-Parra A, Soderstrom K, Ferm M, Ivanyi J, Kiessling R, Klareskog L (1990) Presence of human 65 kD heat shock protein (hsp) in inflamed joints and subcutaneous nodules of RA patients. Scand J Immunol 31:283–288
Kaul G, Thippeswamy H (2011) Role of heat shock proteins in diseases and their therapeutic potential. Indian J Microbiol 51:124–131
Kendal WS (2008) Dying with cancer: the influence of age, comorbidity, and cancer site. Cancer 112:1354–1362
Kern J, Untergasser G, Zenzmaier C, Sarg B, Gastl G, Gunsilius E, Steurer M (2009) GRP-78 secreted by tumor cells blocks the antiangiogenic activity of bortezomib. Blood 114:3960–3967
Khafaga AF, Noreldin AE, Taha AE (2019) The adaptogenic anti-ageing potential of resveratrol against heat stress-mediated liver injury in aged rats: role of Hsp70 and NF-kB signalling. J Therm Biol 83:8–21
Kim HJ, Hwang NR, Lee KJ (2007) Heat shock responses for understanding diseases of protein denaturation. Mol Cells 23:123–131
Klucken J, Shin Y, Masliah E, Hyman BT, McLean PJ (2004) Hsp70 reduces α-synuclein aggregation and toxicity. J Biol Chem 279:25497–25502
Kovalchin JT, Wang R, Wagh MS, Azoulay J, Sanders M, Chandawarkar RY (2006) In vivo delivery of heat shock protein 70 accelerates wound healing by up-regulating macrophage-mediated phagocytosis. Wound Repair Regen 14:129–137
Landry J, Bernier D, Chretien P, Nicole LM, Tanguay RM, Marceau N (1982) Synthesis and degradation of heat shock proteins during development and decay of thermotolerance. Cancer Res 42:2457–2461
Leonardi GC, Accardi G, Monastero R, Nicoletti F, Libra M (2018) Ageing: from inflammation to cancer. Immun Ageing 15:1
Lewthwaite J, Owen N, Coates A, Henderson B, Steptoe A (2002) Circulating human heat shock protein 60 in the plasma of British civil servants: relationship to physiological and psychosocial stress. Circulation 106:196–201
Li J, Hu W, Lan Q (2012) The apoptosis-resistance in t-AUCB-treated glioblastoma cells depends on activation of Hsp27. J Neuro-Oncol 110:187–194
Liao WC, Wu MS, Wang HP, Tien YW, Lin JT (2009) Serum heat shock protein 27 is increased in chronic pancreatitis and pancreatic carcinoma. Pancreas 38:422–426
Libby P (2012) Inflammation in atherosclerosis. Arterioscler Thromb Vasc Biol 32:2045–2051
Lin S-P, Lee Y-T, Wang J-Y, Miller SA, Chiou S-H, Hung M-C, Hung S-C (2012) Survival of cancer stem cells under hypoxia and serum depletion via decrease in PP2A activity and activation of p38-MAPKAPK2-Hsp27. PLoS One 7:e49605
Liu FT, Chen Y, Yang YJ, Yang L, Yu M, Zhao J, Wu JJ, Huang F, Liu W, Ding ZT, Wang J (2015) Involvement of mortalin/GRP75/mthsp70 in the mitochondrial impairments induced by A53T mutant α-synuclein. Brain Res 1604:52–61
Lundy SK, Sarkar S, Tesmer LA, Fox DA (2007) Cells of the synovium in rheumatoid arthritis. T lymphocytes. Arthritis Res Ther 9:202
Macario AJL, De Macario EC (2001) Molecular chaperones and age-related degenerative disorders. In: Advances in cell aging and gerontology. Adv Cell Aging Gerontol 7:131–162. https://doi.org/10.1016/S1566-3124(01)07018-3
Madrigal-Matute J, López-Franco O, Blanco-Colio LM, Muñoz-García B, Ramos-Mozo P, Ortega L, Egido J, Martín-Ventura JL (2010) Heat shock protein 90 inhibitors attenuate inflammatory responses in atherosclerosis. Cardiovasc Res 86:330–337
Mak SK, McCormack AL, Manning-Bog AB, Cuervo AM, Di Monte DA (2010) Lysosomal degradation of α-synuclein in vivo. J Biol Chem 285:13621–13629
Marino Gammazza A, Bavisotto CC, Barone R, de Macario EC, Macario AJ (2016) Alzheimer’s disease and molecular chaperones: current knowledge and the future of chaperonotherapy. Curr Pharm Des 22:4040–4049
Martin-Ventura JL, Duran MC, Blanco-Colio LM, Meilhac O, Leclercq A, Michel J-B, Jensen ON, Hernandez-Merida S, Tuñón J, Vivanco F (2004) Identification by a differential proteomic approach of heat shock protein 27 as a potential marker of atherosclerosis. Circulation 110:2216–2219
Martin-Ventura JL, Leclercq A, Blanco-Colio LM, Egido J, Rossignol P, Meilhac O, Michel J-B (2007) Low plasma levels of Hsp70 in patients with carotid atherosclerosis are associated with increased levels of proteolytic markers of neutrophil activation. Atherosclerosis 194:334–341
Matzinger P (2002) An innate sense of danger. Ann N Y Acad Sci 961:341–342
Mayer M, Bukau B (2005) Hsp70 chaperones: cellular functions and molecular mechanism. Cell Mol Life Sci 62:670
Mayerl C, Lukasser M, Sedivy R, Niederegger H, Seiler R, Wick G (2006) Atherosclerosis research from past to present—on the track of two pathologists with opposing views, Carl von Rokitansky and Rudolf Virchow. Virchows Arch 449:96–103
McLean PJ, Kawamata H, Shariff S, Hewett J, Sharma N, Ueda K, Breakefield XO, Hyman BT (2002) TorsinA and heat shock proteins act as molecular chaperones: suppression of α-synuclein aggregation. J Neurochem 83:846–854
Mehana E-SE, Khafaga AF, El-Blehi SS (2019) The role of matrix metalloproteinases in osteoarthritis pathogenesis: an updated review. Life Sci 234:116786
Merendino AM, Bucchieri F, Campanella C, Marciano V, Ribbene A, David S, Zummo G, Burgio G, Corona DF, Conway de Macario E, Macario AJ, Cappello F (2010a) Hsp60 is actively secreted by human tumor cells. PLoS One 5:e9247
Merendino AM, Bucchieri F, Campanella C, Marcianò V, Ribbene A, David S, Zummo G, Burgio G, Corona DF, de Macario EC (2010b) Hsp60 is actively secreted by human tumor cells. PLoS One 5:e9247
Min JN, Whaley RA, Sharpless NE, Lockyer P, Portbury AL, Patterson C (2008) CHIP deficiency decreases longevity, with accelerated aging phenotypes accompanied by altered protein quality control. Mol Cell Biol 28:4018–4025
Moloney TC, Hyland R, O’Toole D, Paucard A, Kirik D, O’Doherty A, Gorman AM, Dowd E (2014) Heat shock protein 70 reduces α-synuclein-induced predegenerative neuronal dystrophy in the α-synuclein viral gene transfer rat model of Parkinson’s disease. CNS Neurosci Ther 20:50–58
Molvarec A, Prohaszka Z, Nagy B, Szalay J, Fust G, Karadi I, Rigo J Jr (2006) Association of elevated serum heat-shock protein 70 concentration with transient hypertension of pregnancy, preeclampsia and superimposed preeclampsia: a case-control study. J Hum Hypertens 20:780–786
Morrow G, Samson M, Michaud S, Tanguay RM (2004) Overexpression of the small mitochondrial Hsp22 extends Drosophila life span and increases resistance to oxidative stress. FASEB J 18:598–599
Mortaz E, Redegeld FA, Nijkamp FP, Wong HR, Engels F (2006) Acetylsalicylic acid-induced release of Hsp70 from mast cells results in cell activation through TLR pathway. Exp Hematol 34:8–18
Murphy KE, Gysbers AM, Abbott SK, Spiro AS, Furuta A, Cooper A, Garner B, Kabuta T, Halliday GM (2015) Lysosomal-associated membrane protein 2 isoforms are differentially affected in early Parkinson’s disease. Mov Disord 30:1639–1647
Mycko MP, Papoian R, Boschert U, Raine CS, Selmaj KW (2004) Microarray gene expression profiling of chronic active and inactive lesions in multiple sclerosis. Clin Neurol Neurosurg 106:223–229
Nagai Y, Fujikake N, Popiel HA, Wada K (2010) Induction of molecular chaperones as a therapeutic strategy for the polyglutamine diseases. Curr Pharm Biotechnol 11:188–197
Nillegoda NB, Bukau B (2015) Metazoan Hsp70-based protein disaggregases: emergence and mechanisms. Front Mol Biosci 2:57
Njemini R, Lambert M, Demanet C, Mets T (2003) Elevated serum heat-shock protein 70 levels in patients with acute infection: use of an optimized enzyme-linked immunosorbent assay. Scand J Immunol 58:664–669
O’Callaghan-Sunol C, Gabai VL, Sherman MY (2007) Hsp27 modulates p53 signaling and suppresses cellular senescence. Cancer Res 67:11779–11788
Oglesbee MJ, Herdman AV, Passmore GG, Hoffman WH (2005) Diabetic ketoacidosis increases extracellular levels of the major inducible 70-kDa heat shock protein. Clin Biochem 38:900–904
Oroz J, Kim JH, Chang BJ, Zweckstetter M (2017) Mechanistic basis for the recognition of a misfolded protein by the molecular chaperone Hsp90. Nat Struct Mol Biol 24:407–413
Pandey P, Saleh A, Nakazawa A, Kumar S, Srinivasula SM, Kumar V, Weichselbaum R, Nalin C, Alnemri ES, Kufe D (2000) Negative regulation of cytochrome c-mediated oligomerization of Apaf-1 and activation of procaspase-9 by heat shock protein 90. EMBO J 19:4310–4322
Park HS, Lee JS, Huh SH, Seo JS, Choi EJ (2001) Hsp72 functions as a natural inhibitory protein of c-Jun N-terminal kinase. EMBO J 20:446–456
Parodi J, Sepulveda FJ, Roa J, Opazo C, Inestrosa NC, Aguayo LG (2010) Beta-amyloid causes depletion of synaptic vesicles leading to neurotransmission failure. J Biol Chem 285:2506–2514
Parrinello S, Samper E, Krtolica A, Goldstein J, Melov S, Campisi J (2003) Oxygen sensitivity severely limits the replicative lifespan of murine fibroblasts. Nat Cell Biol 5:741–747
Pockley AG (2002) Heat shock proteins, inflammation, and cardiovascular disease. Circulation 105:1012–1017
Pockley AG (2003) Heat shock proteins as regulators of the immune response. Lancet 362:469–476
Pockley AG, Multhoff G (2008) Cell stress proteins in extracellular fluids: friend or foe? Novartis Found Symp 291:86–95; discussion 96–100, 137–140
Pockley AG, Wu R, Lemne C, Kiessling R, de Faire U, Frostegård J (2000) Circulating heat shock protein 60 is associated with early cardiovascular disease. Hypertension 36:303–307
Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, Pike B, Root H, Rubenstein J, Boyer R, Stenroos ES, Chandrasekharappa S, Athanassiadou A, Papapetropoulos T, Johnson WG, Lazzarini AM, Duvoisin RC, Di Iorio G, Golbe LI, Nussbaum RL (1997) Mutation in the α-synuclein gene identified in families with Parkinson’s disease. Science 276:2045–2047
Pratico D (2008) Oxidative stress hypothesis in Alzheimer’s disease: a reappraisal. Trends Pharmacol Sci 29:609–615
Quigney DJ, Gorman AM, Samali A (2003) Heat shock protects PC12 cells against MPP+ toxicity. Brain Res 993:133–139
Radak Z, Chung HY, Naito H, Takahashi R, Jung KJ, Kim HJ, Goto S (2004) Age-associated increase in oxidative stress and nuclear factor kappaB activation are attenuated in rat liver by regular exercise. FASEB J 18:749–750
Radli M, Rudiger SGD (2018) Dancing with the Diva: Hsp90-Client interactions. J Mol Biol 430:3029–3040
Raska M, Weigl E (2005) Heat shock proteins in autoimmune diseases. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 149:243–249
Rayner K, Chen Y-X, McNulty M, Simard T, Zhao X, Wells DJ, de Belleroche J, O’Brien ER (2008) Extracellular release of the atheroprotective heat shock protein 27 is mediated by estrogen and competitively inhibits acLDL binding to scavenger receptor-A. Circ Res 103:133–141
Rocnik E, Chow LH, Pickering JG (2000) Heat shock protein 47 is expressed in fibrous regions of human atheroma and is regulated by growth factors and oxidized low-density lipoprotein. Circulation 101:1229–1233
Rodríguez-Iturbe B, Johnson R (2018) Heat shock proteins and cardiovascular disease. Physiol Int 105:19–37
Ruparelia N, Chai JT, Fisher EA, Choudhury RP (2017) Inflammatory processes in cardiovascular disease: a route to targeted therapies. Nat Rev Cardiol 14:133
Saadeldin IM, Swelum AA, Elsafadi M, Mahmood A, Alfayez M, Alowaimer AN (2018) Differences between the tolerance of camel oocytes and cumulus cells to acute and chronic hyperthermia. J Therm Biol 74:47–54
Saadeldin IM, Swelum AA, Noreldin AE, Tukur HA, Abdelazim AM, Abomughaid MM, Alowaimer AN (2019a) Isolation and culture of skin-derived differentiated and stem-like cells obtained from the Arabian camel (Camelus dromedarius). Animals 9:378
Saadeldin IM, Swelum AA, Tukur HA, Alowaimer AN (2019b) Thermotolerance of camel (Camelus dromedarius) somatic cells affected by the cell type and the dissociation method. Environ Sci Pollut Res Int 26:29490–29496
Saadeldin IM, Abdel-Aziz Swelum A, Elsafadi M, Mahmood A, Osama A, Shikshaky H, Alfayez M, Alowaimer AN, Magdeldin S (2020a) Thermotolerance and plasticity of camel somatic cells exposed to acute and chronic heat stress. J Adv Res 22:105–118
Saadeldin IM, Swelum AA, Zakri AM, Tukur HA, Alowaimer AN (2020b) Effects of acute hyperthermia on the Thermotolerance of cow and sheep skin-derived fibroblasts. Animals 10:545
Salimi S, Shardell MD, Seliger SL, Bandinelli S, Guralnik JM, Ferrucci L (2018) Inflammation and trajectory of renal function in community-dwelling older adults. J Am Geriatr Soc 66:804–811
Salminen A, Paimela T, Suuronen T, Kaarniranta K (2008) Innate immunity meets with cellular stress at the IKK complex: regulation of the IKK complex by Hsp70 and Hsp90. Immunol Lett 117:9–15
Samali A, Cai J, Zhivotovsky B, Jones DP, Orrenius S (1999) Presence of a pre-apoptotic complex of pro-caspase-3, Hsp60 and Hsp10 in the mitochondrial fraction of Jurkat cells. EMBO J 18:2040–2048
Schett G, Redlich K, Xu Q, Bizan P, Groger M, Tohidast-Akrad M, Kiener H, Smolen J, Steiner G (1998) Enhanced expression of heat shock protein 70 (hsp70) and heat shock factor 1 (HSF1) activation in rheumatoid arthritis synovial tissue. Differential regulation of hsp70 expression and hsf1 activation in synovial fibroblasts by proinflammatory cytokines, shear stress, and antiinflammatory drugs. J Clin Invest 102:302–311
Schlesinger MJ (1990) Heat shock proteins. J Biol Chem 265:12111–12114
Schreibelt G, van Horssen J, van Rossum S, Dijkstra CD, Drukarch B, de Vries HE (2007) Therapeutic potential and biological role of endogenous antioxidant enzymes in multiple sclerosis pathology. Brain Res Rev 56:322–330
Sedlackova L, Sosna A, Vavrincova P, Frydl J, Guerriero V, Raynes DA, Hromadnikova I (2011) Heat shock protein gene expression profile may differentiate between rheumatoid arthritis, osteoarthritis, and healthy controls. Scand J Rheumatol 40:354–357
Seibert TA, Hibbert B, Chen Y-X, Rayner K, Simard T, Hu T, Cuerrier CM, Zhao X, de Belleroche J, Chow BJ (2013) Serum heat shock protein 27 levels represent a potential therapeutic target for atherosclerosis: observations from a human cohort and treatment of female mice. J Am Coll Cardiol 62:1446–1454
Shimp SK 3rd, Chafin CB, Regna NL, Hammond SE, Read MA, Caudell DL, Rylander M, Reilly CM (2012) Heat shock protein 90 inhibition by 17-DMAG lessens disease in the MRL/lpr mouse model of systemic lupus erythematosus. Cell Mol Immunol 9:255–266
Siebelt M, Jahr H, Groen HC, Sandker M, Waarsing JH, Kops N, Muller C, van Eden W, de Jong M, Weinans H (2013) Hsp90 inhibition protects against biomechanically induced osteoarthritis in rats. Arthritis Rheum 65:2102–2112
Singleton AB, Farrer M, Johnson J, Singleton A, Hague S, Kachergus J, Hulihan M, Peuralinna T, Dutra A, Nussbaum R, Lincoln S, Crawley A, Hanson M, Maraganore D, Adler C, Cookson MR, Muenter M, Baptista M, Miller D, Blancato J, Hardy J, Gwinn-Hardy K (2003) α-Synuclein locus triplication causes Parkinson’s disease. Science 302:841
Smolen JS, Breedveld FC, Burmester GR, Bykerk V, Dougados M, Emery P, Kvien TK, Navarro-Compan MV, Oliver S, Schoels M, Scholte-Voshaar M, Stamm T, Stoffer M, Takeuchi T, Aletaha D, Andreu JL, Aringer M, Bergman M, Betteridge N, Bijlsma H, Burkhardt H, Cardiel M, Combe B, Durez P, Fonseca JE, Gibofsky A, Gomez-Reino JJ, Graninger W, Hannonen P, Haraoui B, Kouloumas M, Landewe R, Martin-Mola E, Nash P, Ostergaard M, Ostor A, Richards P, Sokka-Isler T, Thorne C, Tzioufas AG, van Vollenhoven R, de Wit M, van der Heijde D (2016) Treating rheumatoid arthritis to target: 2014 update of the recommendations of an international task force. Ann Rheum Dis 75:3–15
Specht HM, Ahrens N, Blankenstein C, Duell T, Fietkau R, Gaipl US, Gunther C, Gunther S, Habl G, Hautmann H, Hautmann M, Huber RM, Molls M, Offner R, Rodel C, Rodel F, Schutz M, Combs SE, Multhoff G (2015) Heat shock protein 70 (Hsp70) peptide activated natural killer (NK) cells for the treatment of patients with non-small cell lung Cancer (NSCLC) after Radiochemotherapy (RCTx) – from preclinical studies to a clinical phase II trial. Front Immunol 6:162
Steinkraus KA, Smith ED, Davis C, Carr D, Pendergrass WR, Sutphin GL, Kennedy BK, Kaeberlein M (2008) Dietary restriction suppresses proteotoxicity and enhances longevity by an hsf-1-dependent mechanism in Caenorhabditis elegans. Aging Cell 7:394–404
Stellas D, Karameris A, Patsavoudi E (2007) Monoclonal antibody 4C5 immunostains human melanomas and inhibits melanoma cell invasion and metastasis. Clin Cancer Res 13:1831–1838
Takayama S, Reed JC, Homma S (2003) Heat-shock proteins as regulators of apoptosis. Oncogene 22:9041
Terry DF, Wyszynski DF, Nolan VG, Atzmon G, Schoenhofen EA, Pennington JY, Andersen SL, Wilcox MA, Farrer LA, Barzilai N, Baldwin CT, Asea A (2006) Serum heat shock protein 70 level as a biomarker of exceptional longevity. Mech Ageing Dev 127:862–868
Tetzlaff JE, Putcha P, Outeiro TF, Ivanov A, Berezovska O, Hyman BT, McLean PJ (2008) CHIP targets toxic α-synuclein oligomers for degradation. J Biol Chem 283:17962–17968
Thompson RC, Allam AH, Lombardi GP, Wann LS, Sutherland ML, Sutherland JD, Soliman MA-T, Frohlich B, Mininberg DT, Monge JM (2013) Atherosclerosis across 4000 years of human history: the Horus study of four ancient populations. Lancet 381:1211–1222
Thuringer D, Jego G, Wettstein G, Terrier O, Cronier L, Yousfi N, Hébrard S, Bouchot A, Hazoumé A, Joly A-L (2013) Extracellular Hsp27 mediates angiogenesis through toll-like receptor 3. FASEB J 27:4169–4183
Tomihara K, Curiel TJ, Zhang B (2013) Optimization of immunotherapy in elderly cancer patients. Crit Rev Oncog 18:573–583
Tsuneki M, Maruyama S, Yamazaki M, Xu B, Essa A, Abé T, Babkair H, Cheng J, Yamamoto T, Saku T (2013) Extracellular heat shock protein A9 is a novel interaction partner of podoplanin in oral squamous cell carcinoma cells. Biochem Biophys Res Commun 434:124–130
Tsutsumi S, Neckers L (2007) Extracellular heat shock protein 90: a role for a molecular chaperone in cell motility and cancer metastasis. Cancer Sci 98:1536–1539
Tytell M, Hooper PL (2001) Heat shock proteins: new keys to the development of cytoprotective therapies. Expert Opin Ther Targets 5:267–287
Urushitani M, Kurisu J, Tateno M, Hatakeyama S, Nakayama K, Kato S, Takahashi R (2004) CHIP promotes proteasomal degradation of familial ALS-linked mutant SOD1 by ubiquitinating Hsp/Hsc70. J Neurochem 90:231–244
Vabulas RM, Ahmad-Nejad P, da Costa C, Miethke T, Kirschning CJ, Hacker H, Wagner H (2001) Endocytosed Hsp60s use toll-like receptor 2 (TLR2) and TLR4 to activate the toll/interleukin-1 receptor signaling pathway in innate immune cells. J Biol Chem 276:31332–31339
van Roon JA, van Eden W, van Roy JL, Lafeber FJ, Bijlsma JW (1997) Stimulation of suppressive T cell responses by human but not bacterial 60-kD heat-shock protein in synovial fluid of patients with rheumatoid arthritis. J Clin Invest 100:459–463
Veereshwarayya V, Kumar P, Rosen KM, Mestril R, Querfurth HW (2006) Differential effects of mitochondrial heat shock protein 60 and related molecular chaperones to prevent intracellular beta-amyloid-induced inhibition of complex IV and limit apoptosis. J Biol Chem 281:29468–29478
von Vietinghoff S, Koltsova EK, Mestas J, Diehl CJ, Witztum JL, Ley K (2011) Mycophenolate mofetil decreases atherosclerotic lesion size by depression of aortic T-lymphocyte and interleukin-17–mediated macrophage accumulation. J Am Coll Cardiol 57:2194–2204
Walker GA, Lithgow GJ (2003) Lifespan extension in C. elegans by a molecular chaperone dependent upon insulin-like signals. Aging Cell 2:131–139
Wang X, Song X, Zhuo W, Fu Y, Shi H, Liang Y, Tong M, Chang G, Luo Y (2009) The regulatory mechanism of Hsp90α secretion and its function in tumor malignancy. Proc Natl Acad Sci U S A 106:21288–21293
Weiss YG, Bromberg Z, Raj N, Raphael J, Goloubinoff P, Ben-Neriah Y, Deutschman CS (2007) Enhanced heat shock protein 70 expression alters proteasomal degradation of IkappaB kinase in experimental acute respiratory distress syndrome. Crit Care Med 35:2128–2138
Wendling U, Paul L, van der Zee R, Prakken B, Singh M, van Eden W (2000) A conserved mycobacterial heat shock protein (hsp) 70 sequence prevents adjuvant arthritis upon nasal administration and induces IL-10-producing T cells that cross-react with the mammalian self-hsp70 homologue. J Immunol 164:2711–2717
Whitesell L, Lindquist SL (2005) Hsp90 and the chaperoning of cancer. Nat Rev Cancer 5:761
Whitham M, Febbraio MA (2010) HSP and diabetes. In: Asea AAA, Pedersen BK (eds) Heat shock proteins and whole body physiology. Springer Netherlands, Dordrecht, pp 3–18
Wick C (2016) Tolerization against atherosclerosis using heat shock protein 60. Cell Stress Chaperones 21:201–211
Williams RS (2000) Heat shock protein 47: a chaperone for the fibrous cap? Circulation 101:1227–1228. https://doi.org/10.1161/01.cir.101.11.1227
Wojdasiewicz P, Poniatowski ŁA, Szukiewicz D (2014) The role of inflammatory and anti-inflammatory cytokines in the pathogenesis of osteoarthritis. Mediat Inflamm 2014:19
Worthington J, Rigby AS, MacGregor AJ, Silman AJ, Carthy D, Ollier WE (1993) Lack of association of increased antibody levels to mycobacterial hsp65 with rheumatoid arthritis: results from a study of disease discordant twin pairs. Ann Rheum Dis 52:542–544
Wu YR, Wang CK, Chen CM, Hsu Y, Lin SJ, Lin YY, Fung HC, Chang KH, Lee-Chen GJ (2004) Analysis of heat-shock protein 70 gene polymorphisms and the risk of Parkinson’s disease. Hum Genet 114:236–241
Xiao Q, Mandal K, Schett G, Mayr M, Wick G, Oberhollenzer F, Willeit J, Kiechl S, Xu Q (2005) Association of serum-soluble heat shock protein 60 with carotid atherosclerosis: clinical significance determined in a follow-up study. Stroke 36:2571–2576
Xie F, Zhan R, Yan L-C, Gong J-B, Zhao Y, Ma J, Qian L-J (2016) Diet-induced elevation of circulating Hsp70 may trigger cell adhesion and promote the development of atherosclerosis in rats. Cell Stress Chaperones 21:907–914
Xilouri M, Brekk OR, Landeck N, Pitychoutis PM, Papasilekas T, Papadopoulou-Daifoti Z, Kirik D, Stefanis L (2013) Boosting chaperone-mediated autophagy in vivo mitigates α-synuclein-induced neurodegeneration. Brain 136:2130–2146
Xu Q, Wick G (1996) The role of heat shock proteins in protection and pathophysiology of the arterial wall. Mol Med Today 2:372–379
Xu L, Chen S, Bergan R (2006) MAPKAPK2 and Hsp27 are downstream effectors of p38 MAP kinase-mediated matrix metalloproteinase type 2 activation and cell invasion in human prostate cancer. Oncogene 25:2987
Xu Q, Metzler B, Jahangiri M, Mandal K (2011) Molecular chaperones and heat shock proteins in atherosclerosis. Am J Phys Heart Circ Phys 302:H506–H514
Yan MH, Wang X, Zhu X (2013) Mitochondrial defects and oxidative stress in Alzheimer disease and Parkinson disease. Free Radic Biol Med 62:90–101
Yap H-Y, Tee SZ-Y, Wong MM-T, Chow S-K, Peh S-C, Teow S-Y (2018) Pathogenic role of immune cells in rheumatoid arthritis: implications in clinical treatment and biomarker development. Cell 7:161
Yu L, Yuan X, Wang D, Barakat B, Williams E, Hannigan G (2014) Selective regulation of p38β protein and signaling by integrin-linked kinase mediates bladder cancer cell migration. Oncogene 33:690–701
Zhang X, He M, Cheng L, Chen Y, Zhou L, Zeng H, Pockley AG, Hu FB, Wu T (2008) Elevated heat shock protein 60 levels are associated with higher risk of coronary heart disease in Chinese. Circulation 118:2687–2693
Zhang X, Xu Z, Zhou L, Chen Y, He M, Cheng L, Hu FB, Tanguay RM, Wu T (2010) Plasma levels of Hsp70 and anti-Hsp70 antibody predict risk of acute coronary syndrome. Cell Stress Chaperones 15:675–686
Zhang X, Wang X, Zhu H, Kranias EG, Tang Y, Peng T, Chang J, Fan G-C (2012) Hsp20 functions as a novel cardiokine in promoting angiogenesis via activation of VEGFR2. PLoS One 7:e32765
Zhang S, Hu Y, Huang Y, Xu H, Wu G, Dai H (2015) Heat shock protein 27 promotes cell proliferation through activator protein-1 in lung cancer. Oncol Lett 9:2572–2576
Zhao M, Shen F, Yin Y, Yang Y, Xiang D, Chen Q (2012) Increased expression of heat shock protein 27 correlates with peritoneal metastasis in epithelial ovarian cancer. Reprod Sci 19:748–753
Zhu J, Quyyumi AA, Wu H, Csako G, Rott D, Zalles-Ganley A, Ogunmakinwa J, Halcox J, Epstein SE (2003) Increased serum levels of heat shock protein 70 are associated with low risk of coronary artery disease. Arterioscler Thromb Vasc Biol 23:1055–1059
Ziegler TR, Ogden LG, Singleton KD, Luo M, Fernandez-Estivariz C, Griffith DP, Galloway JR, Wischmeyer PE (2005) Parenteral glutamine increases serum heat shock protein 70 in critically ill patients. Intensive Care Med 31:1079–1086
Acknowledgements
We would like to thank the Deanship of Scientific Research and RSSU at King Saud University for their technical support.
Disclosure of Interests
All authors declare they have no conflict of interest.
Ethical Approval for Studies Involving Humans
This article does not contain any studies with human participants performed by any of the authors.
Ethical Approval for Studies Involving Animals
This article does not contain any studies with animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Khafaga, A.F., Noreldin, A.E., Saadeldin, I.M. (2020). Role of HSP in the Pathogenesis of Age-Related Inflammatory Diseases. In: Asea, A.A.A., Kaur, P. (eds) Heat Shock Proteins in Inflammatory Diseases. Heat Shock Proteins, vol 22. Springer, Cham. https://doi.org/10.1007/7515_2020_29
Download citation
DOI: https://doi.org/10.1007/7515_2020_29
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-85167-5
Online ISBN: 978-3-030-85168-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)