Abstract
During the last few decades, contamination by anthropogenic chemicals such as persistent organic pollutants (POPs) has spread all over the world as evidenced by their detection in various environmental components and biota including those far from human activities. Particularly, research efforts on field observations and numerical models of global fate of POPs have revealed oceanic water bodies to be a global reservoir and final sink for these toxic contaminants that undergo transport from emission sources and partition between air and water and scavenge to deep-sea layers by various biogeochemical and geophysical processes. This chapter provides an overview of the contamination by POPs and related compounds in deep-sea ecosystems along frontal zones around Japan based on the results of the monitoring studies conducted by our laboratory during the last decade. In the chapter, we focus mainly on two regions, the western North Pacific (WNP), off-Tohoku, Japan, and the East China Sea (ECS). The WNP is a region influenced by various water masses and currents, making it one of the world’s most biologically productive zones. The other region discussed in the chapter, the ECS, is an epicontinental sea with lots of continental inputs. Our studies in these regions were conducted with the objective of understanding the environmental transport and distribution and the specific accumulation characteristics of organohalogen and butyltin compounds in deep-sea organisms. Our results suggest the vertical transport of POPs and related compounds in high productive waters along oceanic fronts and the potential role of deep-sea bed as a final sink and reservoir for these persistent contaminants. Further, to implement and evaluate the effectiveness of international agreements to protect the marine environment from the deleterious effects of POPs, interdisciplinary approaches including studies on biogeochemical and geophysical processes in the ocean as well as field observations are required to delineate the global and regional fate of POPs.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Over the past few decades, large amounts of organic and inorganic contaminants have been released into the environment as a consequence of the worldwide urbanization and agricultural and industrial activities. Marine ecosystems are no exception and they have also been increasingly subject to anthropogenic chemical contamination, receiving contaminants from a variety of diffuse and point sources. The contaminants include industrial chemicals inadvertently released into the environment, as well as those derived from land use activities such as agricultural chemicals applied on crops. One such group of contaminants is the persistent organic pollutants (POPs), comprising various well-known organohalogen contaminants such as polychlorinated biphenyls (PCBs), polychlorinated dibenzo-p-dioxins and furans (PCDD/Fs), and organochlorine pesticides like dichlorodiphenyltrichloroethane (DDT) and its stable metabolites, DDE and DDD (DDTs), chlordane-related compounds (CHLs), hexachlorobenzene (HCB), and hexachlorocyclohexanes (HCHs) (Fig. 1). Besides, certain brominated organic compounds have been considered for inclusion in the list of POPs, i.e., polybrominated diphenyl ethers (PBDEs) and hexabromocyclododecanes (HBCDs) (Fig. 1). These compounds, which are added to electrical and electronic equipment, paints, textiles and building materials as brominated flame retardants (BFRs), are an emerging class of contaminants. In addition to the above contaminants, aquatic pollution by butyltins (BTs), particularly toxic tributyltin (TBT), used as a biocide in antifouling paints for boats and aquaculture nets, has been of concern due to the bioaccumulative potential and deleterious effects of BTs in organisms [1].
Both the emerging and legacy POPs are toxic, are chemically stable, and therefore do not easily degrade in the environment or in organisms. These chemicals tend to partition between various environmental media, such as air, water, soil, sediment, and biota, depending on the physicochemical properties. Furthermore, being semi-volatile, these compounds are subject to co-distillation processes and can be transported globally through the atmosphere, which is well known as a concept of long-range atmospheric transport (LRAT) [2]. As a consequence, these substances have been detected in remote, relatively pristine locations, such as the polar regions and the deep seas. To protect human health and the environment, national and international control measures on the production and usage of POPs have been (or are being) introduced to reduce their emissions to the environment. For instance, the Stockholm Convention, negotiated under the support of the United Nations Environment Program (UNEP), established a globally binding framework initially targeting a group of POPs, informally called the “dirty dozen,” for reduction and eventual elimination [3]. The dirty dozen include several organochlorine pesticides (aldrin, chlordane, dieldrin, endrin, heptachlor, hexachlorobenzene, mirex, toxaphene, and DDT), PCBs, and PCDD/Fs. Besides the pesticide use, HCB was also used for industrial processes. PCBs were widely applied for industrial fluids in electrical equipment. PCDD/Fs and some PCB congeners (i.e., dioxin-like PCBs or coplanar PCBs) are formed as unintentional by-products during incineration and other thermal processes. The Stockholm Convention also includes procedures for identifying and adding substances to the POPs list. In May 2009, nine other POPs, including several BDE congeners present in PBDE commercial mixtures, have been listed under the Stockholm Convention [4]. More recently, HBCD has also been listed to Annex A of the Stockholm Convention at the sixth Conference of Parties (COP 6) in 2013 [5].
Despite a limited number of studies, significant contamination by POPs in deep-sea fishes has been demonstrated in the early 1980s [6–9]. Besides, in recent years, POPs, including BFRs, have been found at appreciable concentrations in deep-sea organisms from various parts of the world [10–28]. Research efforts by various scientific groups in the last few decades have greatly increased our understanding on the global distribution of POPs. The world’s oceans are thought to play an important role in the cycling and removal of POPs [29]. The oceans cover two-thirds of the Earth’s surface and because of their large volume can contain a large inventory of POPs [30]. The sources of POPs to the marine environment are riverine transport, municipal and industrial discharges, continental runoff, and atmospheric deposition in open waters. Atmospheric deposition delivers a large proportion of the POPs present in the oceans, through various mechanisms such as diffusive air–water exchange of POPs and wet and dry deposition processes [31]. Once in the aquatic environment, POPs can be dissolved in the water phase or partition onto colloidal and suspended/settling particulate matter, incorporate into food webs, transfer to the deep waters with the sinking particles, and eventually deposit in bottom sediments. It should be noted as an important role in the global behavior of POPs that the bottom sediments of deep-sea can either act as a final sink or a reservoir for such persistent contaminants. Sediment-sorbed xenobiotics can be taken up by the epibenthic and infaunal biota as they feed. A limited desorption of POPs, independent from that mediated by the bottom currents at the superficial level of the sediment, also occurs in the interstitial water [32]. These features can lead to remobilization of sediment-bound POPs in the entire food web of the deep-sea benthos. Further, the fate of POPs are affected by the hydrodynamics of water masses such as turbulence and advection of water masses and the differential characteristics of coasts with respect to the open sea, i.e., enhanced stratification due to freshwater input from rivers, influence of tides etc. Figure 2 is a conceptual diagram showing the key processes affecting the transport of POPs between the atmosphere, water column, and bottom sediments. Depending on the physicochemical properties of POPs (e.g., log Kaw), their global partition between environmental media and transport processes differ significantly among the compounds, congeners, or isomers [31, 33].
For the past four decades, our laboratory at the Center for Marine Environmental Studies (CMES), Ehime University, Japan, has been conducting research on the contamination status and spatial distribution, temporal trends, behavior, and fate of the above-listed contaminants in various matrices from different ecosystems (reviewed in Tanabe [34]; Tanabe et al. [35]; Tanabe and Ramu [36]). In the following chapter, we review some of our studies dealing with POPs in deep-sea organisms collected from various deep-sea environments. Although it is ideal to directly measure the levels of POPs in seawater to monitor the contamination status of POPs in marine ecosystems, there are limitations such as large volumes of seawater required for the analysis, sensitive analytical techniques crucial for detecting the low concentrations of POPs, and fluctuations of POP concentrations in seawater depending on the weather conditions. Therefore, in our studies, we often employ marine organisms like fish and shellfish as bioindicators to elucidate contamination status and spatial distribution of POPs in marine ecosystems. Aquatic organisms are very efficient in accumulating these contaminants, since, in addition to bioaccumulation through the diet, they are also subject to bioconcentration. Furthermore, data regarding levels and distributions of POPs in marine organisms, especially edible ones (and this includes an increasing number of deep-sea species), are important not only for assessing the state of the ecological environment but also from the human health perspective.
In the chapter, we focus mainly on two regions, the western North Pacific (WNP), off-Tohoku, Japan, and the East China Sea (ECS). The WNP is a region influenced by various water masses and currents making it one of the world’s most biologically productive zones. The other region discussed in the chapter, the ECS, is an epicontinental sea with lots of continental inputs. Both the regions are characterized by the formation of numerous oceanic fronts. Belkin [37] defined oceanic fronts as a narrow zone of enhanced horizontal gradients of water properties (temperature, salinity, nutrients, etc.) that separates broader areas with different water masses or different vertical structure. These fronts are mostly characterized by strong mixing, stirring, enhanced bioproductivity, and ecotones. High productivities around the fronts may enhance flux of POPs into the oceans and their transport to deep waters because of phytoplankton uptake and the vertical flux of the particles play important roles in the biogeochemical cycles of POPs in the oceans [38, 39]. In general, land- and open-ocean-derived materials tend to converge at the frontal zones. The formation of coastal fronts has been suggested to be an important oceanic phenomenon determining the behavior and flux as well as the fate of persistent contaminants in the marine environment [40].
Detailed information on the sampling and analytical methods can be found in the cited papers [14, 41–44]. The reviewed studies may help improve our insights into the contamination status and the fate of legacy POPs and emerging contaminants such as BFRs in the deep-sea environments.
2 Description of the Study Areas
2.1 The Western North Pacific, Off-Tohoku
The study area, the WNP, off-Tohoku, is characterized by a complex oceanographic structure represented by major ocean currents (Fig. 3). The Oyashio, a western boundary current of the Subarctic North Pacific, is a continuation of the East Kamchatka Current (EKC) and is fed by waters from the Western Subarctic Gyre and the Sea of Okhotsk [45]. The primary productivity of the Sea of Okhotsk is very high, especially on the continental shelf due to the relatively high insolation and the nutrient input from the Amur River and Pacific Ocean [46]. Recently, the dense shelf water flowing into Okhotsk Sea Mode Water (OSMW) has been considered as a potential source for exporting large amounts of organic matter and nutrients such as iron from the continental shelf to adjacent ocean interior along Oyashio [47, 48]. The eastward flowing Oyashio forms the Oyashio Front (OYF) which becomes Polar Front (PF) or Subarctic Front (SAF) to the east. The Kuroshio, a western boundary current from the southern tropic area, turns eastward from the eastern coast of Honshu, Japan. Then, warm and saline water is transported by this Kuroshio Extension [49]. The region between the Oyashio Front and the Kuroshio Extension Front is called the Kuroshio–Oyashio Transition Zone (KOTZ) or mixed water region, in which cold and warm waters mix and complex frontal structures are formed [49, 50].
A schematic circulation pattern of the major currents and frontal structures in the Kuroshio–Oyashio transition area, EKC East Kamchatka Current, ESC East Sakhalin Current, OY Oyashio, TWC Tsushima Warm Current, KE Kuroshio Extension, OYF Oyashio Front, PF Polar Front, SAF Subarctic Front, OSMW Okhotsk Sea Mode Water
Thus, the oceanography in the Tohoku area is complex and variable due to the confluence of various currents and water masses. Due to mixing of these water masses, the nutrient-rich subsurface water is brought into the euphotic zone promoting high phytoplankton production [51]. Marine biogeochemical activities including primary production, zooplankton grazing, microbial transformation, aggregation, and degradation processes of organic particles influence the flux of biogenic particles in the ocean. Vertical sinking of particle-associated pollutants such as PCBs may be enhanced by eutrophication since higher primary productivity leads to larger vertical fluxes of particles and organic carbon [38, 52]. This region known as Japan Trench is a depository of biogenic and lithogenic materials that are transported from the coastal shelf [53]. Thus, it can be assumed that a considerable proportion of anthropogenic contaminants may be transported into the deep water of North Pacific through the various biogeochemical processes.
2.2 The East China Sea
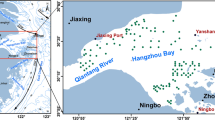
The ECS, located at midlatitudes between 25 and 35°N is an epicontinental sea surrounded by the Ryukyu archipelago, Japan, Korea, China, and Taiwan (Fig. 4). It has a broad continental shelf covering an area of 530 × 103 km2 [54]. The major western boundary current, the Kuroshio, runs along the outer edge of the continental shelf, enters the ECS through the strait between Taiwan and the westernmost island of the Ryukyu Islands, flows northeastward along the shelf slope, and exits to the Philippine Sea after turning eastward near 30°N [55]. Two of the largest rivers in the world, the Yangtze River (Changjiang) and the Yellow River, discharge into the ECS. Thus, the cold, freshwater distributed on the continental shelf and the warm saline Kuroshio water that occupies the area around the shelf water lead to the formation of salinity front near the continental shelf break.
A schematic circulation pattern of the major currents and frontal structures in the East China Sea. TsC Tsushima Current, TWC Taiwan Warm Current, WKCC Western Korea Cold Current, CDW Changjiang Diluted Water, ECSCoW East China Sea Coastal Water, YSCoW Yellow Sea Coastal Water, YSWW Yellow Sea Warm Water. Dashed lines indicate major shelf fronts in the East China Sea
The Kuroshio, the Yangtze River runoff, and the East Asia monsoons are the dominant factors affecting the circulation in the ECS. The ECS continental shelf circulation pattern is characterized by the Kuroshio, Tsushima Current, Taiwan Warm Current, and other coastal waters and shelf fronts [56, 57] (Fig. 4). It has been demonstrated that the Kuroshio strongly influences not only the circulation in the ECS shelf but also its chemistry through water-mass exchanges [56–58].
Riverine runoff is an important mode to transport anthropogenic pollutants from terrestrial sources to adjacent oceans. The Yangtze River flows through densely populated areas with agriculture and industrial activities along both the banks, and Shanghai, the largest city in China, is situated at its mouth. Discharge of industrial wastes, application of fertilizers, pesticides, and herbicides in farming, as well as heavy metal pollution are said to make the Yangtze River one of the most polluted rivers in the world [59]. Li and Daler [60] reported that the environmental pollution of the Yangtze River basin greatly influences the state of the marine environment of the ECS. Consequently, the ECS has attracted much interest as a site for the study of the fate of terrestrial material in the marine environment [61]. The ECS shelf receives a rich supply of nutrients from the Yangtze River and the upwelled Kuroshio subsurface waters. As a result, the ECS shelf is one of the most productive marginal seas in the world [62]. Thus, it is obvious that the ECS receives enormous amounts of anthropogenic pollutants, suspended matter, and nutrients with the riverine runoff. Furthermore, the intensive exchange between the shelf water and the Kuroshio and the high primary productivity observed in this region may facilitate the flux of persistent contaminants to the bottom of the ECS.
3 POPs and Related Compounds in the Western North Pacific, Off-Tohoku
This section provides a synopsis of our studies conducted in the WNP, off-Tohoku [14, 41, 44], with the objective of understanding the distribution and the specific accumulation characteristics of organohalogen and butyltin compounds in deep-sea organisms in this region.
3.1 Contamination Status
Organochlorine compounds (OCs) were detected in all the deep-sea organisms (e.g., deep-sea eels, grenadiers, cods, eelpouts, sculpins, bikumins, flounders, myctophids, lantern sharks, ratfishes, squids, octopus, shrimps, crabs, gastropods) collected during three different time periods (see the above references for more details). The concentrations of PCBs and DDTs were the highest among the OCs analyzed, and the concentrations of other compounds were in the following order CHLs > HCHs ≥ HCB. The predominant accumulation of PCBs and DDTs in deep-sea organisms collected from the WNP, off-Tohoku, agreed with those reported in shallow- and deep-water fishes collected from other locations along the Pacific coast of Japan [12,15] (Table 1). This reflects the higher bioaccumulative properties of PCBs and DDTs in marine food web as well as their significant usage.
Except for some higher trophic level organisms like snubnosed eels (Simenchelys parasitica), which contained some OCs at extremely high concentrations, the concentrations of OCs in deep-sea fish collected from the WNP, off-Tohoku, were generally lower than those in deep-sea organisms collected from other locations in the Atlantic Ocean, Mediterranean Sea, Ireland and Norwegian coasts, and the Arctic region [10, 11, 17–21, 25–27] (Table 2). The relatively low contamination by OCs in the present study area might be due to the smaller usage of these compounds in the WNP region compared to that of the North American and European countries. Besides, factors like variations in analytical methods and fish species (having different biological and ecological characters) may also influence the differences in the OC levels compared between the studies.
Comparing with studies carried out in adjoining areas on deep-sea fishes by our research group (Table 1), the concentrations of PCBs, DDTs, CHLs, and HCB in deep-sea fishes collected from the WNP, off-Tohoku, were comparable or lower than the data so far reported from Suruga and Tosa Bays [12, 15] and the ECS [42]. On the other hand, concentrations of HCHs in deep-sea fishes from this region were higher than those from other locations along the warm Kuroshio Current such as Tosa Bay and the ECS (Table 1). Higher concentrations of HCHs in cold waters along the Oyashio Current than in other offshore waters around Japan were also observed in a study using skipjack tuna (Katsuwonus pelamis) [63]. Due to the high vapor pressure, HCHs are known to rapidly evaporate and be transported from their pollution sources in the tropics and temperate regions to colder regions via the atmosphere [2, 30, 64]. The distribution patterns of HCHs found in the deep-sea organisms reflect the highly transportable nature of HCHs and its accumulation in the cold-water current of the WNP. No significant difference in the contamination status of HCHs between three research periods from 1994 to 2005 in this region (Table 1) is also suggestive of continuous flux of HCHs into the cold waters of the WNP region.
Among the three studies reviewed here, only the study by Takahashi et al. [44] reported the concentrations of PBDEs and HBCDs in deep-sea fishes collected from the western North Pacific, off-Tohoku, in 2005. The concentrations of PBDEs ranged from 1.3 to 8.5 ng/g with a mean of 3.6 ng/g lipid wt, while the concentrations of HBCDs ranged from 5.4 to 45 ng/g with a mean of 22 ng/g lipid wt (except for snubnosed eels). Despite the low levels compared to the other POPs, the detection of PBDEs and HBCDs in deep-sea fishes indicates the widespread presence of such “emerging POPs” even in deep oceans and their long-range transport. Similar to the result above, higher concentrations of HBCDs than PBDEs were observed in skipjack tuna collected from offshore waters around northern Japan [65, 66]. On the other hand, accumulation of PBDEs at higher concentrations than HBCDs was reported in fishes and cetaceans from East and South China Seas [35, 43]. Comparing to the concentration levels of PBDEs (5.1 and 19.9 ng/g lipid wt at mean concentrations) and HBCDs (<1.5 ng/g lipid wt) in two species of deep-sea grenadiers from the Mediterranean Sea [23], relatively high magnitude of contamination by HBCDs was noted in our data on deep-sea fish from the western North Pacific, off-Tohoku. Such differences among the studies and locations may reflect possible long-range transport of HBCDs to cold offshore waters, like HCHs, and/or significant usage of HBCDs (especially for thermal insulating materials) in northern Japan. Although HBCD is currently being proposed to be listed a POP in the Stockholm Convention, the contamination by this BFR in marine ecosystems around Japan may increase or continue in the meantime as its use or disposal, considering the increase in market demand for HBCD in Japan during late 1990s [67].
Concentrations of butyltin compounds (BTs) were reported by Takahashi et al. [14] and de Brito et al. [41]. Takahashi et al. [14] detected BTs in almost all myctophids analyzed (levels up to 46 ng/g wet wt for total BTs (∑BT: MBT + DBT + TBT)). Widespread contamination by BTs in deep-sea organisms from the western North Pacific, off-Tohoku (levels up to 530 ng/g wet wt in the liver of demon grenadier, Coelorinchus gilberti, for ∑BTs), was also reported by de Brito et al. [41]. Although the magnitude of contamination by BTs in open-water ecosystems of the western North Pacific seems to be relatively low, the results indicate the expansion of BT pollution in deep-sea ecosystems.
3.2 Species-Specific Accumulation and Trophic Magnification
Among various fish species analyzed, snubnosed eel (Fig. 5) accumulated hydrophobic organohalogens (with log Kow ~ 6–8) such as PCBs, DDTs, CHLs, and PBDEs at extremely high concentrations (Table 2). This species can be considered as an organism at a high trophic level in the food web due to its strong carnivorous and unique feeding habit; this fish has been found to burrow into the body of large fish such as halibut and shark and feed on their intestine and blood [68]. Relatively higher concentrations of these compounds were also found in some large carnivore and/or bottom-dwelling fishes. These observations suggest that accumulation of the hydrophobic OC and PBDEs is influenced by trophic magnification and/or feeding habits of organisms and their long biological half-lives, i.e., higher uptake rate via food and slower equilibrium between ambient water and body lipids. In contrast, HCHs, HCB, and HBCDs, which have less lipophilicity than the other organohalogen compounds, showed smaller variation in concentrations among species of different trophic levels and feeding habits.
Isotopic signatures based on the ratios of different stable isotopes of elements have been broadly applied to investigate a variety of processes. In particular, stable carbon and nitrogen isotope ratios (δ13C and δ15N) have been used in environmental studies for delineating carbon flows and trophic relationships in continental and marine ecosystems. The stable isotope ratios of nitrogen δ15N reflect diets and represents time-integrated averages of assimilated foods [69]; thus, stable nitrogen isotope analysis has been used to assess the relative trophic level of aquatic biota and provide a quantitative continuous variable for studying the trophic magnification of contaminants within complex food webs [70, 71]. In contrast, the ratios of stable carbon isotopes δ13C, in biota, can help to elucidate trophic interactions by establishing the relative contribution of marine (or pelagic) versus coastal (or benthic) carbon sources [72]. Thus, the quantification of both stable carbon and nitrogen isotope ratios can provide valuable information on the feeding ecology of biota and its potential influence on the trophic enrichment of contaminants [70, 73].
To understand the trophic magnification of organohalogen compounds, δ15N and δ13C were determined in fishes collected in 2005. Their correlations to the concentrations of organohalogen compounds were discussed [44]. A wide range of δ13C values among fishes was observed, implying the existence of multiple carbon sources in the food web of this region. Significant positive correlations were found between δ15N (‰) and lipid normalized concentrations of PCBs, DDTs, and PBDEs (p < 0.05) showing their high trophic magnification potential in marine food webs (Fig. 6). No such relationship was found for HCB, HCHs, and HBCDs (Fig. 6). The rapid elimination through gills to surrounding water and by excretion via other means may prevent the trophic magnification of these compounds. In addition, less trophic magnification observed for some organohalogen compounds in this study may be due to the smaller difference in trophic levels of fishes. In fact, there were no significant correlations between δ15N values and the concentrations of almost all the compounds analyzed when the data of snubnosed eels was excluded (only a significant relationship for PBDEs with p < 0.05). Thus, the accumulation profiles of organohalogen compounds in deep-sea fishes reflect not only their trophic transfer but also the different sources of the contaminants in the food web and/or species-specific distribution of fishes. Especially, some bottom-dwelling fishes including snubnosed eels showed significant lower δ13C values than the other fish species, reflecting influence of substances originated from sediments. Therefore, the data of high trophic bottom-dwelling fishes were excluded for further discussion on spatial and vertical distribution of contaminants.
A considerable variation was observed in the concentrations of BTs among tissues/organs and species. Among the tissues/organs analyzed, higher concentrations of BTs were found in the liver of fish (97 ± 212 ng ∑BT/g wet wt) and viscera of crabs (17 ± 17 ng ∑BT/g wet wt) [41]. Nevertheless, in the case of BTs, relatively high concentrations were observed not only in larger carnivore fishes (e.g., 9.7 ng ∑BT/g wet wt in the whole body of Pacific grenadier) but also in small fishes (up to 46 ng ∑BT/g wet wt in the whole body of myctophid fishes) and squids (up to 44 ng ∑BT/g wet wt in the whole body of Japanese hooked squid) [14, 41]. It is evident from previous studies that the bioaccumulation properties of BTs (i.e., organometallic compounds) were principally different from organohalogen compounds having hydrophobic and/or lipophilic nature. Less biomagnification of BTs was also documented in some food webs along Japanese coasts [74, 75].
3.3 Distribution in Relation to the Water-Mass Structure
Even among fishes showing similar trophic levels, considerable variation in the concentrations of POPs and related compounds was observed, which cannot be explained only by their feeding habits and trophic magnification. It has been documented that fishes can be used as suitable bioindicators to elucidate the environmental levels and distributions of persistent and lipophilic compounds in water bodies after careful consideration of their biological and ecological parameters [76].
Takahashi et al. [14] studied the accumulation profiles of OCs and BTs in relation to the sampling locations and diel vertical migration modes of myctophid species to delineate the spatial and vertical distribution of the contaminants in the WNP. While concentrations of OCs and BTs varied among species, no significant difference in the concentrations of fishes were observed between sampling locations (in the area: 38–41°N, 143–147°E). On the other hand, the accumulation patterns of contaminants showed a specific trend in accordance with the migration types (Fig. 7). Except for a migrant species D. theta, higher concentrations of PCBs, DDTs, and CHLs were found in non- or less-migratory species living in deeper waters. On the contrary, HCHs, HCB, and BTs accumulated at higher concentrations in migratory species as compared to nonmigratory fishes. A similar trend can also be suggested from mean concentrations of OCs and BTs in deep-sea fishes collected from the WNP, off-Tohoku [41] (Fig. 8). In general (except for snubnosed eels), higher concentrations of PCBs, DDTs, and CHLs were found in fishes from deeper waters (~1,000 m). On the contrary, HCHs, HCB, and BTs accumulated at higher concentrations in fishes from shallower waters.
Concentrations and residue patterns of PCBs, DDTs, HCHs, and BTs in myctophid fishes from the western North Pacific, off-Tohoku, with different migration types. Each bar indicates the concentration in a composite sample which was prepared from pooled and homogenized individual specimen belonging to the same species collected from the same sampling location [14]
These observations suggest the vertical distribution of these contaminants in the water column of this region. Tanabe and Tatsukawa [77] showed that the concentrations of HCHs significantly decreased with depth in the open-water column of the Pacific Ocean because of their volatility and high atmospheric mobility. Long-range atmospheric transport and cold condensation of volatile POPs, as noted in the above section, may enhance their flux to the surface waters in this WNP region. On the other hand, results of these studies are also suggestive of the vertical transport of hydrophobic OCs and their accumulation in benthic deep-sea organisms. OCs with lower water solubility and higher affinity for adsorption to particles would be transported to deeper waters and incorporated into the marine food web more readily than more water-soluble compounds. With regard to BTs, higher concentrations were found in shallow-water than deep-sea fishes. Recent input of BTs from the usage in surface waters (e.g., antifouling) could be well over the vertical transportation rate of these compounds, which results in higher levels in shallow waters.
In addition, the structure of the water mass around the WNP should be considered in evaluating the vertical profiles of contaminants. As noted in the description of the studied area, it has been suggested that the North Pacific Intermediate Water (NPIW), which is found at depths of 300–800 m in the North Pacific subtropical gyre, originates from the water mass of OSMW, and new NPIW is formed along the Kuroshio–Oyashio interfrontal zone [50]. The dense shelf water flowing into the OSMW has been considered as a potential source for exporting large amounts of organic matter and nutrients such as iron from the continental shelf to adjacent ocean interior along Oyashio. This means that organic pollutants as well as the shelf-originated nutrients can be transported from the continental shelf to NPIW. Despite limited data available, elevated background levels of OCs such as DDTs and PCBs in the Okhotsk Sea and East Siberia regions have been suggested by several studies on air and water [78, 79] and wild animals such as seals and sea eagles [80, 81]. The sampling depths and locations in the study of Takahashi et al. [14] and de Brito et al. [41] overlapped with the area where an intrusion of water mass from the Okhotsk Sea into the mesopelagic zone and the formation of NPIW occur. In this context, the intermediate water in the study area can be expected to have higher concentrations of OCs such as PCBs and DDTs that originate from more contaminated Okhotsk Sea.
3.4 Compositions and Temporal Trends of Organohalogen Compounds
Influence of the water-mass structure and material transport around the Okhotsk Sea and Oyashio current on the vertical profiles of OCs can be also found in the compositions of DDTs in deep-sea fishes from the WNP, off-Tohoku. Among the DDT compounds, p,p'-DDE and p,p'-DDT were the predominant constituents in almost all the deep-sea fishes (Figs. 9 and 10) [14, 44]. Similarly high proportions of p,p'-DDT (> 40%) were also found in Japanese common squid (Todarodes pacificus) from off-Tohoku, Japan, although the location is far away from any possible sources of DDT [82]. Such DDT composition (i.e., high proportions of a parent compound, p,p'-DDT) observed in fishes and squids from the WNP, off-Tohoku, is apparently different from those reported in previous studies from Suruga and Tosa Bays [12, 15] and in other recent researches from the Mediterranean Sea [25, 26] and North Atlantic Ocean [20, 27], where p,p'-DDE, a stable degradation compound, was dominant among DDTs in almost all the fishes analyzed. In this context, the intermediate water in the study area can be expected to have relatively fresh input of DDTs. Particularly in myctophids, higher proportions of p,p'-DDT were observed in nonmigratory fishes than in those of migratory ones (p < 0.05, Mann–Whitney U-test) (Fig. 9). This agrees with the concentration profile of DDTs as noted above.
Compositions of DDT compounds and HCH isomers in myctophid fishes from the western North Pacific, off-Tohoku. Each bar indicates the composition in a composite sample which was prepared from pooled and homogenized individual specimen belonging to the same species collected from the same sampling location [14]
Among the HCHs isomers, α-HCH was the predominant isomer in all the deep-sea fishes in the NWP, off-Tohoku (Figs. 9 and 10). It has been reported that the ratio of α-HCH to total HCH concentrations in seawater and fish has a tendency to increase with the increasing latitude [30, 63].α-HCH is preferentially transported to northern colder regions due to higher vapor pressure among the HCH isomers.
Regarding to the temporal change in the composition of OCs, the percentages of p,p'-DDT in myctophids collected in 1994 (~40%) was apparently lower than those in myctophids collected from Yaizu, Suruga Bay, in 1976 (58%) [83]. This suggests the reduction of fresh input of DDTs in the WNP during the last few decades. The decreasing p,p'-DDT proportion in total DDT compounds has also been reported in the study on temporal trend of OCs in northern fur seals from the Pacific Coast of Japan [84]. In contrast to the composition of DDT compounds, no significant difference in the proportion of α-HCH (~60%) was observed between the myctophids collected in 1976 and 1994. Such a small variation in the temporal trend of HCH isomer composition has also been reported in the northern fur seals [84]. It may be attributable to the global transport of HCHs to high-latitude regions and/or the water-mass inflow from the Okhotsk Sea. Further monitoring along the Kuroshio–Oyashio interfrontal zone including marginal seas such as the Okhotsk Sea with geophysical and biogeochemical studies is required to delineate the distributions and fate of anthropogenic contaminants.
4 POPs and Related Compounds in the East China Sea
In this section our recent studies in the ECS [42, 43] have been reviewed for understanding the distribution and the specific accumulation characteristics of organohalogen and butyltin compounds in various deep-sea organisms (e.g., deep-sea eels, sea perches, hairtails, argentines, grenadiers, splitfins, flounders, gurnards, myctophids, lantern sharks, dogfish sharks, skates, squids, shrimps, prawns, lobsters, crabs, sea anemones; see the above references for more details).
4.1 Contamination Status
Among the organohalogen compounds analyzed in deep-sea organisms from the ECS, the concentrations of DDTs were the highest and those of the other compounds were approximately in the order of PCBs > CHLs > HCB > HBCDs > PBDEs = HCHs [42, 43] (Table 3). Deep-sea fishes from the ECS had the highest concentrations of DDTs among the data reported so far from our group for the WNP region (Table 1). Rapid industrial development and population growth in coastal areas of China have resulted in significant environmental pollution and damage to the aquatic ecosystems. From the 1950s to the 1980s, DDT was widely used in agriculture in China before it was legally banned in 1983 [85]. In the last two decades, many agricultural lands in China have been developed for commercial uses, thus, accelerating the remobilization of previously buried insecticides/pesticides. Yuan et al. [86] reported that the large-scale usage of DDT in agricultural practices and the subsequent runoff into the waterways have resulted in the high levels of DDTs in the freshwater, estuarine, and marine environment of China. In the case of PCBs, the concentrations in deep-sea fishes from ECS were comparable to that of deep-sea fishes from Tosa Bay [15] and off-Tohoku, Japan [14, 41, 44], but were significantly lower than that of deep-sea fishes from Suruga Bay [12] and other locations of the world (Table 2). On the other hand, the concentrations of HCHs and HCB in deep-sea organisms from the ECS were about one or two orders of magnitude lower than those of DDTs and PCBs. HCH isomers are less lipophilic when compared to other OCs, and, thus, they have lower biomagnification factors in aquatic ecosystems [87]. Significant fluxes (i.e., volatilization) of more volatile POPs, HCHs and HCB, to the atmosphere in the waters of low-latitude regions [2,30, 64] may also be attributable to their less contamination in the ECS along the Kuroshio Current.
Unlike PCBs and DDT, which are largely a legacy of the past, BFRs such as PBDEs and HBCDs have been banned from usage since late 1990s–2000s or even currently used in various electronic devices, furniture and textiles. Being additive flame retardants they can leak out of the treated materials during the life cycle of the product, causing a continuous contamination of the environment even after the regulations on their production/application were implemented. Total concentrations of PBDEs and HBCDs in various deep-sea organisms from the ECS ranged from 0.31 to 57 ng/g lipid wt. and 0.15 to 210 ng/g lipid wt., respectively (Table 3). Reports on PBDEs and HBCDs in marine ecosystems and food webs, particularly for the deep-sea environment, are relatively scarce. The concentrations of PBDEs in deep-sea fishes from the ECS were higher than those from the WNP, off-Tohoku [44]. The detection of PBDEs and HBCDs in deep-sea organisms from the ECS indicates that these compounds are also transportable to the deep oceans, similar to that of other POPs. The sources of these BFRs in the ECS could be from the various manufacturing operations as well as e-waste recycling activities along the Chinese coast [88]. The recycling of e-wastes could mobilize these BFRs from the electronic components into the environment.
The contamination status of organohalogen compounds in the ECS is probably affected by the various hydrological processes, notably the Yangtze River discharge and the Kuroshio Current. The Yangtze River flowing through big cities and important agricultural areas with an annual discharge of more than 9.0 × 1011 m3 [89] is a significant source of anthropogenic pollutants from the terrestrial environment to the ECS. On the other hand, the Kuroshio, flowing to the northeast along the continental slope, is a unique source of oceanic materials of open-ocean origin to the shallow continental shelf [90].
Concentrations of butyltin compounds (BTs) and other organotin compounds including triphenyltin (TPT) were reported by Tanabe et al. [42]. BTs and TPT were detected in almost all deep-sea organisms analyzed (levels up to 62 ng/g wet wt in blackbelly lantern shark, Etmopterus lucifer, for ∑BT, and 40 ng/g wet wt in garden eel, Conger myriaster, for TPT). Although the magnitude of contamination by BTs in ECS seems to be relatively low comparing with that of the western North Pacific, off-Tohoku, the results indicate widespread contamination by organotin compounds including TPT in various deep-sea ecosystems around the East Asian region.
4.2 Species-Specific Accumulation and Composition of Organohalogen Compounds
The concentrations of most of the organohalogen compounds in deep-sea organisms from the ECS varied considerably between the various species analyzed. Factors like food web characteristics, habitat, and feeding strategies can contribute to the exposure of deep-sea organisms to organohalogen compounds. Higher concentrations of organohalogen compounds were found in fishes like garden eel (Conger myriaster), Japanese split fin (Synagrops japonicus), lantern shark (Etmopterus lucifer), dogfish shark (Squalus japonicus), sharpnose sevengill shark (Heptranchias perlo), and green eyes (Chlorophthalmus acutifrons). All these species are deep-sea species and feed at high trophic levels of the food web. Thus, the species-specific differences in the accumulation patterns of the organohalogen compounds observed are due to biomagnification and/or feeding habits of the organisms. In general, HCHs and HCB, which have lower molecular weights and are less lipophilic than the other organohalogen compounds, exhibited smaller variation in concentrations among species and trophic levels owing to their faster rates of attaining equilibrium with water concentrations [83, 91]. However, unicorn grenadier (Caelorinchus productus) showed very high levels of HCB (up to 290 ng/g lipid wt.). The other grenadier species from the ECS also showed elevated levels of HCB. High levels of HCB were also found in the same species from Tosa Bay, Japan [15], and off-Tohoku, Japan [41]. So far, it is not clear why grenadier species accumulate such high levels of HCB.
Among the DDT compounds, p,p'-DDE or p,p'-DDT were the predominant constituents in almost all the deep-sea organisms. The percentages of p,p'-DDT in shallow-water and deep-sea fishes, 37% and 40%, respectively, were higher than in the deep-sea fishes from Suruga Bay and Tosa Bay [12, 15] and other locations of the world such as the Mediterranean Sea [25, 26] and North Atlantic Ocean [20, 27], indicating that there is fresh input of DDT from China or other Southeast Asian countries to the ECS. Higher percentages of p,p'-DDT were also found in skipjack tuna (Katsuwonus pelamis) from the ECS [63]. In case of HCH composition, β-HCH was the dominant isomer in all the deep-sea organisms, and this may be because of its persistent nature. α-HCH which is the major isomer in technical HCH mixture was detected in most of the samples, but the percentage composition of α-HCH was far less when compared to deep-sea fishes from the WNP, off-Tohoku [14, 41, 44]. In spite of the usage of technical HCH mixture in China and nearby countries, the low levels of α-HCH may be due to the rapid volatilization of α-HCH in the lower latitudes [30].
Of the fourteen PBDE congeners analyzed, a total of ten congeners from tri- to deca-BDE were identified in the deep-sea organisms from the ECS. The congener profiles of PBDEs varied between the species and, in most cases, the congeners from tetra- to hexa-BDEs were predominant (Fig. 11). The higher brominated congeners from octa- to deca-BDE were detected in very few samples, suggesting the relatively low bioaccumulation potential as well as the low bioavailability of these congeners. Congener-specific biotransformation of PBDEs undoubtedly plays an important role in the bioaccumulation behavior of PBDEs. BDE 209 was occasionally present at concentrations above its detection limit. It is noteworthy that BDE 209 was the dominant congener in case of an echinoderm (Holothuroidea sp.) and in flatfish (Chascanopsetta lugubris), and this can be explained by their sediment-dwelling nature. BDE 209 is mainly associated with sediments and is a major congener in suspended particles and sediments and related environments. The low detection of BDE 209 in other deep-sea organisms could be due to either its large molecular size which lowers its uptake rates or its relatively rapid biotransformation [92]. However, the presence of BDE 209 in deep-sea organisms from the ECS indicates that this congener probably is capable of long-range transport and dispersal. Among the HBCD isomers, α- and γ-HBCDs were detected in all the samples, while β-isomer was below detection limits in most of the samples (Fig. 11). Similar to BDE 209, higher proportion of γ-HBCD was observed in sediment-dwelling organisms.
4.3 Trophic Magnification and Sources of Contaminants in the Food Web
As noted above in the section for the WNP, the quantification of both stable carbon and nitrogen isotope ratios can provide valuable information on the feeding ecology of biota and its potential influence on the trophic enrichment of contaminants. [43] examined δ15N and δ13C in deep-sea organisms from the ECS to assess trophic magnification of organohalogen contaminants in the food web of the ECS. The δ15N values in the deep-sea organisms from the ECS ranged from 7.18‰ to 14.17‰. No clear relationships were observed between δ15N (‰) and concentrations (both in wet and lipid wt) for all the contaminants. In the aquatic environment, the uptake of contaminants by biota usually includes direct partitioning from the surrounding environment and ingestion of contaminated dietary sources, whereas the loss processes include direct partitioning, biotransformation, growth dilution, maternal transfer to offspring, and egestion. Besides, intensive biological N2 fixation by cyanobacterium, Trichodesmium, which plays an important role of primary production in the ECS, can affect variation of δ15N in individual organisms and entire food web [93]. The large difference in nutrient and organic matter sources between the sampled locations and specimens can be plausible reasons for the lack of relationship between δ15N (‰) and concentrations of organohalogen compounds in the ECS food web, as will be explained later.
Generally, the terrigenous organic matter is depleted in 13C, while the marine organic matter is usually enriched in 13C [94, 95]. Since the δ13C of marine planktonic sources is different from those of most terrestrial organic material sources, the carbon isotopic composition can be used as a tracer to distinguish marine from terrigenous organics. In the present study, the δ13C values in the deep-sea organisms from the ECS ranged from −15.54‰ to −20.87‰. Zhou et al. [96] reported the mean δ13C values of suspended particulate matter (SPM) in the Changjiang Estuary to be −22.6‰. Therefore, the deep-sea organisms from the ECS with low δ13C (−20.87‰) could indicate the input of terrigenous materials to the ECS. In the present study, strong negative relationships were observed for log-normalized lipid weight concentrations and δ13C (‰) for all the analyzed organohalogen compounds (Fig. 12, data shown only for PBDEs). Thus, the negative relationships observed between the organohalogen concentrations and δ13C (‰) indicate that organisms showing the higher concentrations than the other species accumulate these compounds from terrigenous (coastal benthic) sources rather than oceanic (pelagic) organic matter. Such specific pattern also reflects that steep slope in the concentrations of nutrients, terrigenous materials, and, possibly, contaminants from the coastal front zone is affected by Yangtze River discharge to oceanic waters along the Kuroshio Current.
4.4 Distribution and Transport of POPs into Deep Waters
Apart from the species-specific accumulation of organohalogen compounds, the variation in the concentrations of the organohalogen compounds among the organisms could be related to the distribution of these compounds in the marine ecosystem. Even among the fishes showing similar feeding habits, considerable variations in the concentrations of organohalogen compounds were observed, which cannot be explained only by their feeding habits and food web magnification. To understand spatial and vertical distribution of the contaminants in the ECS, the fishes were grouped into two categories, shallow water (<200 m) and deep water (>200 m), based on their sampling depths. Interestingly, it was found that the concentrations of almost all the contaminants (except for PCBs) were significantly higher in fishes from greater depths than that from shallower depths (p < 0.05 for DDTs and HBCDs and p < 0.05 for CHLs, PBDEs, and HBCDs, Mann–Whitney U-test) (Fig. 13).
As it was mentioned before, the ECS shelf receives large amounts of freshwater, suspended matter, and nutrients with the Yangtze River discharge [62]. Furthermore, Gong et al. [97] reported that the ECS has a moderately high primary production (108–997 mg/m2/d with a mean value of 425 mg/m2/d). Thus, the high primary productivity and input of terrestrial materials with the Yangtze River discharge leads to an increase in the particulate matter. Kao et al. [61] tracked the export flux of organic matter from the shelf to the deep-sea by comparing carbon and nitrogen isotopic characteristics of sedimentary organic matter all over the ECS and the adjacent Okinawa Trough. The study found similar δ13C and δ15N values between sediments from the inner shelf near the China coast and from the Okinawa Trough, suggesting that the ECS shelf is a source of particulate matter to the deeper waters of the ECS.
In the marine environment, organohalogen compounds are either dissolved in the water phase or associated with suspended particulate matter or dissolved organic carbon depending on their hydrophobicity and organic carbon–water partition coefficients (KOC). The particulate matter, on which POPs sorb by the process of flocculation and aggregation to be eventually deposited into the deep waters, could be of natural origin, like detritus, fecal pellets, and living planktonic organisms, or from an anthropogenic source. Studies have shown that eutrophication results in large vertical fluxes of particle-associated pollutants and organic carbon to the deep waters [38, 52]. Thus, all the examined organohalogen compounds being hydrophobic are expected to have high affinity to particulate matter and get transported to deep waters. Volatilization of semi-volatile compounds like HCHs and HCB to the atmosphere from the surface waters of ECS due to the high temperatures in the subtropical region could also contribute to the vertical gradient of their concentrations. On the other hand, biological and ecological factors which can influence the results obtained from various organisms should be considered for further discussions on behavior of POPs. It is necessary to develop a useful model including key biological and ecological parameters for further “normalization” of the contaminant concentrations in organisms (instead of simple lipid normalization) as well as to use better “bioindicators” comprising of same/similar species for future research.
5 Conclusions and Perspectives
It is noteworthy that higher concentrations of POPs and related compounds in deeper waters were indicated by using bioindicators such as fishes in the WNP, off-Tohoku, and ECS, which have been documented to be high productive frontal zones around East Asia. Expansion of contamination by emerging POPs including PBDEs into the deep-sea ecosystems was also suggested by our research. Our recent ocean survey on POPs in sediments from the ECS reported significantly higher concentrations of DDTs and PCBs in deep-sea sediment from the Okinawa Trough than those in the shallower continental shelf [98]. These results suggest the vertical transport of POPs and related compounds in the high productive waters along the oceanic fronts and the potential role of deep-sea bed as a final sink and reservoir for these persistent contaminants. The accumulation and distribution of the POPs and related compounds observed in the marine ecosystems reflects their physicochemical properties such as vapor pressure and hydrophobicity as well as biological and ecological factors of organisms. In addition, riverine discharges, advection by oceanic currents, and formation of oceanic fronts are some examples of the physical transport processes that influence the redistribution of POPs. Further interdisciplinary approach including studies on biogeochemical and geophysical processes in the ocean as well as field observations are required to delineate the global and regional fate of POPs.
Mathematical models are one of indispensable tools to describe the chemodynamics of POPs, including the mass budget, half-life, and long-range transport potential. Models have been used to study the role of atmospheric transport in the multi-compartmental global fate and distribution of persistent substances. However, high-resolution global models that predict nonsteady dynamics of POPs in oceans need to be developed to understand the potential impact of POPs on ecosystems, human health, and their ultimate fate. In this regard, our group has developed a model called the Finely Advanced Transboundary Environmental model (FATE) [99, 100]. The FATE is a global high-resolution multi-compartment model that computes nonsteady POPs biogeochemical cycles in and across the five environmental compartments (atmosphere, ocean, vegetation, soil, and cryosphere). This model enables to predict the three-dimensional distributions of POPs as a result of advective transports, and to quantify the amount of PCB exports to the deep ocean by several physical and biogeochemical processes including phytoplankton detritus settling. Furthermore, we have developed a three-dimensional high-resolution transport model for regional scale POPs fate in the ECS [101], which will be linked and evaluated by our POP observations in the ECS for three consecutive years from 2008 to 2010 [98]. Using such state-of-the-art models, we were able to predict global and regional sinks and reservoirs of POPs which could provide new insights into our current understanding of the fate and transport of POPs.
Recently, we have also started looking at the chemical aftermath of the Tohoku earthquake (the Great Eastern Japan Earthquake) and tsunami that took place on March 11th, 2011 [102]. Three nuclear reactors at Fukushima Daiichi Nuclear Power Station in the tsunami area suffered hydrogen explosions, and leaked radioactive substances including 29.3 PBq of 137Cs were estimated to be released into the ocean [103]. These and other radioactive materials are now polluting the global environment, and it has been cautioned by many that these may accumulate in the biotic compartments and may ultimately reach human. This phenomenon needs the maximum attention of scientists working on the aftereffects of the Great Eastern Japan Earthquake. In addition, the tsunami had wiped out a strip of coast supporting a wide range of land uses and industries. Petro- and agrochemical plants, iron foundries, steel works, and automotive, electronics, food processing, paper, plastics, and pharmaceutical plants were among those that suffered damage. Numerous reports available indicate that various chemicals with potential environmental and health hazards may have been present at many facilities in heavily impacted locations and that old electrical equipment containing PCBs was washed away [104]. The complexity of the waste generated during the Great Eastern Japan Earthquake and tsunami may pose a serious threat of environmental pollution, which should also be monitored along with the effects of nuclear disaster. Our previous study [105] showed that the levels of 137Cs was the lowest in the species of marine mammals obtained from off-Tohoku, Japan, when compared with the specimens caught from other parts of the world such as Lake Baikal, Black Sea, North Sea, and Northern Canadian waters. As reviewed in this chapter, the data on POPs and related compounds in various organisms in the WNP, off-Tohoku, Japan, was also based on the observations in the region where the Great Eastern Japan Earthquake occurred. Thus, we would like to reiterate here that work on POPs and other emerging contaminants (e.g., brominated flame retardants, perfluorochemicals, pharmaceutical and personal care products, toxic elements) as well as radionuclides on the specimens from this region now can give an insight into the most discussed environmental problem in the area. The papers reviewed here can provide the baseline data for comparison for studying the possibility of build-up of the chemical contaminants in the marine ecosystems along northeastern Japan. We hope that our outcomes and future trials on the marine pollution can contribute to avoid any possible catastrophe by such chemicals around our country and the world.
References
Fent K (1996) Ecotoxicology of organotin compounds. Crit Rev Toxicol 26:1–117
Wania F, Mackay D (1996) Tracking the distribution of persistent organic pollutants. Environ Sci Technol 30:390A–396A
UNEP (2005) Ridding the World of POPs: a guide to the Stockholm convention on persistent organic pollutants. http://www.pops.int
UNEP (2009) Report of the Conference of the Parties of the Stockholm Convention on Persistent Organic Pollutants on the work of its fourth meeting. United Nations Environment Programme, 2009 UNEP/POPs/COP.4/38
Sixth meeting of the conference of the parties to the stockholm convention. http://chm.pops.int/TheConvention/ConferenceoftheParties/Meetings/COP6/tabid/3074/mctl/ViewDetails/EventModID/870/EventID/396/xmid/10240/Default.aspx. Accessed 20 Nov 2013
Barber RT, Warlen SM (1979) Organochlorine insecticide residues in deep sea fish from 2500 m in the Atlantic Ocean. Environ Sci Technol 13:1146–1148
Krämer W, Buchert H, Reuter U, Biscoito M, Maul DG, Le Grand G, Ballschmiter K (1984) Global baseline pollution studies IX: C6-C12 organochlorine compounds in surface-water and deep-sea fish from the eastern North Atlantic. Chemosphere 13:1255–1267
Stegeman JJ, Kloepper-Sams PJ, Farrington JW (1986) Monooxygenase induction and chlorobiphenyls in the deep-sea fish Coryphaenoides armatus. Science 231:1287–1289
Melzian B, Zoffmann C, Spies RB (1987) Chlorinated hydrocarbons in lower continental slope fish collected near the Farallon Islands, California. Mar Pollut Bull 18:388–393
Berg V, Ugland KI, Hareide NR, Aspholm PE, Polder A, Skaare JU (1997) Organochlorine contamination in deep-sea fish from Davis Strait. Mar Environ Res 44:135–148
Berg V, Polder A, Skaare JU (1998) Organochlorines in deep-sea fish from the Nordfjord. Chemosphere 38:275–282
Lee JS, Tanabe S, Takemoto N, Kubodera T (1997) Organochlorine residues in deep-sea organisms from Suruga Bay, Japan. Mar Pollut Bull 34:250–258
Takahashi S, Lee JS, Tanabe S, Kubodera T (1998) Contamination and specific accumulation of organochlorine and butyltin compounds in deep-sea organisms collected from Suruga Bay, Japan. Sci Total Environ 214:49–64
Takahashi S, Tanabe S, Kawaguchi K (2000) Organochlorine and butyltin residues in mesopelagic myctophid fishes from the western North Pacific. Environ Sci Technol 34:5129–5136
Takahashi S, Hayashi S, Kasai R, Tanabe S, Kubodera T (2001) Contamination of deep-sea organisms from Tosa Bay, Japan by organochlorine and butyltin compounds. In: Fujita T, Saito H, Takeda M (eds) Deep-sea Fauna and Pollutants in Tosa Bay. National Science Museum Monographs, vol 20. National Science Museum, Tokyo, pp 363–380
de Boer J, Wester PG, Klamer JC, Lewis WE, Boon JP (1998) Do flame retardants threaten ocean life? Nature 394:28–29
Looser R, Froescheis O, Cailliet GM, Jarman WM, Ballschmiter K (2000) The deep-sea as a final sink of semi volatile persistent organic pollutants? Part 11: organochlorine pesticides in surface and deep-sea dwelling fish of the North and South Atlantic and the Monterey Bay Canyon (California). Chemosphere 40:661–670
Froescheis O, Looser R, Cailliet GM, Jarman WM, Ballschmiter K (2000) The deep-sea as a final global sink of semivolatile persistent organic pollutants? Part 1: PCBs in surface and deep-sea dwelling fish of the North and South Atlantic and the Monterey Bay Canyon (California). Chemosphere 40:651–660
Solé M, Porte C, Albaigés J (2001) Hydrocarbons, PCBs and DDT in the NW Mediterranean deep-sea fish Mora moro. Deep-Sea Res 48:495–513
Mormede S, Davies IM (2003) Horizontal and vertical distribution of organic contaminants in deep-sea fish species. Chemosphere 50:563–574
Storelli MM, Storelli A, Addabbo RD, Barone G, Marcotrigiano GO (2004) Polychlorinated biphenyl residues in deep-sea fish from the Mediterranean Sea. Environ Intern 30:343–349
Ramu K, Kajiwara N, Mochizuki H, Miyasaka H, Asante KA, Takahashi S, Nishida S, Tanabe S (2006) Occurrence of organochlorine pesticides, polychlorinated biphenyls and polybrominated diphenyl ethers in deep-sea fishes from the Sulu Sea. Mar Pollut Bull 52:1827–1832
Covaci A, Losada S, Roosens L, Vetter W, Santos FJ, Neels H, Storelli A, Storelli MM (2008) Anthropogenic and naturally occurring organobrominated compounds in two deep-sea fish species from the Mediterranean Sea. Environ Sci Technol 42:8654–8660
Unger MA, Harvey E, Vadas GG, Vecchione M (2008) Persistent pollutants in nine species of deep-sea cephalopods. Mar Pollut Bull 56:1498–1500
Storelli MM, Perrone VG, Marcotrigiano GO (2007) Organochlorine contamination (PCBs and DDTs) in deep-sea fish from the Mediterranean Sea. Mar Pollut Bull 54:1962–1989
Storelli MM, Losada S, Marcotrigiano GO, Roosens L, Barone G, Neels H, Covaci A (2009) Polychlorinated biphenyl and organochlorine pesticide contamination signatures in deep-sea fish from the Mediterranean Sea. Environ Res 109:851–856
Lemaire B, Priede IG, Collins MA, Bailey DM, Schtickzelle N, Thomé JP, Rees JF (2010) Effects of organochlorines on cytochrome P450 activity and antioxidant enzymes in liver of roundnose grenadier Coryphaenoides rupestris. Aquat Biol 8:161–168
Castro Jiménez J, Rotllant G, Ábalos M, Parera J, Dachs J, Company JB, Calafat A, Abad E (2013) Accumulation of dioxins in deep-sea crustaceans, fish and sediments from a submarine canyon (NW Mediterranean). Prog Oceanogr 118:260–272. http://www.sciencedirect.com/science/article/pii/S0079661113001304
Palm A, Cousins I, Gustafsson Ö, Axelman J, Grunder K, Broman D, Brorström-Lundén E (2004) Evaluation of sequentially-coupled POP fluxes estimated from simultaneous measurements in multiple compartments of an air-water-sediment system. Environ Pollut 128:85–97
Iwata H, Tanabe S, Sakai N, Tatsukawa R (1993) Distribution of persistent organochlorines in the oceanic air and surface seawater and the role of ocean on their global transport and fate. Environ Sci Technol 27:1080–1098
Wania F, Axelman J, Broman D (1998) A review of processes involved in the exchange of persistent organic pollutants across the air-sea interface. Environ Pollut 102:3–23
Knezovich JP, Harrison FL, Wilhelm RG (1987) The bioavailability of sediment-sorbed organic chemicals: a review. Water Air Soil Pollut 32:233–245
Lohmann R, Breivik K, Dachs J, Muir D (2007) Global fate of POPs: current and future research directions. Environ Pollut 150:150–165
Tanabe S (2007) Contamination by persistent toxic substances in the Asia-Pacific region. Dev Environ Sci 7:773–817
Tanabe S, Ramu K, Isobe T, Takahashi S (2008) Brominated flame retardants in the environment of Asia-Pacific: an overview of spatial and temporal trends. J Environ Monit 10:188–197
Tanabe S, Ramu K (2010) Environmental Specimen Bank (es-BANK) of Ehime University, Japan – current status and future perspectives. In: Isobe T, Nomiyama K, Subramanian A, Tanabe S (eds) Interdisciplinary studies on environmental chemistry. Environmental specimen bank: exploring possibility of setting-up esbs in developing countries, vol 4. TERRAPUB, Tokyo, pp 1–5
Belkin IM (2002) Front. In: Nybakken JW, Broenkow WW, Vallier TL (eds) Interdisciplinary encyclopedia of marine sciences, vol 1. Grolier, Danbury, pp 433–436
Dachs J, Eisenreich SJ, Hoff RM (2000) Influence of eutrophication on air-water exchange, vertical fluxes, and phytoplankton concentrations of persistent organic pollutants. Environ Sci Technol 34:1095–1102
Dachs J, Lohmann R, Ockenden WA, Mejanelle L, Eisenreich SJ, Jones KC (2002) Oceanic biogeochemical controls on global dynamics of persistent organic pollutants. Environ Sci Technol 36:4229–4237
Tanabe S, Nishimura A, Hanaoka S, Yanagi T, Takeoka H, Tatsukawa R (1991) Persistent organochlorines in coastal fronts. Mar Pollut Bull 22:344–351
de Brito APX, Takahashi S, Ueno D, Iwata H, Tanabe S, Kubodera T (2002) Organochlorine and butyltin residues in deep-sea organisms collected from the western North Pacific, off-Tohoku, Japan. Mar Pollut Bull 45:348–361
Tanabe S, Ramu K, Mochizuki H, Miyasaka H, Okuda N, Muraoka M, Kajiwara N, Takahashi S, Kubodera T (2005) Contamination and distribution of persistent organochlorine and organotin compounds in deep-sea organisms from East China Sea. In: Hasegawa K, Shinohara G, Takeda M (eds) Deep-sea Fauna and Pollutants in Nansei Islands. National Science Museum Monographs, vol 29, National Science Museum, Tokyo, pp 453–476
Ramu K, Kajiwara N, Isobe T, Takahashi S, Miyasaka H, Kubodera T, Tanabe S (2007) Organohalogen compounds in deep sea organisms from East China Sea: levels, profiles and fate. Organohalogen Compd 69:437–440
Takahashi S, Oshihoi T, Ramu K, Isobe T, Ohmori K, Kubodera T, Tanabe S (2010) Organohalogen compounds in deep-sea fishes from the western North Pacific, off-Tohoku, Japan: contamination status and bioaccumulation profiles. Mar Pollut Bull 60:187–196
Shimizu Y, Yasuda I, Ito SI (2001) Distribution and circulation of the coastal oyashio intrusion. J Phys Oceanogr 31:1561–1578
Sorokin YI, Sorokin PY (1999) Production in the sea of Okhotsk. J Plankton Res 21:201–230
Nakatsuka T, Toda M, Kawamura K, Wakatsuchi M (2004) Dissolved and particulate organic carbon in the Sea of Okhotsk: transport from continental shelf to ocean interior. J Geophys Res 109:C09S14. doi:10.1029/2003JC001909
Nishioka J, Ono T, Saito H, Nakatsuka T, Takeda S, Yoshimura T, Suzuki K, Kuma K, Nakabayashi S, Tsumune D, Mitsudera H, Johnson WK, Tsuda A (2007) Iron supply to the western subarctic Pacific: importance of iron export from the Sea of Okhotsk. J Geophys Res 112, C10012. doi:10.1029/2006JC004055
Yasuda I, Okuda K, Shimizu Y (1996) Distribution and modification of North Pacific intermediate water in the Kuroshio–Oyashio interfrontal zone. J Phys Oceanogr 26:448–465
Yasuda I (1997) The origin of the North Pacific intermediate water. J Geophys Res 102:893–909
Honda MC, Imai K, Nojiri Y, Hoshi F, Sugawara T, Kusakabe M (2002) The biological pump in the northwestern North Pacific based on fluxes and major components of particulate matter obtained by sediment-trap experiments (1997–2000). Deep-Sea Res II 49:5595–5625
Jeff JD, Eisenreich SJ, Paterson MJ, Beaty KJ, Hecky R, Elser JJ (1999) Biogeochemical cycling of PCBs in lakes of variable trophic status: a paired-lake experiment. Limnol Oceanogr 44:889–902
Otosaka S, Noriki S (2000) REEs and Mn/Al ratio of settling particles: horizontal transport of particulate material in the northern Japan Trench. Mar Chem 72:329–342
Qin YS, Zhao YY, Chen LR, Zhao SL (eds) (1996) Geology of the East China Sea. Science Press, Beijing
Ichikawa H, Beardsley RC (2002) The current system in the Yellow and East China Seas. J Oceanogr 58:77–92
Hickox R, Belkin I, Cornillon P, Shan Z (2000) Climatology and seasonal variability of ocean fronts in the East China, Yellow and Bohai seas from satellite SST data. Geophys Res Lett 27:2945–2948
Chen CTA (2008) Chemical and physical fronts in the Bohai, Yellow and East China seas. J Mar Syst 78:394–410
Liu KK, Tang TY, Gong GC, Chen LY, Shiah FK (2000) Cross-shelf and along-shelf nutrient fluxes derived from flow fields and chemical hydrography observed in the southern East China Sea off northern Taiwan. Cont Shelf Res 20:493–523
Wong CM, Williams CE, Pittock J, Collier U, Schelle P (2007) World’s top 10 rivers at risk. WWF International, Gland, Switzerland, http://assets.panda.org/downloads/worldstop10riversatriskfinalmarch13.pdf. Accessed 20 July 2013
Li D, Daler D (2004) Ocean pollution from land-based sources: East China sea, China. Ambio 33:107–113
Kao SJ, Linb FJ, Liu KK (2003) Organic carbon and nitrogen contents and their isotopic compositions in surficial sediments from the East China Sea shelf and the southern Okinawa Trough. Deep-Sea Res II 50:1203–1217
Hung JJ, Chen CH, Gong GC, Sheu DD, Shiah FK (2003) Distributions, stoichiometric patterns and cross-shelf exports of dissolved organic matter in the East China Sea. Deep-Sea Res II 50:1127–1145
Ueno D, Takahashi S, Tanaka H, Subramanian A, Fillman G, Nakata H, Lam PKS, Zheng J, Muchtar M, Prudente M, Chung KH, Tanabe S (2003) Global pollution monitoring of PCBs and organochlorine pesticides using skipjack tuna as a bioindicator. Arch Environ Contam Toxicol 45:378–389
Beyer A, Mackay D, Matthies M, Wania F, Webster E (2000) Assessing long-range transport potential of persistent organic pollutants. Environ Sci Technol 34:699–703
Ueno D, Kajiwara N, Tanaka H, Subramanian A, Fillmann G, Lam PKS, Zheng GJ, Muchtar M, Razak H, Prudente M, Chung KH, Tanabe S (2004) Global pollution monitoring of polybrominated diphenyl ethers using skipjack tuna as a bioindicator. Environ Sci Technol 38:2312–2316
Ueno D, Alaee M, Marvin C, Muir DCG, Macinnis G, Reiner E, Crozier P, Furdui VI, Subramanian A, Fillmann G, Lam PKS, Zheng GJ, Muchtar M, Razak H, Prudente M, Chung KH, Tanabe S (2006) Distribution and transportability of hexabromocyclododecane (HBCD) in the Asia-Pacific region using skipjack tuna as a bioindicator. Environ Pollut 144:238–247
Watanabe I, Sakai S (2003) Environmental release and behavior of brominated flame retardants. Environ Intern 29:665–682
Caira JN, Benz GW, Borucinska J, Kohler NE (1997) Pugnose eels Simenchelys parasiticus (Synaphobranchidae) from the heart of a shortfin mako Isurus oxyrinchus (Lamnidae). Environ Biol Fish 49:139–144
Hobson KA, Welch HE (1992) Determination of trophic relationships within a high arctic marine food web using δ13C and δ15N analysis. Mar Ecol Prog Ser 84:9–18
Fisk AT, Hobson KA, Norstrom RJ (2001) Influence of chemical and biological factors on trophic transfer of persistent organic pollutants in the Northwater Polynya food web. Environ Sci Technol 35:732–738
Ruus A, Ugland KI, Skaare JU (2002) Influence of trophic position on organochlorine concentrations and composition patterns in a marine food web. Environ Toxicol Chem 21:2356–2364
France RL, Peters RH (1997) Ecosystem differences in the trophic enrichment of 13C in aquatic food webs. Can J Fish Aquat Sci 54:1255–1258
Hobson KA, Fisk A, Karnovsky N, Holst M, Gagnon JM, Fortier M (2002) A stable isotope (δ13C, δ15N) model for the North Water Polynya food web: implications for evaluating trophodynamics and the flow of energy and contaminants. Deep-Sea Res II 49:5131–5150
Takahashi S, Tanabe S, Kubodera T (1997) Butyltin residues in deep-sea organisms collected from Suruga Bay, Japan. Environ Sci Technol 31:3103–3109
Takahashi S, Mukai H, Tanabe S, Sakayama K, Miyazaki T, Masuno H (1999) Butyltin residues in livers of humans and wild terrestrial mammals and in plastic products. Environ Pollut 106:213–218
Tanabe S, Subramanian A (2006) Bioindicator of POPs – monitoring in developing countries. Kyoto University Press/Trans Pacific Press, Kyoto
Tanabe S, Tatsukawa R (1983) Vertical transport and residence time of chlorinated hydrocarbons in the open ocean water column. J Oceanogr Soc Jpn 39:53–62
Iwata H, Tanabe S, Ouchi E, Tatsukawa R (1995) Persistent organochlorines in air and water from east Siberia. In: Abstracts, second SETAC (Society of Environmental Toxicology and Chemistry) World Congress. SETAC, Vancouver, p 77
McConnell LL, Kucklick JR, Bidleman TF, Ivanov GP, Chernyak SM (1996) Air−water gas exchange of organochlorine compounds in Lake Baikal, Russia. Environ Sci Technol 30:2975–2983
Nakata H, Tanabe S, Tatsukawa R, Koyama Y, Miyazaki N, Belikov S, Boltunov A (1998) Persistent organochlorine contaminants in ringed seals (Phoca hispida) from the Kara sea, Russian Arctic. Environ Toxicol Chem 17:1745–1755
Iwata H, Watanabe M, Kim EY, Gotoh R, Yasunaga G, Tanabe S, Masuda Y, Fujita S (2000) Contamination chlorinated hydrocarbons and lead in Steller’s sea eagle and white-tailed sea eagle from Hokkaido, Japan. In: Ueta M, McGrady MJ (eds) First symposium on Steller’s and white-tailed sea eagles in East Asia. Wild Bird Society of Japan, Tokyo, pp 91–106
Ueno D, Inoue S, Ikeda K, Tanaka H, Yamada H, Tanabe S (2003) Specific accumulation of polychlorinated biphenyls and organochlorine pesticides in Japanese common squid as a bioindicator. Environ Pollut 125:227–235
Tanabe S, Tanaka H, Tatsukawa R (1984) Polychlorobiphenyls, DDT, and hexachlorocyclohexane isomers in the western North Pacific ecosystem. Arch Environ Contam Toxicol 13:731–738
Tanabe S, Sung JK, Choi DY, Baba N, Kiyota M, Yoshida K, Tatsukawa R (1994) Persistent organochlorine residues in northern fur seal from the Pacific coast of Japan since 1971. Environ Pollut 85:305–314
Chen W, Zhang L, Xu L, Wang X, Hong L, Hong H (2002) Residue levels of HCHs, DDTs and PCBs in shellfish from coastal areas of east Xiamen Island and Minjiang estuary, China. Mar Pollut Bull 45:385–390
Yuan D, Yang D, Wade TL, Qian Y (2001) Status of persistent organic pollutants in the sediment from several estuaries in China. Environ Pollut 114:101–111
Colombo JC, Bilos C, Campanaro M, Rodriguez Presa MJ, Catoggio JA (1995) Bioaccumulation of polychlorinated biphenyls and chlorinated pesticides by the Asiatic clam Corbicula fluminea: its use as sentinel organism in the Rio de La Plata Estuary, Argentina. Environ Sci Technol 29:914–927
Ramu K, Kajiwara N, Sudaryanto A, Isobe T, Takahashi S, Subramanian A, Ueno D, Zheng GJ, Lam PKS, Takada H, Zakaria MP, Viet PH, Prudente M, Tana TS, Tanabe S (2007) Asian mussel watch program: contamination status of polybrominated diphenyl ethers and organochlorines in coastal waters of Asian countries. Environ Sci Technol 41:4580–4586
Jiang JH, Wang SM (2004) Primary analysis of water resource, disasters and environment in the Changjiang River catchment. Quat Sci 24:512–517 (in Chinese)
Lie HJ, Cho CH (2002) Recent advances in understanding the circulation and hydrography of the East China Sea. Fish Oceanogr 11:318–328
Ellgehausen H, Guth JA, Esser HO (1980) Factors determining the bioaccumulation potential of pesticides in the individual compartments of aquatic food chains. Ecotoxicol Environ Saf 4:137–157
de Wit CA (2002) An overview of brominated flame retardants in the environment. Chemosphere 46:583–624
Minagawa M, Wada E (1986) Nitrogen isotope ratios of red tide organisms in the East China Sea: a characterization of biological nitrogen fixation. Mar Chem 19:245–259
Wada E, Minagawa M, Mizutani H, Tsuji T, Imaizumi R, Karasawa K (1987) Biogeochemical studies on the transport of organic matter along the Otsuchi River watershed, Japan. Estuar Coast Shelf Sci 25:321–336
Middelburg JJ, Nieuwenhuize J (1998) Carbon and nitrogen isotopes in suspended matter and sediments from the Scheldt Estuary. Mar Chem 60:217–225
Zhou J, Wu Y, Zhang J, Kang Q, Liu Z (2006) Carbon and nitrogen composition and stable isotope as potential indicators of source and fate of organic matter in the salt marsh of the Changjiang Estuary, China. Chemosphere 65:310–317
Gong GC, Wen YH, Wang BW, Liu GJ (2003) Seasonal variation of chlorophyll a concentration, primary production and environmental conditions in the subtropical East China Sea. Deep-Sea Res II 50:1219–1236
Watanabe N, Yamano S, Shimizu J, Takahashi S (2011) Distribution of persistent organic pollutants (POPs) in the East China Sea. Rep Hydrographic Oceanogr Res 47:72–82 (in Japanese)
Kawai T, Handoh IC, Takahashi S (2009) The rise of the Finely-Advanced Transboundary Environmental model (FATE): a state-of-the-art model prediction of the global sink of persistent organic pollutants. Organohalogen Compd 71:1610–1615
Kawai T, Handoh IC, Suzuki N (2011) The roles of marine phytoplankton and ocean circulation in determining the global fate of polychlorinated biphenyls. In: Omori K, Guo X, Yoshie N, Fujii N, Handoh IC, Isobe A, Tanabe S (eds) Interdisciplinary studies on environmental chemistry—marine environmental modeling & analysis. TERRAPUB, Tokyo, pp 169–178
Ono J, Takahashi D, Guo X, Takahashi S, Takeoka H (2011) Process study on numerical simulation for persistent organic pollutants in the East China Sea. In: Omori K, Guo X, Yoshie N, Fujii N, Handoh IC, Isobe A, Tanabe S (eds) interdisciplinary studies on environmental chemistry—marine environmental modeling & analysis. TERRAPUB, Tokyo, Japan, pp 197–204
Christoudias T, Lelieveld J (2013) Modelling the global atmospheric transport and deposition of radionuclides from the Fukushima Dai-ichi nuclear accident. Atmos Chem Phys 13:1425–1438
Tanabe S (2011) Great eastern japan earthquake – possible marine environmental contamination by toxic pollutants. Mar Pollut Bull 62:883–884
Bird WA, Grossman E (2011) Chemical aftermath: contamination and cleanup following the Tohoku earthquake and tsunami. Environ Health Perspect 119:A290–A301
Yoshitome R, Kunito T, Ikemoto T, Tanabe S, Zenke H, Yamauchi M, Miyazaki N (2003) Global distribution of radionuclides (137Cs and 40 K) in marine mammals. Environ Sci Technol 37:4597–4602
Acknowledgements
Our studies reviewed here were conducted as part of a joint research project entitled “Study on Deep-Sea Fauna and Conservation of Deep-Sea Ecosystem” between the Department of Zoology, the National Museum of Nature and Science, and the Center for Marine Environmental Studies, Ehime University. We thank the late Prof. Kouichi Kawaguchi, Dr. Masatoshi Moku, and the crew and staff members of the R/V Tansei-maru, Ocean Research Institute, The University of Tokyo; Dr. Tsunemi Kubodera, National Museum of Nature and Science; and the crew and staff members of the R/V Wakataka-maru, Fisheries Research Agency, for their help on the collection of valuable samples and information of deep-sea organisms. Our studies were also supported by Global Center of Excellence Program of the Japanese Ministry of Education, Culture, Sports, Science and Technology and Grants-in-Aid for Scientific Research (S) (No. 20221003) and (B) (21310043) from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Takahashi, S., Karri, R., Tanabe, S. (2014). Contamination by Persistent Organic Pollutants and Related Compounds in Deep-Sea Ecosystems Along Frontal Zones Around Japan. In: Belkin, I.M. (eds) Chemical Oceanography of Frontal Zones. The Handbook of Environmental Chemistry, vol 116. Springer, Berlin, Heidelberg. https://doi.org/10.1007/698_2013_252
Download citation
DOI: https://doi.org/10.1007/698_2013_252
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-65837-6
Online ISBN: 978-3-662-65839-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)