Abstract
Introduction
Little is known about the incidence and symptomatology of pineal cysts in children. Until now, the proper management of this group of patients has not been established.
Purpose
The purpose of this study was to evaluate the epidemiological and clinical features of pineal cysts in children and adolescents and to try to find guidelines for their management.
Methods and results
We analyzed 24 patients (17 girls, mean age 9, and 7 boys, mean age 14) with pineal cysts found as the only pathology on MRI. Six patients were treated surgically (excision of the cysts via a supracerebellar-infratentorial approach) because of the progression of neurological symptoms or the enlargement of the cyst at follow-up. In this group of patients, no surgery-related complications were noted, nor was residual cyst observed on postoperative MRI. In 4 cases, histological examination revealed simple cysts, but in 2 cases pineocytomas were diagnosed. Preoperative symptoms disappeared except light headache in 2 cases and in 1 case no improvement was obtained. The remaining 18 patients had a mean follow-up of 38 months (range 24–60 months). None of the cysts diminished or collapsed. We also measured the circadian pattern of melatonin secretion as well as β-HCG and AFP levels in serum before surgery. We found very high night levels of melatonin in both of the patients with pineocytomas, while the patients with pineal cysts showed normal or depressed melatonin secretion profile.
Conclusion
We concluded that though most pineal cysts were clinically benign they should be followed up for many years. If the cyst grows larger in follow-up MRI study and neurological symptoms are progressive, surgical treatment should be performed. In the authors' opinion, one of the markers discriminating benign and neoplastic lesions may be melatonin.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lesions of the pineal gland constitute 0.4–1.0% of all intracranial tumors [4]. They may be divided into four basic categories:
-
1.
Germ cell tumors
-
2.
Pineal parenchymal tumors
-
3.
Pineal interstitial cell tumors
-
4.
Cysts [21]
The pineal cysts occur in all ages, from the fetal period to senility, with predominance in adults in the fourth decade of life [2]. The natural history is unknown. They can be classified as neoplastic or benign as well as those lined by glial tissue, ependyma, or surrounded by the normal texture of the pineal parenchyma [10]. In the post-mortem examinations, the pineal cysts are found in 25–41% of otherwise normal pineal glands [30]. This high rate of occurrence is due to recording even the smallest lesions in this investigation [9].
In the MRI investigation, the rate of occurrence of the pineal cysts is much lower, ranging from 0.6% in children to 2.6% in adults [26]. Pineal cysts are diagnosed when the lesion is 8 mm long or greater in a single plane [4, 15]. They are well-circumscribed, homogenous, round formations that are only slightly hyperintense relative to CSF on T1-weighted images. On T2-weighted and proton density-weighted images they are hyperintense relative to CSF [27]. Kjos et al. connected these features with proteinaceous fluid content [14]. Some authors, however, classify lesions of 5 mm diameter as cysts, especially in cases in which the thin rim of enhancement is visible, after contrast material application [24]. This is probably the result of the undeveloped blood-brain barrier in the surrounding pineal tissue [7]. The smallest pineal cyst dimensions described in the literature are 2×2×2 mm [11].
In most cases, the pineal cysts are asymptomatic or the symptoms are nonspecific and poorly pronounced. In the adult population, the following sets of symptoms were isolated:
-
1.
Paroxysmal headache with gaze paresis
-
2.
Chronic headache, gaze paresis, papilledema, and hydrocephalus
-
3.
Pineal apoplexy with acute hydrocephalus [33]
The relationship between the pineal cysts and various symptoms in children has not been established.
At present, much effort is being devoted to defining clear principles of proceeding with pineal cysts, especially the development of suitable diagnostic procedures and clinical management. The differential diagnosis of simple cysts and neoplastic lesions with cystic components, such as astrocytoma, pineocytoma, or pineoblastoma, is of special importance [6, 19, 28]. Therefore, a suitable marker for cyst malignancy is sought.
The aim of this paper was the development of objective guidelines regarding the management of pineal cyst-like lesions in children and adolescents. For this purpose, the clinical and epidemiologic data were analyzed. The circadian pattern of melatonin secretion, as well as alpha-fetoprotein (AFP) and beta-chorionic gonadotropin (β-HCG) in serum were investigated in order to find their correlation with tumor malignancy.
Materials and methods
Twenty-four pediatric cases of pineal cysts larger than 5 mm, which were found to be the only pathology on MRI, were analyzed. There were 17 girls and 7 boys in the group studied. The age distribution of our patients is presented in Table 1.
Observations and results
Clinical symptoms, which were the reasons for performing MRI, are shown in Table 2.
Headache, vertigo, and visual disturbances were most commonly found. Parinaud's syndrome, which is a characteristic manifestation of the quadrigeminal plate compression, was found in only 2 patients.
In 13 patients the cyst caused disturbances in CSF flow through the aqueduct and gliosis at area around the aqueduct. However, in only 3 of these 13 cases we found mild to moderate widening of the ventricles. None of the patients presented hydrocephalus with radiological features of raised intracranial pressure.
The MR scans were also analyzed. The size of the cyst in anteroposterior and craniocaudal directions was measured on midline sagittal T1-weighted images. In 19 out of 24 cases the cyst's largest diameter ranged from 8 to 20 mm and in next 5 out of 24 cases the diameter was larger than 20 mm. No significant difference in cyst size was observed between boys and girls.
In 12 out of 24 cases enhancement of the cyst's wall with gadolinium was found. In the remaining 12 cases no reaction after gadolinium injection was obtained.
All the children have been followed-up with clinical examination repeated every 6 months and MRI performed once a year. The mean follow-up duration was 38 months, ranging from 24 to 60 months. The cyst diameters were stable in 23 out of 24 cases. In one child we revealed the progression of the cyst and she was treated surgically.
Six out of 24 children and adolescents were treated surgically. The mean age in this group was 11.5 years, ranging from 7 to 15 years. The surgical group consisted of 4 girls and 2 boys. The clinical manifestation was the same as in the whole group. We did not find any specific symptoms that allowed us to predict the necessity of surgery.
The diameter of the cysts was larger than 20 mm in 4 out of 6 cases. This means that all cases except one with cysts larger than 20 mm were treated surgically. Analyzing MRI enhancement of the cyst wall with gadolinium was also found in 4 out of 6 cases, with both cases of pineocytoma among them. MRI revealed ventricular dilatation (mild and moderate) in 2 cases.
All the patients were subjected to marker analysis. We determined the levels of AFP and β-HCG in plasma. In all cases we found normal levels of markers. We also determined the pattern of melatonin secretion asking if melatonin could be a marker of pineal cell tumors. Figure 1 shows the results obtained. Very high night levels of melatonin were found in both patients with pineocytomas, while the other patients showed normal or depressed melatonin secretion profiles.
The characteristics of the operated patients are presented in Table 3.
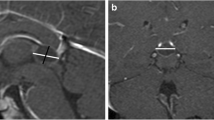
All patients were operated on using the infratentorial-supracerebellar approach. We did not experience any postoperative complications. Total cyst removal was obtained in all cases. One patient (case 2) was treated first by endoscopic fenestration of the cyst and later, because of the regrowth of the cyst, using the infratentorial-supracerebellar approach. This procedure allowed total removal of the cyst. The extent of cyst removal was confirmed and later followed up with MRI in all cases (Fig. 2). So far there has been no cyst regrowth in any of the cases, even in those diagnosed as pineocytomas. The mean follow-up duration in the "surgical group" was 31 months, range 11–40 months.
In 5 cases histological examination revealed simple cysts but in 2 cases it revealed pineocytomas. Both patients with pineocytomas were boys with no specific symptoms. At first, the diameter of the cysts was stable on follow-up MRI; however, in case 2 regrowth of the cyst was found after endoscopic fenestration. On MRI moderate ventricular dilatation was found in 1 case and enhancement with gadolinium of the cyst wall in both cases.
Analyzing the outcome, we found that 3 patients were free of symptoms after surgery, in the next 2 patients only light headache persisted, and in 1 case no improvement was obtained.
Discussion
The results presented in this paper demonstrate for the first time that large symptomatic pineal cysts are also found in children and young people under 18. Of special interest is the fact that as many as 5 children affected were under 7 years old.
In our opinion, the initially large pineal cysts in children constitute a certain development variant of this gland. According to Copper's hypothesis, they are produced by the sequestration of the pineal recess of the third ventricle [2]. The fact that some parts of the large pineal cysts described had an ependymal lining seems to corroborate this [23].
The ependymal lining probably degenerates over time due to distension; therefore, large pineal cysts are often found with a glial lining [31]. Parts of them even retain a connection with the ventricular system, which has been demonstrated to induce their further enlargement [12]. The rapid growth of a cyst is generally connected with certain clinical manifestation. It also creates a suspicion of malignant process. There is also the possibility of the presence of certain rare complications, none of which were found in the group of children analyzed by us. The bleeding to the cyst lumen [20], development of papilloma of the choroid plexus [27], rapid rupture of the cyst with resulting aseptic cerebrospinal meningitis [13], or even sudden death of the patient were described [18].
The MRI follow-up of the pineal cysts made by us demonstrated no significant differences in their size, except for one case. However, numerous other studies did indicate the increase in pineal cysts during the pubescence [24], even though there is a growth arrest of the pineal gland throughout childhood [25]. Therefore, most authorities advocate monitoring the pineal cysts for many years, especially in this period [26, 29, 33]. According to Fetell et al. if the lesion is enlarging, MRI follow-up examination should be performed every 2–3 months [6].
Large pineal cysts cause different constellations of symptoms [17]. For this reason, their diagnosis in children is not straightforward and differs from diagnosis in adults. In children, it is especially important to obtain an accurate medical history from the parents or custodians, as well as careful physical examination of the child. For instance, the headache, one of the most common symptoms of pineal cysts, are difficult to evaluate objectively even in adults, and—to make matters worse—headaches are found in 67–82% of 15-year children [16]. Comparing the clinical symptoms in children with symptoms described by other authors in adults, we did not notice any significant differences [5, 17, 31, 33]. Most often they were mild to moderate headaches, vertigo, visual disturbances, neurological symptoms, and in 2 cases, Parinaud's symptom.
We did not find hydrocephalus, which is relatively frequent in adult patients [17]. Some authors make speculations about the connection between hydrocephalus and headaches [6]. In our opinion, the headache is caused by the disturbances of the CSF flow through the cerebral aqueduct.
We have found signs of mass and disturbances of the CSF flow through the cerebral aqueduct, with the presence of gliosis (areas of elevated signal level in the vicinity of the cerebral aqueduct) in about half of the cases. According to another theory, the cause of the headaches is the pressure of the pineal cyst on the vein of Galen [29]. Perhaps the truth lies between the two and all hypotheses are true. This may be corroborated by the fact that removing the cyst alleviates the symptoms [6].
The imaging of cyst lesions within the pineal gland encounters many problems. On MRI, small cysts may be difficult to discriminate from calcification [15]. Calcification often accompanying the cyst does not show as characteristic signal areas on MR; therefore, for the small cysts found on MRI, comparative analysis with CT is needed [11].
Substantial difficulties still exist in the differential diagnosis of pineal cysts and malignant lesions. The diagnosis of nonneoplastic pineal cysts cannot be based on a single diagnostic criterion [7, 21, 29, 33]. For instance, it is not certain whether the MRI signal of the cystic component of a low-grade astrocytoma is always more hyperintense than pineal cysts [6]. Furthermore, according to Engel et al. signal characteristics of pineocytomas are very similar to glial pineal cysts [4]. Fleege et al. have proved that in spite of using double evaluation criteria (imaging and clinical), the incorrect neoplasm diagnosis was made in 14 out of 19 symptomatic pineal cysts [7].
In the opinion of Fetell et al., the evaluation of the protein content in the cyst fluid could enable the discrimination of such lesions [6]. Unfortunately, no authors have verified this hypothesis. Moreover, in normal conditions, after contrast material administration, the pineal gland tissue is enhanced [24, 26]. However, after the pressing of the parenchyma by the cyst, the effect may be weakened and as a result, this fact might mean interpretation as a neoplastic lesion [6]. In the group of children analyzed by us, the enhancing of the cyst wall was present in half of the cases.
The surgery was performed in 7 of our children in connection with the progress of symptoms or the increase in cyst observed on MRI. In 5 cases the ventricles were not or only slightly dilated and therefore an open cyst resection using the infratentorial supracerebellar approach was performed [8]. Alternative solutions are the occipital transtentorial, the transcallosal, and the transventricular approaches [33]. We did not experience any postoperative complications. In cases of pineal cysts with hydrocephalus, the most often used form of management is the endoscopic resection of the pineal cysts, stereotactically or free-hand [17]. Because in one of our children a significant widening of the ventricles had been observed, we first performed an endoscopic fenestration of the cyst. The rapid regrowth of the cyst induced us to the open classic surgery as above. The cyst has proven to be neoplastic.
In the histological examinations, a pineal cyst was diagnosed in 5 cases and a pineocytoma in 2. On the basis of our experience and the data of other authors, it is important for the confirmation of the diagnosis to provide the largest fragment of the lesion possible [6, 7, 11]. A macroscopic observation of the cyst during the surgery is also of great importance, especially the appearance of the fluid. In benign lesions the fluid is frequently clear, while in astrocytomas the liquid is brown [6]. In the material obtained by us, the content of the pineal cyst was light yellow, water-clear fluid, even in pineocytomas.
The markers of pineal tumors have been sought for many years. One of them is melatonin. However, there are contradictory reports in the world literature on this topic [1, 3, 22, 32].
Our investigation indicated a normal or only slightly depressed melatonin level in all analyzed children, except those with pineocytomas. In children with pineocytoma the increase of melatonin secretion at night was observed. We suppose that excessive secretion of melatonin may exist in true pineal tumors like pineocytomas, while in lesions causing destruction or compression of the pineal gland, low values of melatonin are found.
In conclusion, the authors suggest the observation of the pineal cysts for many years, even if the majority of them are clinically benign. If the cyst shows growth on successive MR images or a progression of the clinical symptoms is observed, surgery is indicated. Searching for other new markers of the neoplastic processes is also necessary to facilitate screening tests, formulation of diagnosis, and monitoring of the treatment. In the authors' opinion, one of the markers discriminating benign and neoplastic lesions may be melatonin.
References
Commentz JC, Helmke K (1995) Precocious puberty and decreased melatonin secretion due a hypothalamic hamartoma. Horm Res 44:271–275
Copper ERA (1944) Cystic hydrops of the pineal gland. J Nerv Ment Dis 99:552–572
Dempsey RJ, Chandler WF (1984) Abnormal serum melatonin levels in patients with intrasellar tumors. Neurosurgery 15:815–819
Engel U, Gottschalk S, Niehaus L, Lehmann R, May C, Vogel S, Janisch W (2000) Cystic lesions of the pineal region—MRI and pathology. Neuroradiology 42:399–402
Fain JS, Tomlinson FH, Scheithauer BW, Parisi JE, Fletcher GP, Kelly PJ, Miller GM (1994) Symptomatic glial cysts of the pineal gland. J Neurosurg 80:454–460
Fetell MR, Bruce JN, Burke AM, Cross DT, Torres RAA, Powers JM, Stein BM (1991) Non-neoplastic pineal cysts. Neurology 41:1034–1040
Fleege A, Miller GM, Fletcher GP, Fain JS, Scheithauer BW (1994) Benign glial cysts of the pineal gland: unusual imaging characteristic with histologic correlation. Am J Neuroradiol 15:161–167
Fukui M, Natori Y, Matsushima T, Nishio S, Ikezaki K (1998) Operative approaches to the pineal region tumors. Childs Nerv Syst 14:49–52
Hasegava A, Ohtsubo F, Mori W (1987) Pineal gland in old age; quantitative and qualitative morphological study of 168 human autopsy cases. Brain Res 33:113–11
Hirato J, Nakazato Y (2001) Pathology of pineal region tumors. J Neurooncology 54:239–249
Jinkins JR, Xiong L, Reiter RJ (1995) The midline pineal "eye": MR and CT characteristic of the pineal gland with and without benign cyst formation. J Pineal Res 19:64–71
Kang HS, Kim DG, Han DH (1998) Large glial cyst of the pineal gland: a possible growth mechanism. Case report. J Neurosurg 88:138–140
Kitayama J, Toyoda K, Fujii K, Ibayashi S, Sugimori H, Sadoshima S, Fujishima M (1996) Recurrent aseptic meningitis caused by rupture of a pineal cyst. No To Shinkei 48:1147–1150
Kjos BO, Brant-Zawadzki M, Kucharczyk W, Kelly WM, Norm D, Newton TH (1985) Cystic intracranial lesions: MR imaging. Radiology 155:363–369
Kluczewska E, Lorenz-Giec A, Bażowski P, Mandera M, Baron J, Zieliński Z (1999) Analiza i symtomatologia torbieli szyszynki w badaniach rezonansu magnetycznego. Neurol Neurochir Pol 33:1033–1043
Lewis DW, Ashwal S, Dahl G, Dorbad D, Hirtz D, Prensky A, Jarjour I (2002) Practice parameter: evaluation of children and adolescent with recurrent headaches. Neurology 59:490–499
Michielsen G, Benoit Y, Baert E, Meire F, Caemaert J (2002) Symptomatic pineal cysts: clinical manifestation and management. Acta Neurochir (Wien) 144:233–242
Milroy CM, Smith CL (1996) Sudden death due to a glial cyst of the pineal gland. J Clin Pathol 49:267–269
Momozaki N, Ikezaki K, Abe M, Fukui M, Fujii K, Kishikawa T (1992) Cystic pineocytoma—case report. Neurol Med Chir (Tokyo) 32:169–171
Mukherjee KK, Banerji D, Sharma R (1999) Pineal cyst presenting with intracystic and subarachnoid haemorrhage: report of a case and review of the literature. Br J Neurosurg 13:189–192
Neatherlin JS (1985) Pineal region brain tumors. J Neurosurg Nurs 17:349–354
Neuwelt EA, Lewy AJ (1983) Disappearance of plasma melatonin after removal of a neoplastic pineal gland. N Engl J Med 308:1132–1135
Russel DS, Rubinstein LJ (1977) Non-neoplastic cyst. In: Pathology of tumours of the nervous system. Arnold, London, p 295
Sawamura Y, Ikeda J, Ozawa M, Minoshima Y, Saito H, Abe H (1995) Magnetic resonances images reveal a high incidence of asymptomatic pineal cysts in young women. Neurosurgery 37:11–15
Schmidt F, Penka B, Trauner M, Reinsperger L, Ranner G, Ebner F, Waldhauser F (1995) Lack of pineal growth during childhood. J Clin Endocrinol Metab 80:1221–1223
Sener RN (1995) The pineal gland: a comparative MR imaging study in children and adults with respect to normal anatomical variations and pineal cysts. Pediatr Radiol 25:245–248
Steven DA, McGinn GJ, McClarty BM (1996) A choroid plexus papilloma arising from an incidental pineal cyst. Am J Neuroradiol 17:939–942
Sugiyama K, Arita K, Okamura T, Yamasaki F, Kajiwara Y, Ueda H, Kurisu K (2002) Detection of a pineoblastoma with large central cyst in young child. Childs Nerv Syst 18:157–160
Tamaki N, Shirataki K, Lin T, Masumura M, Katayama S, Matsumoto S (1989) Cysts of the pineal gland. A new clinical entity to be distinguished from tumors of the pineal region. Childs Nerv Syst 5:172–176
Tapp E (1979) The histology and pathology of the human pineal gland. In: Kappers JA, Pevet P (eds) The pineal gland of vertebrates including MA. Elsevier/North-Holland, Amsterdam, pp 481–500
Vaquero J, Martinez R, Escandon J, Bravo G (1988) Symptomatic glial cysts of the pineal gland. Surg Neurol 30:468–470
Vorkapic P, Waldhauser F, Bruckner R, Biegelmayer C, Schmidbauer M, Pendl G (1987) Serum melatonin level: a new neurodiagnostic tool in pineal region tumors? Neurosurgery 21:817–824
Wisoff JH, Epstein F (1992) Surgical management of symptomatic pineal cysts. J Neurosurg 77:896–900
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mandera, M., Marcol, W., Bierzyńska-Macyszyn, G. et al. Pineal cysts in childhood. Childs Nerv Syst 19, 750–755 (2003). https://doi.org/10.1007/s00381-003-0813-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-003-0813-2